Detection method for dissolution determination of flunarizine hydrochloride capsules
A technology of flunarizine hydrochloride and a detection method, which is applied in the field of analytical chemistry, can solve the problems of unsuitable flunarizine hydrochloride capsule dissolution, reduced column efficiency, shortened chromatographic column life and the like, and achieves low cost and high column efficiency. , the effect of reducing the test cost
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
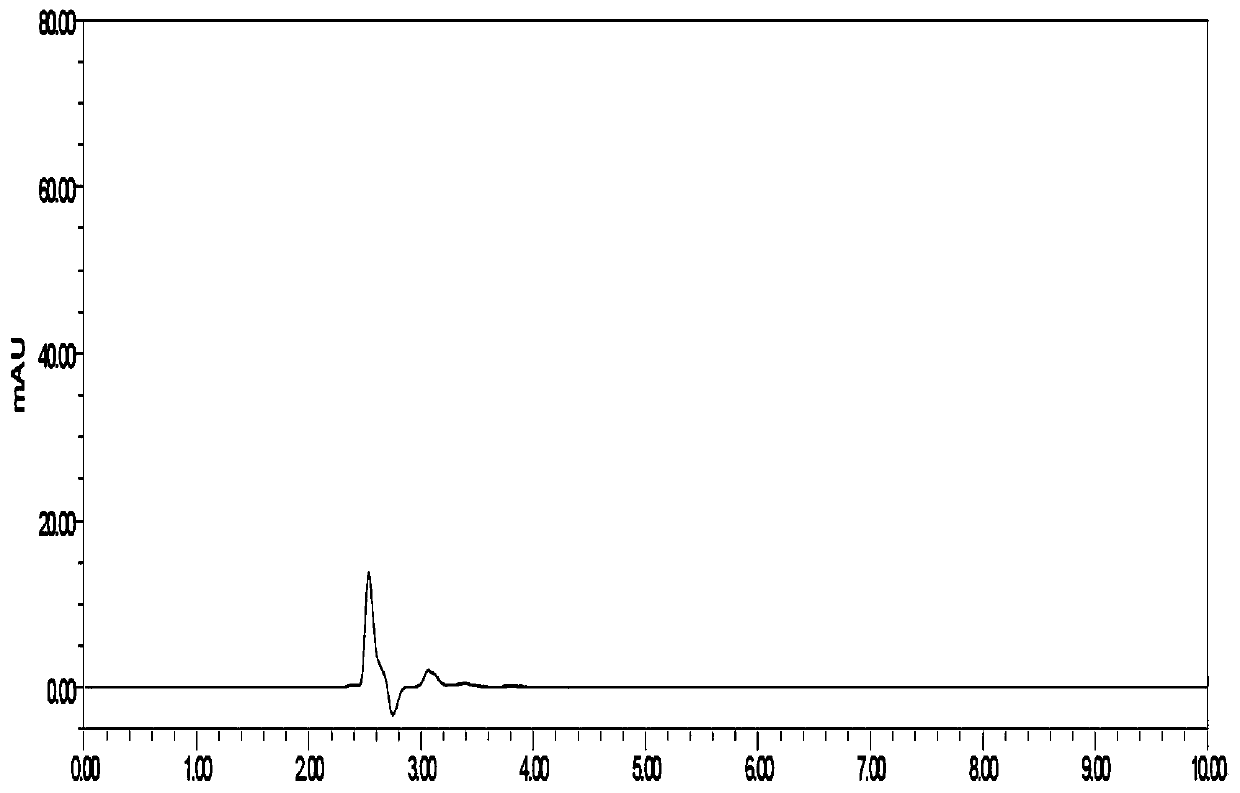
[0040] Embodiment 1: A kind of detection method of flunarizine hydrochloride capsule dissolution assay
[0041] Instruments and Conditions:
[0042] High performance liquid chromatography: DIONEXU3000;
[0043] Chromatographic column: YMC-PackODSC18 (4.6*250mm, 5μm);
[0044] Mobile phase: Methanol-phosphate buffer (take 1.20 g of sodium dihydrogen phosphate, add water to dissolve and dilute to 1000 ml, add 4 ml of triethylamine, adjust the pH to 3.5 with phosphoric acid) (75:25);
[0045] Column temperature: 35°C;
[0046] Flow rate: 1.0ml / min;
[0047] Detection wavelength: 253nm;
[0048] Injection volume: 20 μl.
[0049] Implementation steps:
[0050] (1) Preparation of dissolution medium: Weigh 2.00 g of SDS, 6.00 g of sodium dihydrogen phosphate (7.80 g of sodium dihydrogen phosphate dihydrate), 0.90 g of sodium hydroxide, and dilute with water to 1000 ml.
[0051](2) Take 10.89mg and 11.02mg of flunarizine hydrochloride as control, put them in a 100ml measuring b...
Embodiment 2
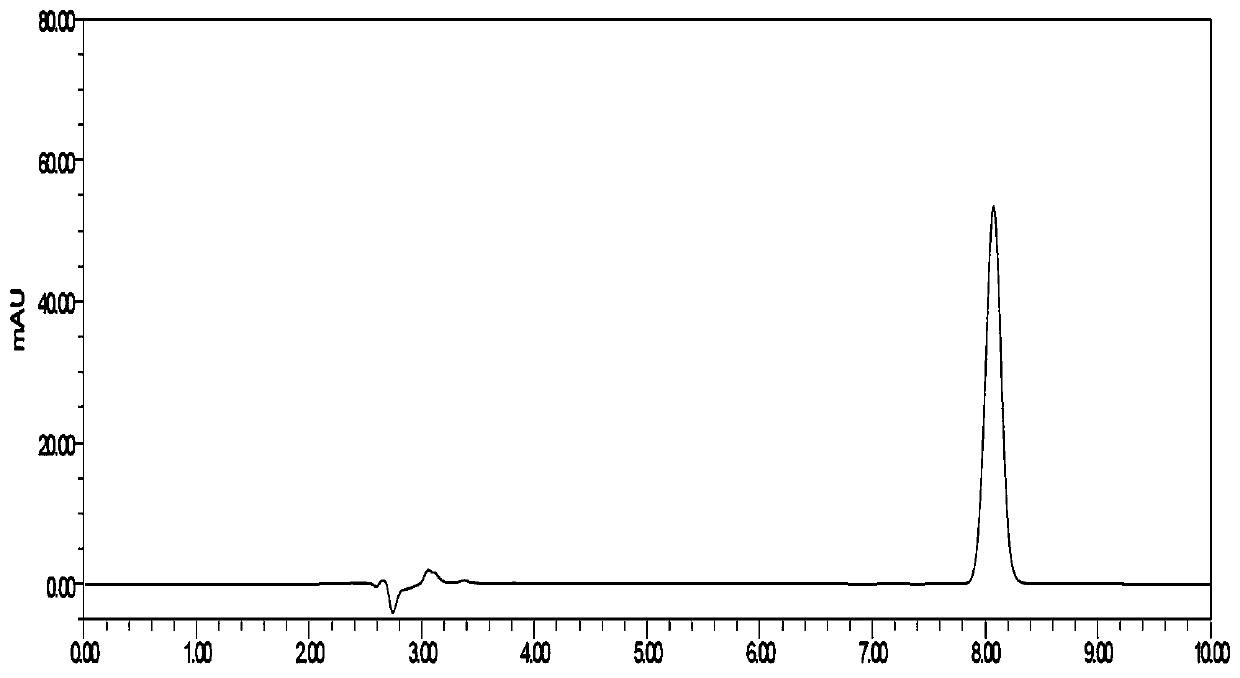
[0054] Embodiment 2: a kind of detection method of flunarizine hydrochloride capsule dissolution assay:
[0055] Instruments and Conditions:
[0056] High performance liquid chromatography: DIONEXU3000;
[0057] Chromatographic column: YMC-PackODSC18 (4.6*250mm, 5μm);
[0058] Mobile phase: Methanol-phosphate buffer (take 1.20 g of sodium dihydrogen phosphate, add water to dissolve and dilute to 1000 ml, add 4 ml of triethylamine, adjust the pH to 3.5 with phosphoric acid) (75:25);
[0059] Column temperature: 35°C;
[0060] Flow rate: 1.0ml / min;
[0061] Detection wavelength: 253nm;
[0062] Injection volume: 20 μl.
[0063] Implementation steps:
[0064] (1) Preparation of dissolution medium (with SDS added): Weigh 2.00 g of SDS, 6.00 g of sodium dihydrogen phosphate (7.80 g of sodium dihydrogen phosphate dihydrate), 0.90 g of sodium hydroxide, and dilute to 1000 ml with water.
[0065] (2) Preparation of dissolution medium (without SDS): Weigh 6.00 g of sodium dihydr...
Embodiment 3
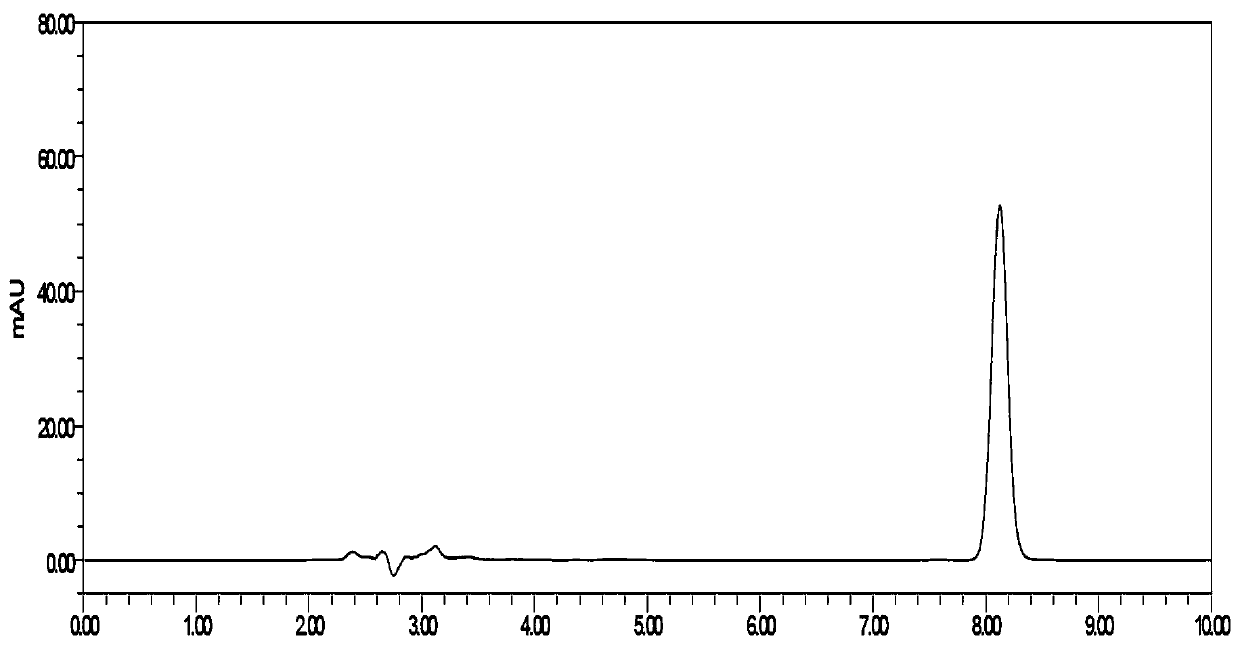
[0072] Embodiment 3: the detection method of a kind of flunarizine hydrochloride capsule dissolution assay in the prior art (Comparative case)
[0073] Instruments and Conditions:
[0074] High performance liquid chromatography: DIONEX U3000;
[0075] Chromatographic column: YMC-Pack ODS C18 (4.6*250mm, 5μm);
[0076] Mobile phase: methanol-phosphate buffer (take 1.36g of potassium dihydrogen phosphate, add water to dissolve and dilute to 1000ml, add 4ml of triethylamine, adjust the pH value to 3.5 with phosphoric acid) (75:25);
[0077] Column temperature: 35°C;
[0078] Flow rate: 1.0ml / min;
[0079] Detection wavelength: 253nm;
[0080] Injection volume: 20 μl.
[0081] Implementation steps:
[0082] (1) Preparation of dissolution medium: Weigh 2.00 g of SDS, 6.00 g of sodium dihydrogen phosphate (7.80 g of sodium dihydrogen phosphate dihydrate), 0.90 g of sodium hydroxide, and dilute with water to 1000 ml.
[0083] (2) Take 11.13mg and 11.35mg of flunarizine hydro...
PUM
| Property | Measurement | Unit |
|---|---|---|
| wavelength | aaaaa | aaaaa |
| wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More