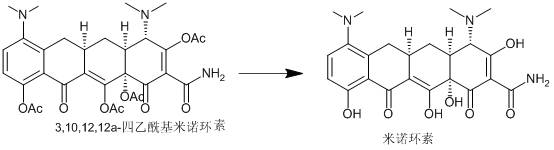
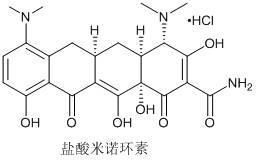
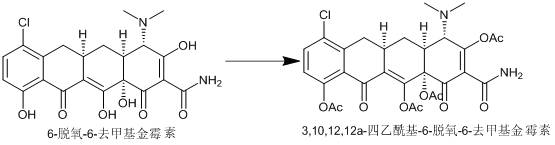
Synthesis method of minocycline hydrochloride
A technology of minocycline hydrochloride and its synthesis method, which is applied in the field of minocycline hydrochloride, can solve the problems of easy explosion in diazotization reaction, cumbersome operation, and difficulty in removal, so as to improve environmental friendliness and operability, Reduce environmental pollution and reduce the effect of hydrogenation times
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0039] In a stainless steel hydrogenation autoclave, add 20g of demethylaureomycin hydrochloride and 120ml of methanol, add 30g of p-toluenesulfonic acid, add 2g of Raney nickel, replace nitrogen three times, then flush with hydrogen to a pressure of 2Mpa, react at 40°C for 6h, and detect by HPLC The reaction is considered to be complete when the content of desmethylchlortetracycline in hydrochloric acid is ≤2%. Cool down to 0°C, add 300ml of water, add aqueous sodium bicarbonate solution to adjust the pH to about 8, and precipitate a solid, which can be obtained by suction filtration to obtain Intermediate I, a light yellow solid 17g, yield 88%, HPLC purity 97.5%. 1 H NMR (500 MHz, DMSO ) δ 7.53 (s, 1H), 7.31 (s, 2H), 6.84 (s, 1H), 3.68 (s, 1H), 3.28 (s, 1H), 3.07 – 2.76 (m, 3H), 2.61 (s, 1H), 2.26 (s, 6H), 1.74 (s, 2H).
Embodiment 2
[0041] In a stainless steel hydrogenation autoclave, add 20g of demethylaureomycin hydrochloride and 200ml of ethanol, add 15g of methanesulfonic acid, add 6g of Raney nickel, replace with nitrogen three times, then flush with hydrogen to a pressure of 3.5Mpa, react at 45°C for 8h, and detect by HPLC The reaction is considered to be complete when the content of desmethylchlortetracycline in hydrochloric acid is ≤2%. Cool down to 0°C, add 300ml of water, add aqueous sodium bicarbonate solution to adjust the pH to about 8, and precipitate a solid, which can be obtained by suction filtration to obtain Intermediate I, a light yellow solid 16.5g, yield 85%, HPLC purity 97.8%.
Embodiment 3
[0043]In a stainless steel hydrogenation autoclave, add 20g of demethylaureomycin hydrochloride and 280ml of methanol, add 15g of methanesulfonic acid, add 10g of Raney nickel, replace nitrogen three times, then flush with hydrogen to a pressure of 5Mpa, react at 50°C for 5h, and detect the reaction by HPLC The reaction is considered complete when the content of desmethylchlortetracycline hydrochloride is ≤2%. Cool down to 0°C, add 300ml of water, add aqueous sodium bicarbonate solution to adjust the pH to about 8, precipitate a solid, and obtain intermediate I by suction filtration, a light yellow solid of 18.2 g, yield 94%, HPLC purity 95.6%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More