New method for preparation of netupitant key intermediate N-methyl-4-(2-methyl phenyl)-6-(4-methyl-1-piperazinyl)-3-pyridylamine
A key technology of methyl phenyl and tannamide, which is applied in the field of pharmaceutical synthesis and achieves the effects of easy availability of raw materials, mild reaction conditions and simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0039] Preparation of N-methoxyformyl-6-(4-methyl-1-piperazinyl)-4-(2-methylphenyl)-3-pyridinamine (compound of formula II):
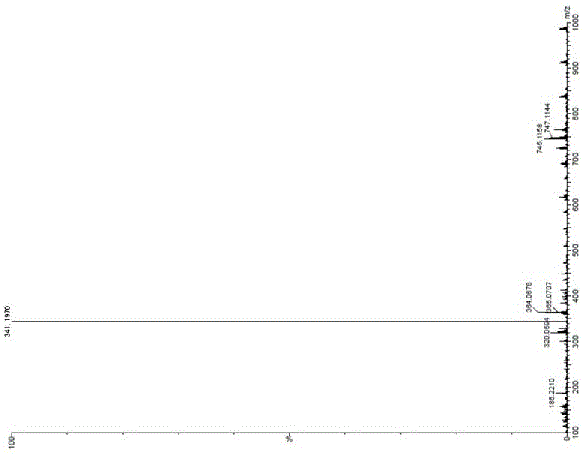
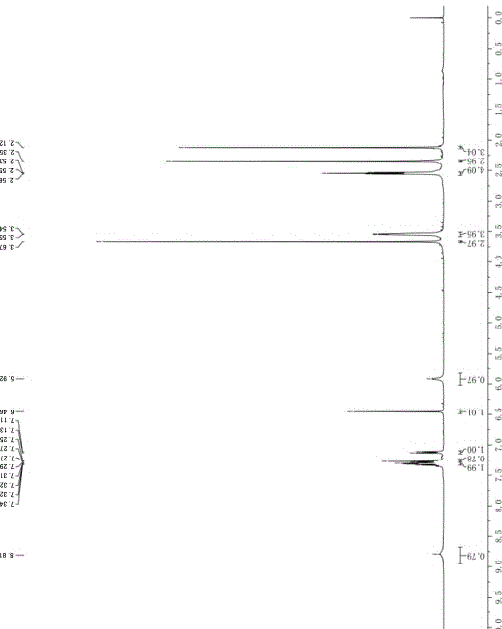
[0040] Add 70 g of 6-(4-methyl-1-piperazinyl)-4-(2-methylphenyl) nicotinamide into a one-necked flask, dissolve it with 1 L of anhydrous methanol, and then stir and cool down in an ice bath Finally, 80 g of iodobenzene diacetate and 26 g of potassium hydroxide were added, and the mixture was stirred and reacted in an ice bath for 1 hour. TLC monitored the completion of the reaction. After concentration, add 1L of water to the reaction liquid successively, extract 3 times with ethyl acetate, combine the organic layers, then wash twice with saturated sodium chloride solution, dry the organic layer with anhydrous sodium sulfate, filter, and concentrate to obtain yellow Oil 65g. The yield is 85%. 1 H-NMR (CDCl 3 ,400MHz): δ =8.81(brs,1H),7.34~7.25(m,3H),7.12(d, J =8Hz),6.46(s,1H),5.92(s,1H),3.67(s,3H),3.55~3.54(m,4H),2.55(t, J =4Hz,4H),2.35(s,3H),2...
Embodiment 2
[0047] Preparation of N-methoxyformyl-6-(4-methyl-1-piperazinyl)-4-(2-methylphenyl)-3-pyridinamine (compound of formula II):
[0048] Under ice-bath conditions, 4g of iodobenzene diacetate was added in batches to 3g of 6-(4-methyl-1-piperazinyl)-4-(2-methylphenyl)nicotinamide and 4gCH 3 In the chloroform solution of ONa, after the addition was completed, the reaction was continued to be stirred and reacted for 3 h under an ice bath; the reaction was complete as monitored by TLC. Add 10 mL of water to the reaction solution to quench the reaction, extract 3 times with chloroform, combine the organic layers, then wash twice with saturated sodium chloride solution, dry the organic layer with anhydrous sodium sulfate, filter, and concentrate to give yellow oil 2.4 g, yield 62%.
[0049]
[0050] The solvent in this embodiment can also be replaced by other types of solvents, such as: acetone, N,N-dimethylformamide, dimethyl sulfoxide, methanol, and the like.
Embodiment 3
[0052] Preparation of N-methyl-6-(4-methyl-1-piperazinyl)-4-(2-methylphenyl)-3-pyridinamine (compound of formula III):
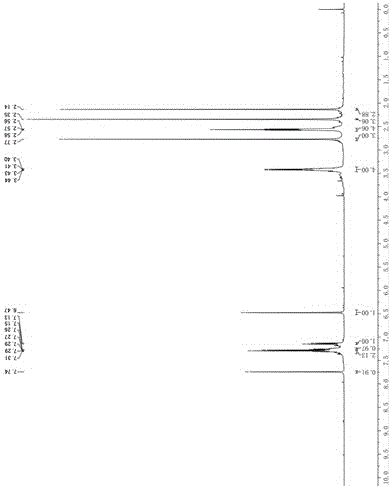
[0053] Dissolve 40 g of the compound of formula II in 1.5 L of toluene, place in an ice-water bath, and add dropwise 200 ml of red aluminum solution dissolved in 1 L of toluene. During the dropping process, the reaction is violent, and the temperature is kept below 50°C. After the dropwise addition was completed, the reaction was heated in an oil bath at 50° C., and the reaction was complete as monitored by TLC. The reaction solution was slowly added dropwise to 1N NaOH solution under ice bath, then ethyl acetate was added to extract the aqueous phase, and the organic layer was washed with saturated sodium chloride solution until neutral, dried with anhydrous sodium sulfate, filtered and concentrated to obtain brown yellow 30.5 g of oil, yield: 62%. 1 H-NMR (CDCl 3 ,400MHz): δ =7.74(s,1H),7.31~7.13(m,4H),6.47(s,1H),3.44~3.40(m,4H),2.77(s,3H),2.57(t, J =...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com