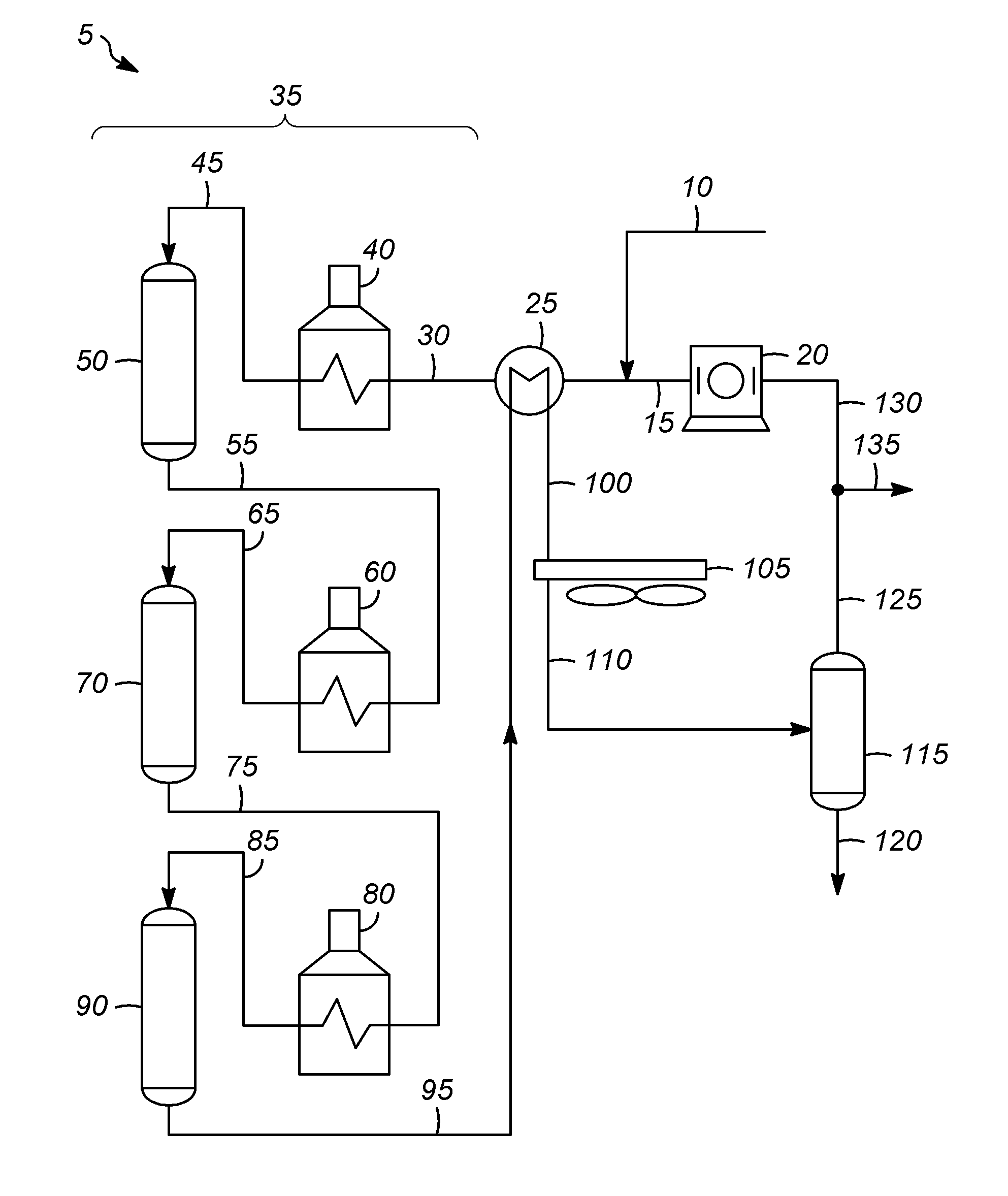
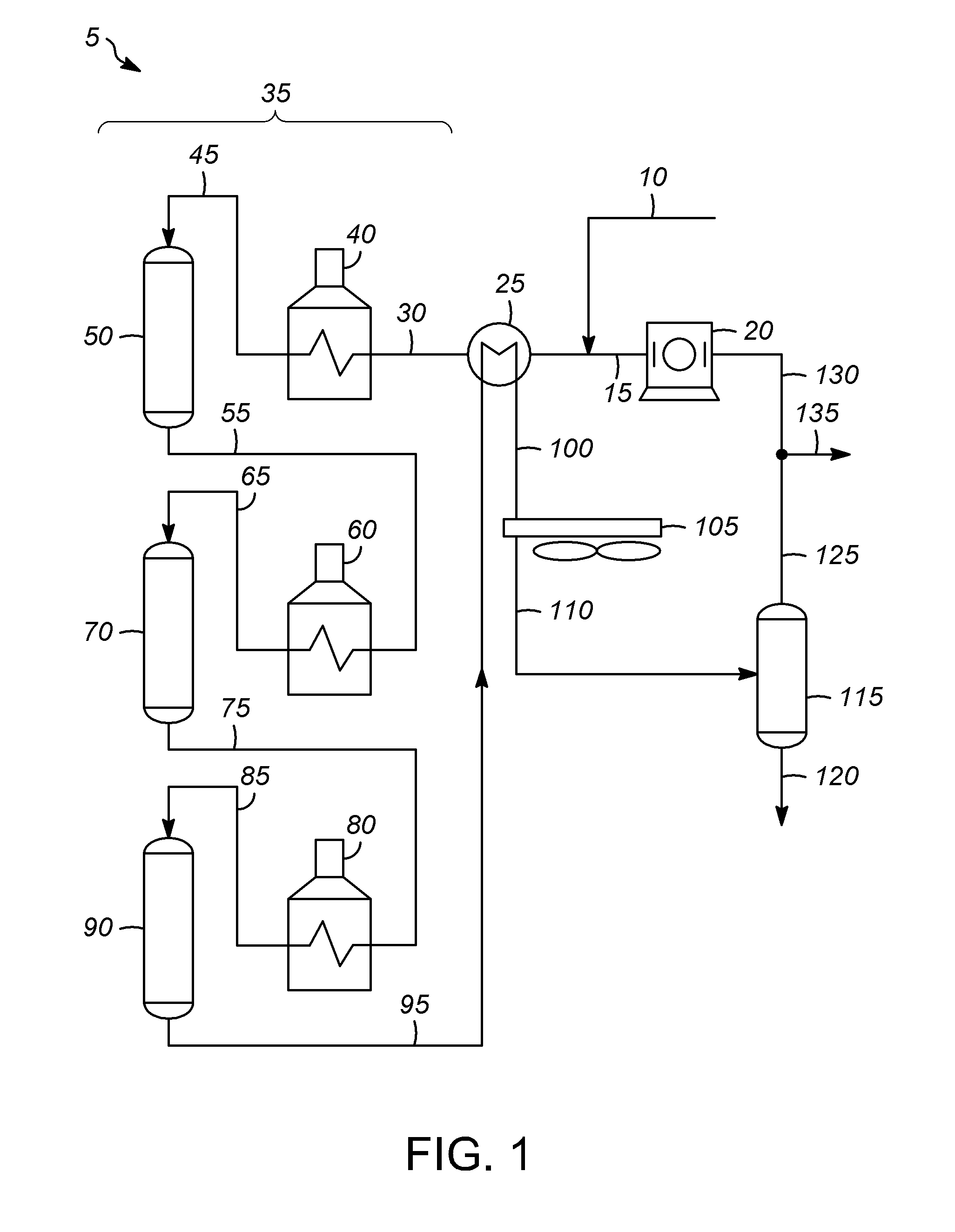
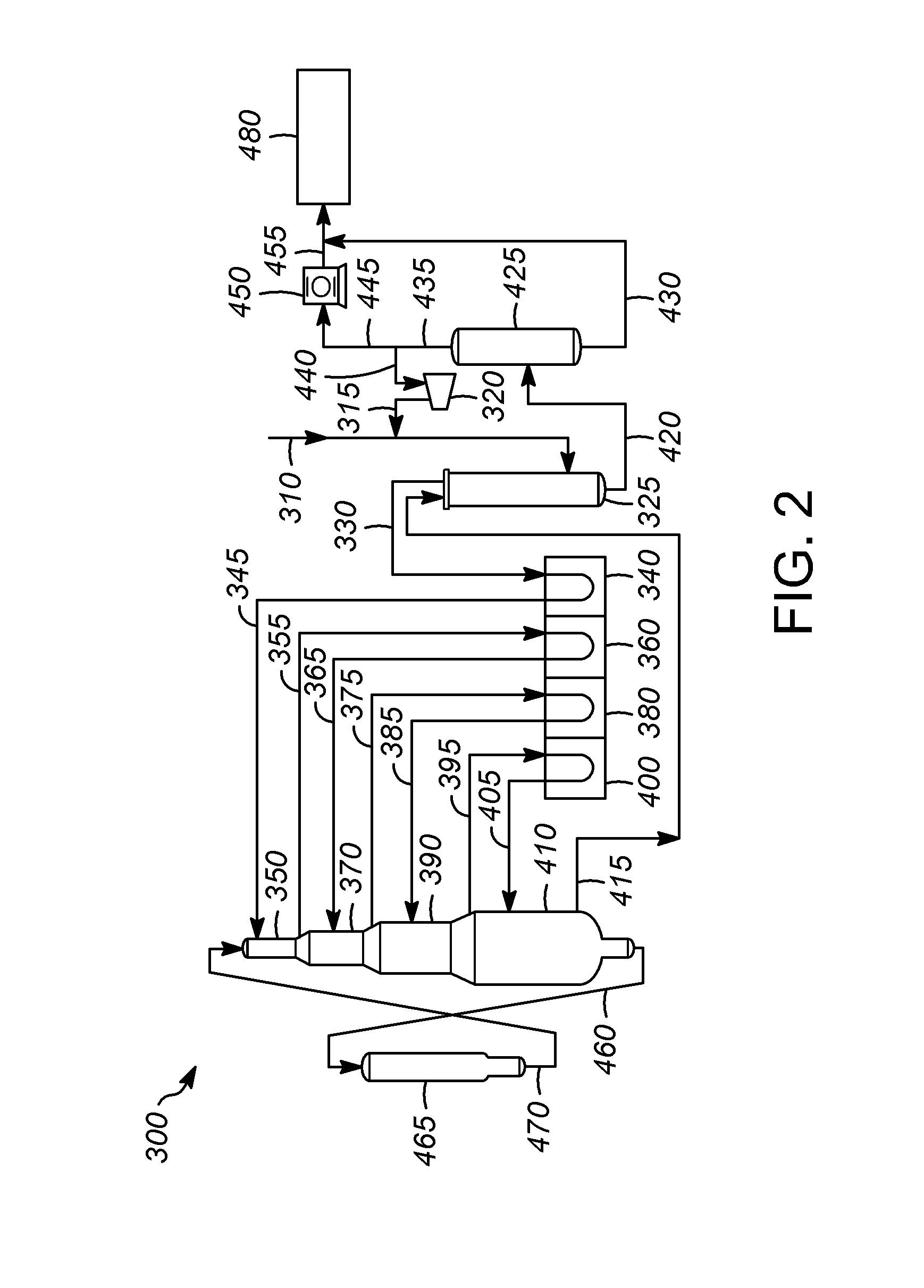
Many types of
hydrocarbon processing operations are carried out under relatively harsh operating conditions, including high temperatures and / or pressures and within various harsh chemical environments.
This and other
specialty austenitic stainless steels have been used in these applications but are susceptible to high temperature H2S,
sulfur, and
chloride-SCC
corrosion and high temperature
hydrogen attack issues that are present in these processes.
Many metals, including austenitic stainless steels, can be subject to a highly localized form of
corrosion known as stress-corrosion
cracking (SCC).
SCC often takes the form of branching cracks in apparently ductile material and can occur with little or no advance warning.
One particularly harsh environment in which austenitic stainless steels are typically observed to undergo stress corrosion
cracking is an environment containing halides, usually in the form of inorganic chlorides.
The presence of chlorides along with an aqueous phase and tensile stresses can result in chloride stress corrosion
cracking (“chloride-SCC”) of austenitic stainless steels.
In addition, while high temperatures may reduce the amount of time required for a particular chloride concentration to result in chloride-SCC, lower temperatures can cause chlorides to condense on surfaces, thereby increasing the concentration of the chlorides on the surfaces.
Thus, chloride-SCC can be problematic at many temperature ranges.
One particularly problematic area of chloride-SCC is in condensers where chloride condenses and concentrates on surfaces of the vessel.
The
precipitation of
chromium depletes the
chromium content adjacent to the grain boundaries, forming
chromium depleted zones and drastically reducing the corrosion and / or cracking resistance in corrosive environments in these zones.
The PTA can
attack the chromium depleted zones formed by
sensitization, causing corrosion and ultimately polythionic
acid stress corrosion cracking (PTA-SCC) where the vessel is put under tensile stresses either by being pressurized or by having residual stresses from, for example,
welding during fabrication.
Each of these processes is
time consuming and impractical during the operation of an
oil refinery complex because it requires additional materials and additional
downtime of the particular equipment to perform the purge or
neutralization steps.
Moreover, the catalyst in the reactor can be poisoned if trace levels of the chemicals remain, which is often the case.
However, such austenitic stainless steels are also susceptible to PTA-SCC as a result of
exposure to polythionic acid, since the operating conditions of many hydrocarbon treatment processes fall within the time at temperature at which
sensitization occurs.
Similarly, these materials are still susceptible to chloride-SCC through
exposure to chlorides,
oxygen, water, and stress at sufficient times and temperatures.
The need for special procedures during shutdown and startup of a
refinery complex affects not only costs, but also production time since they take a certain amount of time to carry out.
However, even with these precautions,
chloride corrosion of welded plate heat exchangers is often observed when
liquid water is present in the feed leading to chloride pitting, chloride pitting due to under-deposit corrosion, and when a water
dew-point occurs during regeneration of a
fixed bed reforming unit leading to chloride pitting, and chloride-SCC due to the presence of chlorides,
oxygen, and water.
Any failure due to corrosion or cracking will reduce product quality by contaminating the product
stream with the feed stream.
In addition, some welded plate heat exchangers are damaged by thermal stress which can cause mechanical damage to the
heat exchanger.
This type of mechanical damage accounts for the majority of all damage that causes bundle cross leakage.
Flow mal-distributions can occur when there is a
fouling of the bundle, when there is a sudden plugging of the bundle, when there is plugging of a
distributor, or when there are low velocities resulting in poor liquid flow up in the bundle.
Thermal stress damage that causes bundle cross leaking is costly to the
end user because it results in cross-leakage of the higher pressure stream into the lower pressure stream resulting in a reduction in
heat transfer efficiency, as well as potentially contaminating a product stream with a feed stream.
In corrosion and SCC cases, the bundles cannot be repaired because the damage typically occurs to most of or all the plate channels, requiring replacement of the bundle or the entire
heat exchanger.
However, when 10-20% or more of the channels are plugged, the resulting increase in pressure-drop can significantly reduce
throughput.
Furthermore, repaired bundles can be more susceptible to further damage by thermal stresses.
 Login to View More
Login to View More