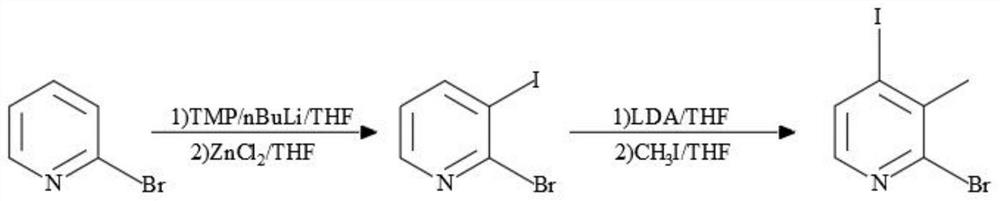
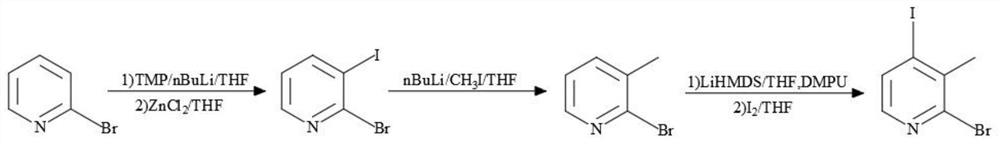
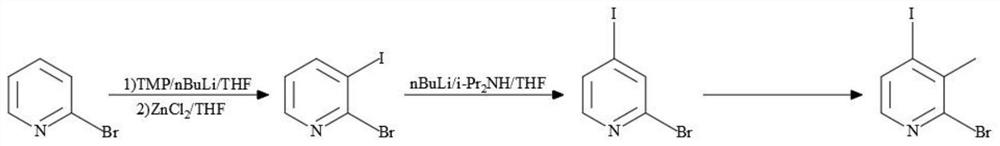
Synthesis process of 2-bromo-4-iodo-3-methylpyridine
A technology for the synthesis of picoline, which is applied in the field of synthesis of 2-bromo-4-iodo-3-picoline, can solve the problems of no literature reports in the synthesis, and achieve good product quality and cheap and easy-to-obtain raw materials , the effect of high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 11
[0031] Dissolve 2,2,6,6-tetramethylpiperidine (10.9g, 69mmol, 1.1eq) in tetrahydrofuran (40mL), replace the system with nitrogen three times, cool to below -20°C, and add n- Butyllithium (32mL, 69mmol, 1.1eq), there is a large amount of exothermic and degassing phenomenon during the dropping process, and the temperature control during the process does not exceed -20°C. After the dropwise addition, the temperature is controlled below -70°C for 1 hour. Bromopyridine (10.0g, 63mmol, 1.0eq), after the addition is complete, react at a temperature of -70~-60°C for 2h, then add 1M tetrahydrofuran solution of anhydrous zinc chloride (69mL, 69mmol, 1.1eq), continue to Reaction 1h. A mixed solution of iodine (24.0g, 95mmol, 1.5eq) and tetrahydrofuran (50mL) was added dropwise, and the reaction was continued for 0.5h. Monitoring shows that there are few remaining raw materials, stop the reaction, naturally rise to room temperature, add 100mL of saturated aqueous ammonium chloride soluti...
Embodiment 12
[0033] Dissolve 2,2,6,6-tetramethylpiperidine (49.2g, 348mmol, 1.1eq) in tetrahydrofuran (250mL), replace the system with nitrogen three times, cool to below -50°C, and add n- Butyllithium (158mL, 348mmol, 1.1eq), during the dropwise addition process, there is a large amount of exothermic and gas-releasing phenomena, the process temperature control does not exceed 0°C, and the temperature is controlled below 0°C for 0.5 hours, the system is cooled to below -70°C again, drop Add 2-bromopyridine (50.0g, 316mmol, 1.0eq), after the addition is complete, control the temperature at -70~-60°C for 2h, then add anhydrous magnesium chloride (33.1g, 348mmol, 1.1eq), continue the reaction for 0.5 h, generating a brown-green solid suspension. A mixed solution of iodine (80.3g, 316mmol, 1.0eq) and tetrahydrofuran (200mL, 4.0vol) was added dropwise, and the reaction was continued for 0.5h, turning into a red-black solution. Monitoring showed that there were few remaining raw materials, and ...
Embodiment 21
[0036] Dissolve 2-bromo-3-iodopyridine (10g, 35mmol, 1.0eq) in THF (100mL, 10vol), stir until completely dissolved, warm to -70°C, add dropwise n-BuLi n-hexane solution (14mL , 35mmol, 1.0eq), the dropwise addition was completed, stirred for 10min, then added dropwise methyl iodide (15g, 105mmol, 3.0eq), continued to stir for 30min, TLC showed that no raw material remained, raised the temperature to 0°C, and slowly added 50mL of saturated sulfurous acid dropwise Sodium hydrogen aqueous solution quenched the reaction, and the reaction system was naturally raised to room temperature, added ethyl acetate (100mL × 2) for extraction, combined organic phases, washed with 25% sodium chloride aqueous solution (100mL), and allowed to stand for liquid separation. The phase was concentrated under reduced pressure until no liquid emerged, and weighed to obtain 1.8 g of liquid, with a yield of 30%.
[0037]Dissolve 2-bromo-3-methylpyridine (10g, 58mmol, 1.0eq) in a mixture of tetrahydrofur...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More