Meclofenoxate hydrochloride microcapsule and method for preparing injection thereof
A technology of meclofenxate hydrochloride and microcapsules, which are applied in the field of meclofenxate hydrochloride freeze-dried preparations and their preparation, can solve the problems of unsatisfactory effect, high cost, limited wide application and the like, and improve bioavailability. , slow release, reduce the effect of hydrolysis reaction
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
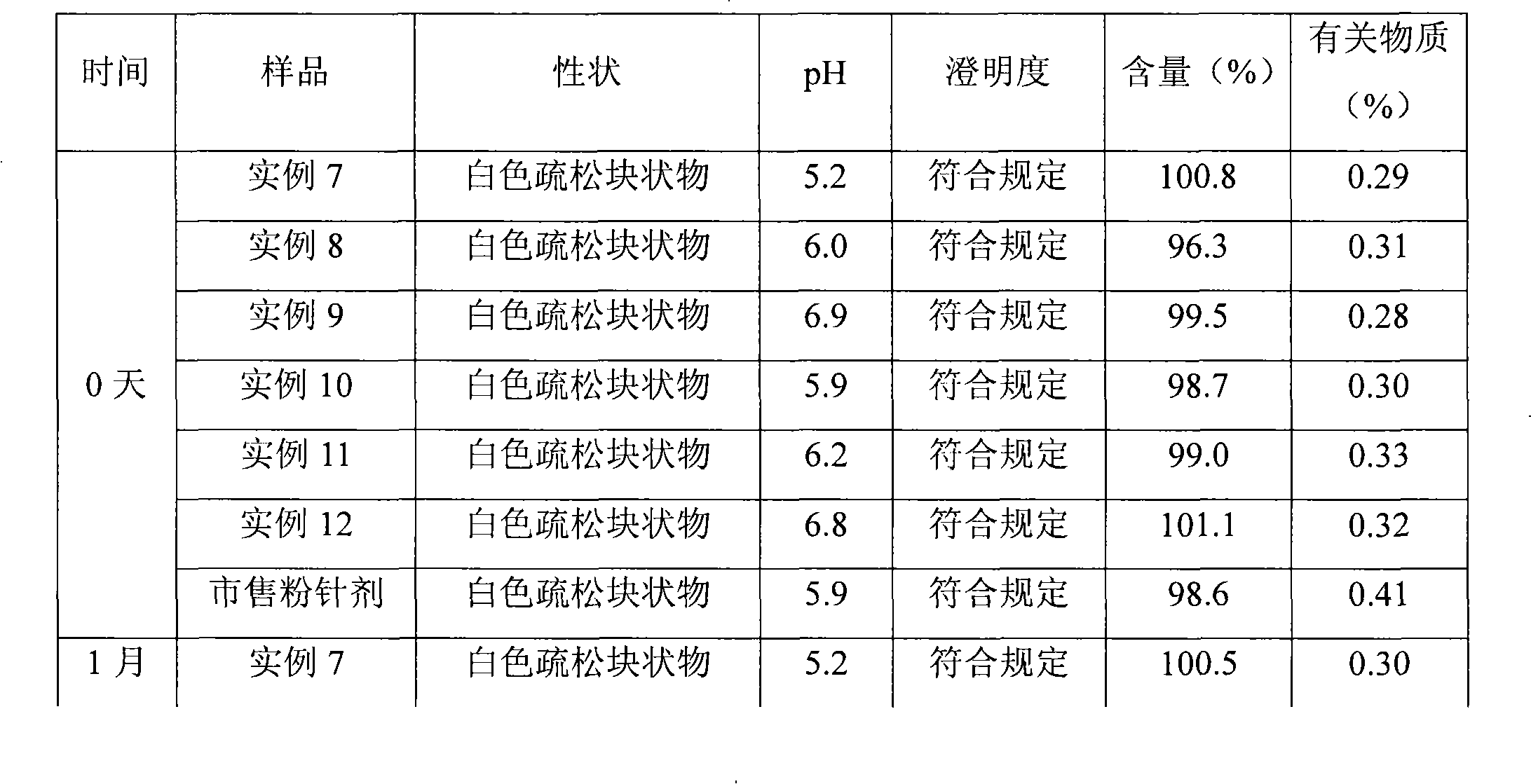
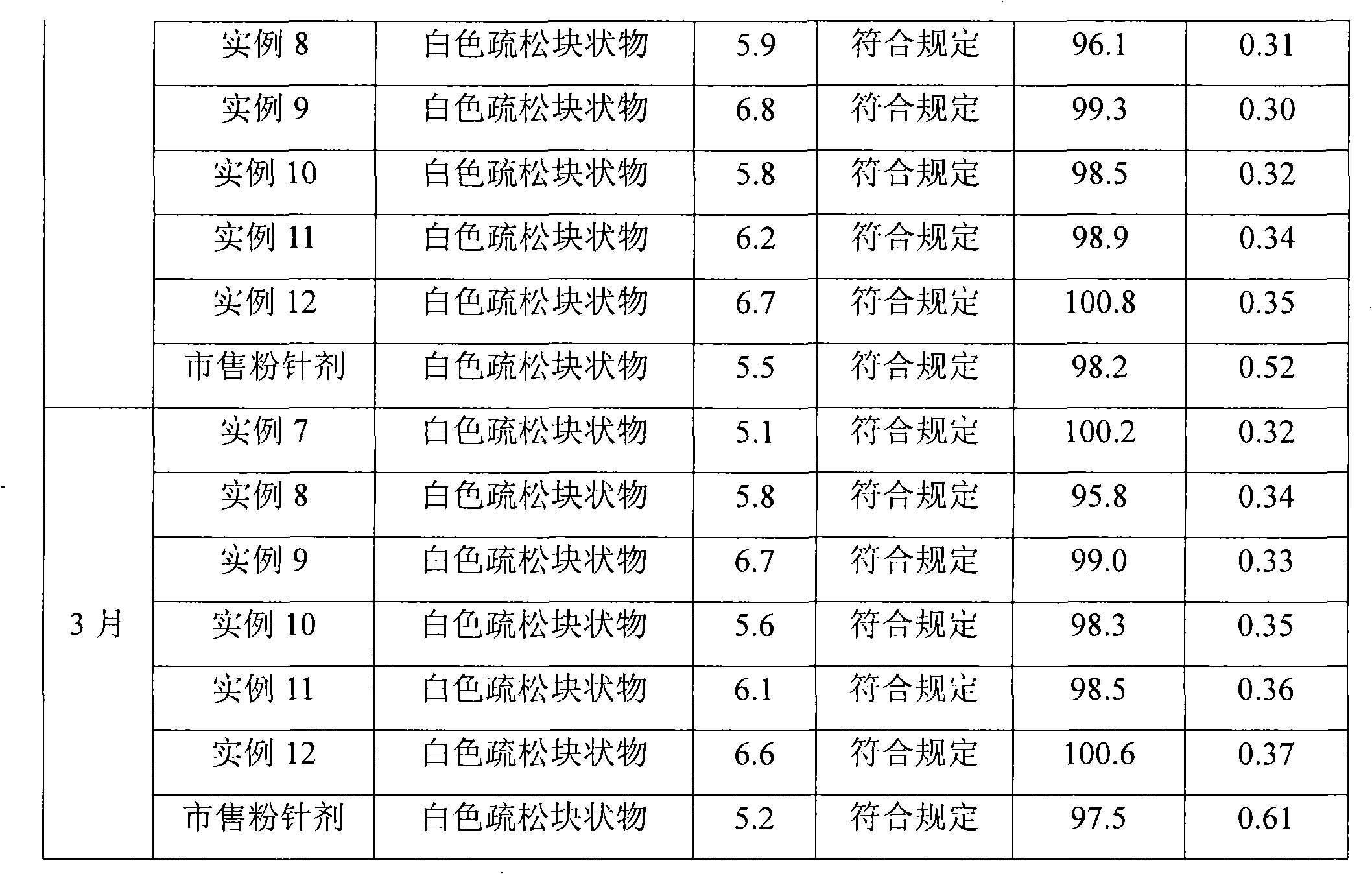
Embodiment 1
[0060] Meclofenoxate Hydrochloride 60g
[0061] Gelatin 180g
[0062] Dextran 150g
[0063] Span 20g
[0064] Poloxamer 20g
[0065] Preparation Process
[0066] (1) 60g meclofenoxate hydrochloride is added in the water for injection, dissolves completely;
[0067] (2) add 180g gelatin and 20g span to step (1) gained solution, prepare water-in-oil emulsion with mechanical stirring method;
[0068] (3) Add 500ml of 30% dextran solution and 20g poloxamer to the solution obtained in step (2), and prepare water-in-oil-in-water multiple emulsion by mechanical stirring;
[0069] (4) the potassium hydroxide solidified liquid that is 3mol / L is dripped by the solubility of step (3) gained multiple emulsion in the high-voltage electrostatic field by the orifice injection, and the electric field voltage is 2.2kv, and the injection speed is 5ml / min After solidification for 10 minutes, the microcapsules were filtered out, and the microcapsules were washed with water until the pH was ...
Embodiment 2
[0071] Meclofenoxate Hydrochloride 250g
[0072] Gelatin 800g
[0073] Dextran 300g
[0075] Polyoxyethylene Hydrogenated Castor Oil 100g
[0076] Preparation Process
[0077] (1) 250g meclofenoxate hydrochloride is added in the water for injection, dissolves completely;
[0078] (2) Add 800g gelatin and 100g egg yolk lecithin to the solution obtained in step (1), prepare water-in-oil emulsion with high-pressure homogenization;
[0079] (3) Add 600ml of 50% dextran solution and 100g polyoxyethylene hydrogenated castor oil to the solution obtained in step (2), prepare water-in-oil-in-water multiple emulsion with high-pressure homogenization;
[0080] (4) the sodium ethylate solidified liquid that is 6mol / L is dripped by the solubility of step (3) gained multiple emulsion in the high-voltage electrostatic field by the orifice injection, and the electric field voltage is 4.4kv, and the injection speed is 20ml / min, After solidification for 60 ...
Embodiment 3
[0082] Meclofenoxate Hydrochloride 100g
[0083] Gelatin 250g
[0084] Dextran 180g
[0085] Soy Lecithin 50g
[0086] Hydrogenated Soy Lecithin 50g
[0087] Tween 80 50g
[0088] Polyoxyethylene Stearate 50g
[0089] Preparation Process
[0090] (1) 100g meclofenoxate hydrochloride is added in the water for injection, dissolves completely;
[0091] (2) Add 250g gelatin, 50g soybean lecithin and 50g hydrogenated soybean lecithin to the solution obtained in step (1), prepare water-in-oil emulsion with mechanical stirring;
[0092] (3) Add 30% dextran solution 600ml, 50g Tween 80 and 50g polyoxyethylene stearate to step (2) gained solution, prepare water-in-oil-in-water multiple emulsion with mechanical stirring;
[0093] (4) the sodium hydroxide solidified liquid that is 5mol / L is the sodium hydroxide solidified liquid that the solubility of step (3) gained compound emulsion is dripped into the high-voltage electrostatic field by the orifice injection, and the electric f...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More