Composition for treating diabetes comprising long-acting insulin conjugate and long-acting insulinotropic peptide conjugate
a technology of insulinotropic peptides and conjugates, which is applied in the direction of peptide/protein ingredients, drug compositions, metabolic disorders, etc., can solve the problems of increasing the risk of cardiovascular disease and insulin resistance, aversion to needles, and difficulty in handling injection devices, so as to improve the duration of in-vivo efficacy and stability, improve the effect of diabetes treatment, and stabilize the blood glucose control
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
on of Long-Acting Insulin Conjugate
[0091]For PEGylation of the N-terminus of immunoglobulin Fc region with 5K PropionALD(3) PEG (PEG with three propylaldehyde groups, NOF, Japan), the immunoglobulin Fc region and PEG were reacted at a molar ratio of 1:2 with the immunoglobulin Fc concentration of 10 mg / mL at 4° C. for 4.5 h. At this time, the reaction was conducted in a 100 mM potassium phosphate (pH 6.0), and a reducing agent, 20 mM SCB (NaCNBH3) was added thereto. The mono-PEGylated immunoglobulin Fc was purified from the reaction mixture using a SOURCE 15Q (GE Healthcare) purification column.
[0092]In order to prepare an insulin-PEG-immunoglobulin Fc conjugate having 90% or more modification of phenylalanine (B1F) at position 1 of the beta chain of insulin, the mono-PEGylated immunoglobulin Fc and insulin were prepared at a molar ratio of 4:1 and at the total protein concentration of 20 mg / mL, and reacted with each other at 4° C. for 20 h. 100 mM potassium phosphate (pH 6.0) was u...
example 2
on of Long-Acting Exendin-4 Conjugate
[0093]3.4 k PropionALD (2) PEG was reacted with the lysine (Lys) of CAexendin-4 using imidazo-acetyl exendin-4 (CAexendin-4, AP, USA). Among the two Lys isomer peaks, the last isomer peak (positional isomer of Lys27), which has more reaction and which is easily distinguishable from the N-terminal isomer peaks, was used for the coupling reaction.
[0094]The reaction was performed at a molar ratio of peptide:immunoglobulin Fc of 1:8, and a total concentration of proteins of 60 mg / mL at 4° C. for 20 h. The reaction was performed in a solution of 100 mM K-P (pH 6.0), and 20 mM SCB was added as a reducing agent. The coupling reaction solution was purified through two purification columns. First, SOURCE Q (XK 16 mL, Amersham Biosciences) was used to remove a large amount of immunoglobulin Fc which had not participated in the coupling reaction. Using 20 mM Tris (pH 7.5) and 1 M NaCl with salt gradients, the immunoglobulin Fc having relatively weak binding...
example 3
Test on Concurrent Administration of Long-Acting Insulin Conjugate and Long-Acting Exendin-4 Conjugate in STZ (Streptozotocin)-Induced Diabetes
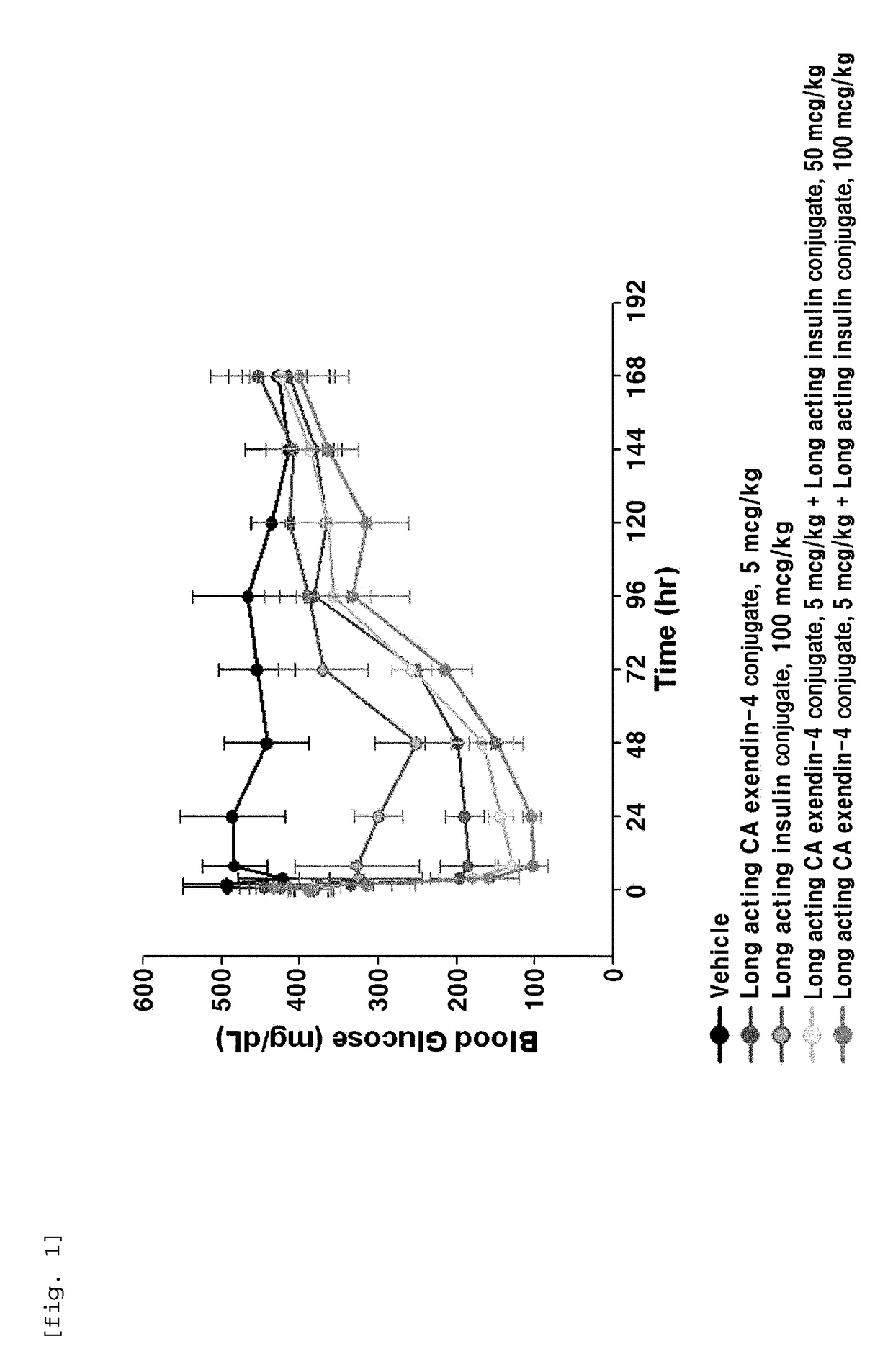
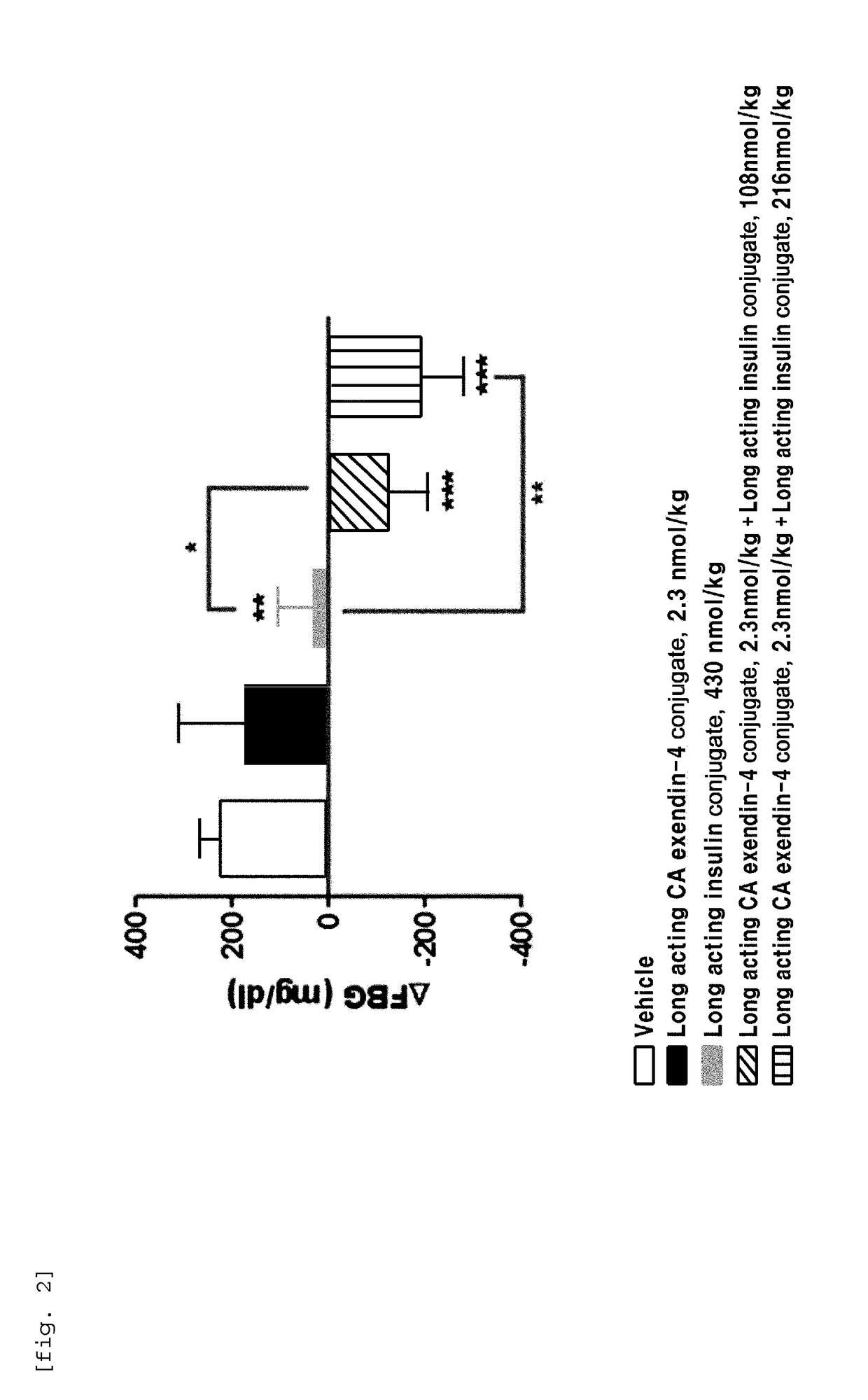
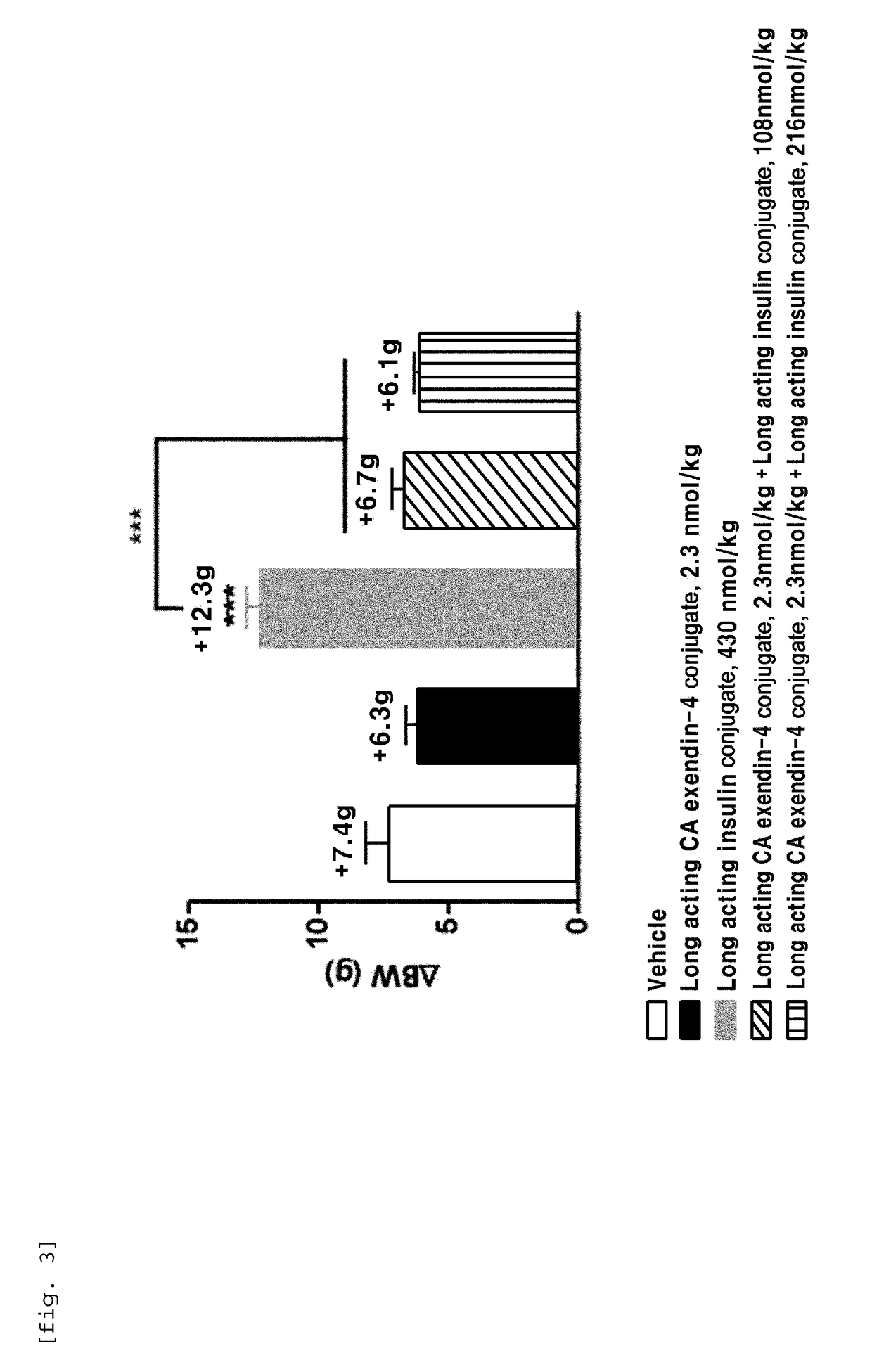
[0101]In order to perform in vivo efficacy test on administration of the compositions including the long-acting insulin conjugate and the long-acting exendin-4 conjugate prepared in Examples 1 and 2 or concurrent administration of the long-acting insulin conjugate and the long-acting exendin-4 conjugate, changes in the blood glucose levels were examined using STZ (Streptozotocin)-induced diabetic mice.
[0102]Diabetes was induced in 7-week old C57BL / 6 mice fasted for 16 h by intraperitoneal injection of STZ (50 mg / kg, 1 mg / mL) in 10 mM citrate buffer (pH 4.5) for 5 consecutive days. After 2 days, 1-2 drops of blood was taken from the tail vein using a 26 G syringe, and the blood glucose was measured using a glucometer (OneTouch Ultra, LifeScan, Inc., USA). Diabetes induction was determined by the measured blood glucose (350-600 mg / dL).
[0103]Dia...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com