[0022]Some embodiments of the present invention may also be useful as stabilizing elements or “molecular cytoskeletons” in the alignment of materials. For example, when combined with materials such as liquid crystals, polymeric networks as described herein may stabilize and / or accelerate the alignment of one or more liquid crystal phases. That is, the polymeric networks may serve as a structural reinforcing group to stabilize, for example, liquid crystals in a desired orientation, tilt angle, or
mesophase, in some cases, without need for an
external source of energy. This may be useful in the fabrication of bistable devices, such as
electronic paper, which may require materials to maintain a particular orientation in the absence of an
external energy source. Another
advantage of the present invention may be the ability to accelerate and / or enhance the switching events in devices, such as
liquid crystal devices. For example, many
liquid crystal devices involve the heterogeneous alignment of a liquid crystal, wherein, in the absence of an
external source of energy, the alignment of a liquid crystal sample in a particular orientation may begin at the interface between the liquid crystal and at least one surface (e.g., rubbed or treated surface), wherein the surface interacts with (e.g., orients) the liquid crystal at the surface, followed by propagation of the alignment through the bulk of the sample. In some cases, heterogeneous alignment may occur on a timescale that may be slow, relative to the desired rate of switching. In contrast, polymeric networks of the invention may be present throughout the bulk of the material and may facilitate homogeneous alignment of a liquid crystal, i.e., from a first orientation to a second orientation, which may occur more rapidly than heterogeneous alignment. Thus, devices of the invention may switch between different liquid crystal states with greater speed and uniformity, resulting in devices having faster response times and enhanced performance.
[0023]In some embodiments, the present invention provides compositions comprising polymeric networks comprising the
assembly of a plurality of
polymer molecules, wherein each
polymer molecule comprises at least one intermolecular interacting group at or near a terminal end of the
polymer molecule. The polymer molecules may be connected to an adjacent polymer molecule via the interacting group, wherein the interacting group forms a bond with an adjacent polymer. The bond may be non-
covalent bond or a
covalent bond. In some cases, the bond is a non-
covalent bond such as a
hydrogen bond, ionic bond, dative bond, or Van der Waals interaction. In some embodiments, the polymer molecules may be connected to an adjacent polymer molecule via a
hydrogen bond. In some embodiments, the polymer molecules may be connected to an adjacent polymer molecule via a covalent bond. The polymer molecules may assemble via the interacting groups to form a polymeric network having a greater
chain length than an individual polymer molecule
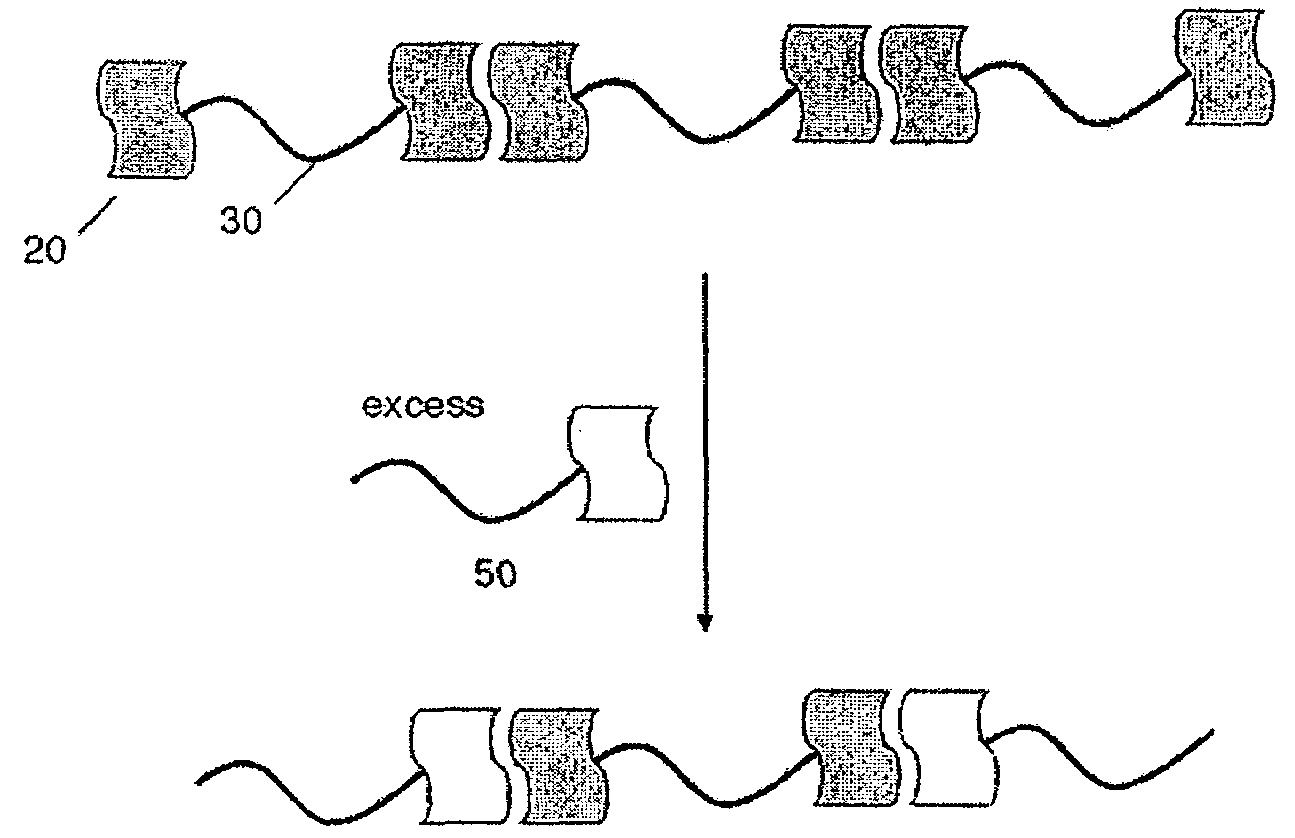
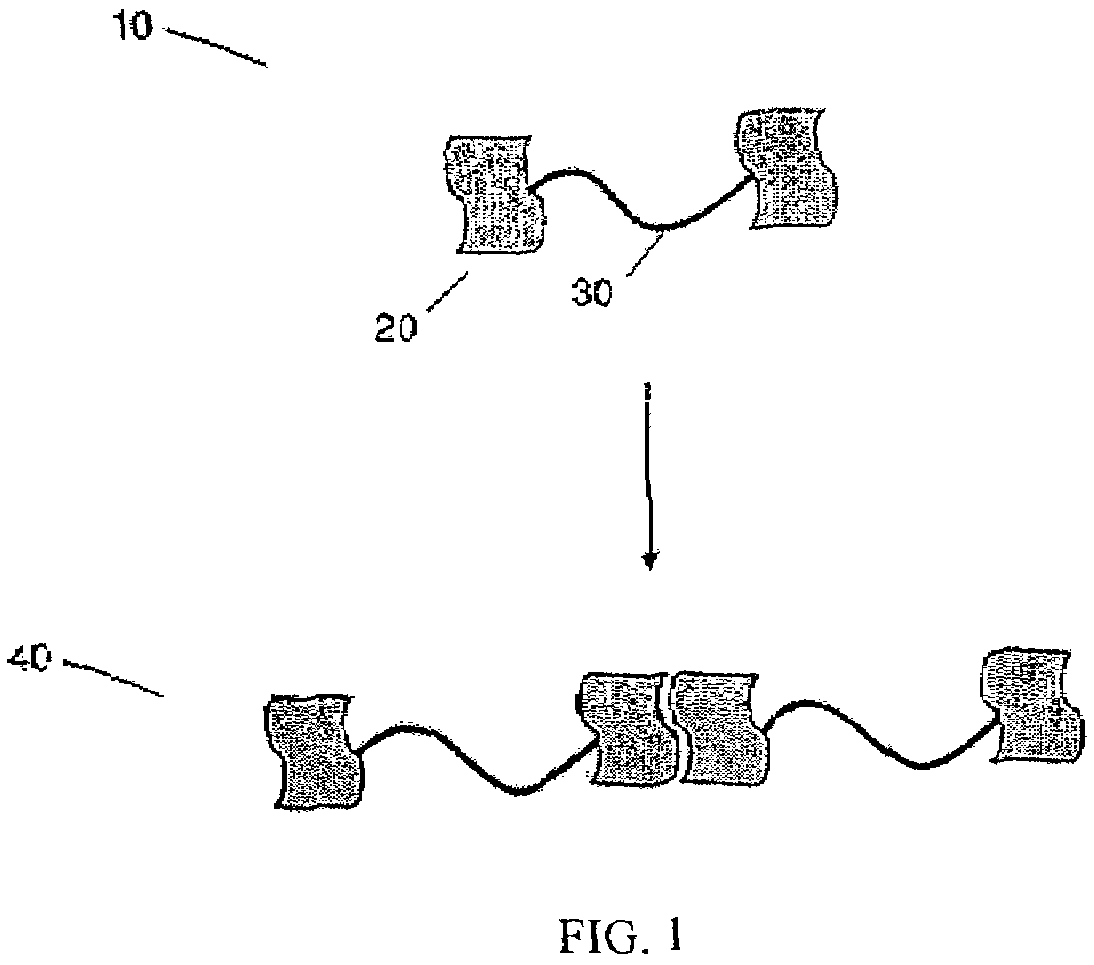
[0024]As shown schematically in FIG. 1, polymer molecule 10 may comprise interacting groups 20 attached to the terminal ends of polymer 20.
Polymer molecule 10 may then form a bond, such as a
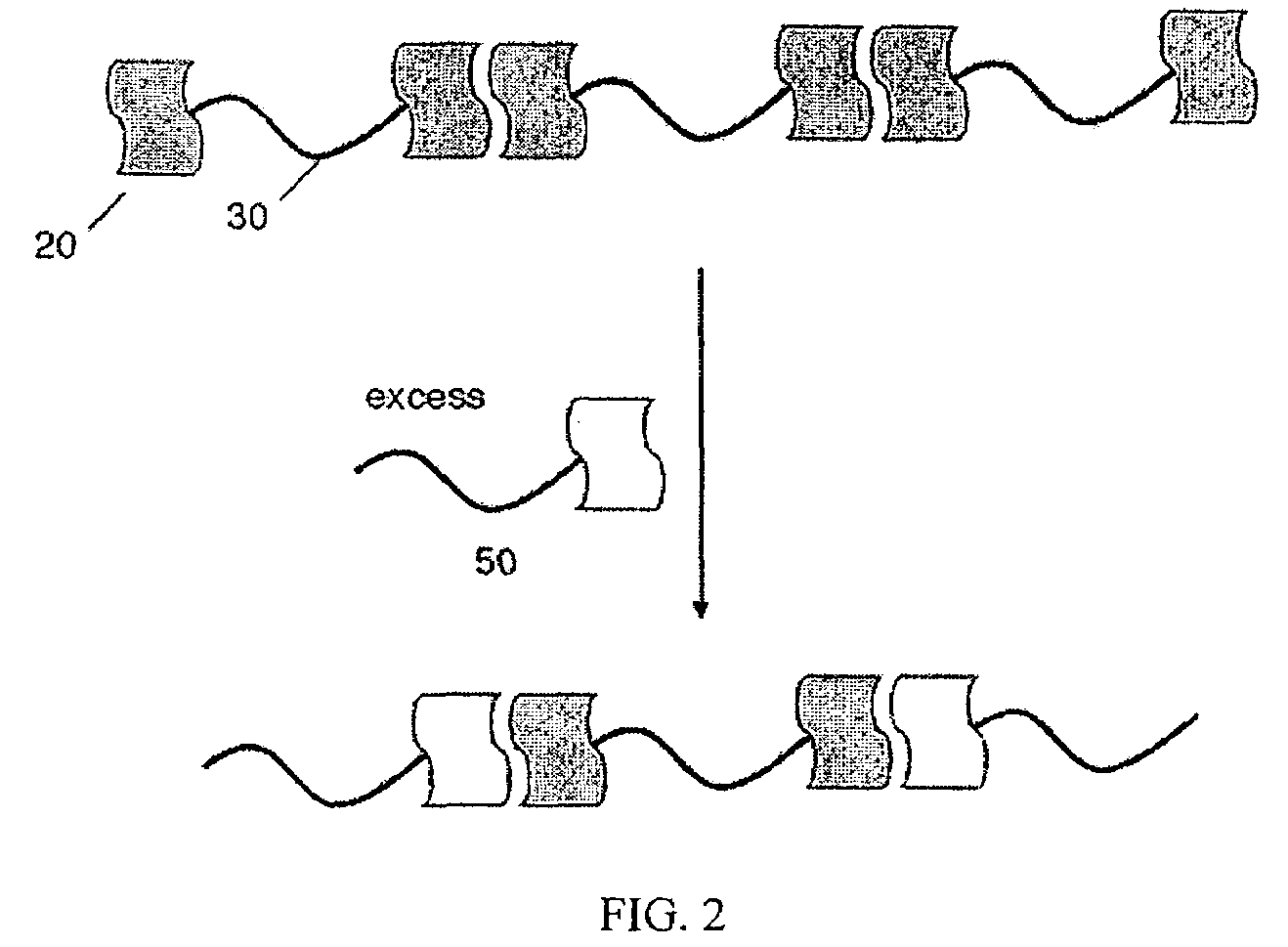
hydrogen-bond, for example, with an adjacent polymer molecule to form polymeric network 40. The polymer molecule may comprise interacting groups positioned at or near both terminal ends of the polymer molecule, allowing formation of a polymeric network that may extend throughout the bulk of a given volume and may have essentially infinite length within the volume. In some embodiments, at least 50% of the polymer molecules are connected to an adjacent polymer molecule via the at least one interacting group. In other embodiments, at least 60%, at least 70%, at least 80%, or, at least 90% of the polymer molecules are connected to an adjacent polymer molecule via the at least one interacting group. In some cases, formation of the polymeric network may be reversible. As shown schematically in FIG. 2, addition of an excess of a species 50 comprising a single interacting group may disrupt the polymeric network, wherein the individual polymer molecules interact with the species 50 rather than with an adjacent polymer molecule.
[0025]The polymeric network comprising the
assembly of a plurality of polymer molecules may advantageously have a greater dichroic ratio than a dichroic ratio of an individual polymer molecule that is essentially identical to an individual polymer molecule of the polymeric network, but lacking the interacting group, under essentially identical conditions. As used herein, the term “dichroic ratio” is given its ordinary meaning in the art and refers to the ratio of the polarized absorption of a species along two different axes of the species. The dichroic ratio may be defined by the formula,DR=(A∥−A⊥) / (A∥+A⊥),wherein A∥ is the absorption measured along a direction that is parallel to an expected alignment of a species, while A⊥ is the polarized absorption along a direction that is perpendicular to the expected alignment of the species. In some cases, A∥ refers to the alignment of a species along the
long axis of the species. The dichroic ratio of a material generally indicates the extent to which the material may be aligned along a particular direction, wherein a large dichroic ratio may indicate a high degree of alignment. In some embodiments, the dichroic ratio of the polymeric network is at least 10%, at least 20%, at least 30% at least 40%, at least 50%, at least 60%, or, at least 70% greater than the dichroic ratio of a polymer molecule that is essentially identical to an individual polymer molecule of the
polymer network, but lacking the interacting group, under essentially identical conditions. In some cases, the dichroic ratio may be increased by 80%, 90%, or even greater.
[0026]As used herein, a first species may be exposed to a set of “essentially identical conditions” as a second species by subjecting the first species and second species to a similar or substantially identical set of environmental parameters, such as temperature, pressure, pH,
solvent, concentration, chemical
reagent,
atmosphere (e.g.,
nitrogen,
argon,
oxygen, etc.),
electric field,
electromagnetic radiation, other source of
external energy, or the like, for a similar or identical period of time.
[0027]In some cases, compositions of the invention may further comprise a
host material with which the polymeric network is mixed. For example, the
host material may be a
solvent, polymer, gel, liquid crystal, other materials capable of forming anisotropic phases, or materials that are miscible with the polymeric network. In some embodiments, the
host material comprises a liquid crystal. In some cases, the polymeric network may be molecularly dissolved in the host material. As used herein, a “molecularly dissolved” polymeric network refers to a polymeric network that may be soluble within (e.g., does not phase-separate from) a host material while remaining an intact, polymeric network. That is, while the
polymer network may be soluble within the host material at the
molecular level, there may be little or no disruption in the assembly of the individual polymer molecules. In some cases, upon switching of the host material and
polymer network between different orientations, the polymer network may remain intact. In some cases, upon switching of the host material and polymer network between different orientations, the polymer network may undergo slight disruption but may then rapidly re-assemble such that the alignment of the host material is essentially unaffected by the slight disruption. Compositions comprising polymeric network mixed with and / or molecularly dissolved in host materials may have use in a wide variety of applications, as described more fully below.
 Login to View More
Login to View More