Compound feed for pregnant sows, preparation method of compound feed and use method of compound feed
A technology for compound feed and pregnant sows, applied in the field of pig feed, can solve the problems of affecting the economic benefits of breeding, lack of exercise of sows, physical damage of sows, etc., to improve estrus rate, accelerate absorption and utilization, and excretion of harmful substances , the effect of extending the service life
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
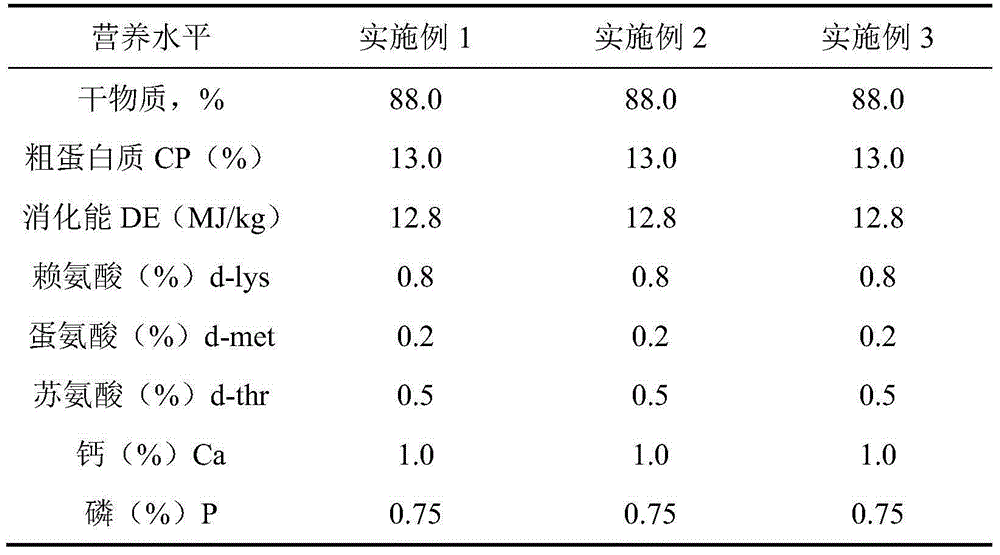
[0024] The compound feed for pregnant sows in this embodiment mainly consists of the following components in parts by weight: 52 parts of corn, 8.3 parts of soybean meal, 10 parts of wheat, 25 parts of bran, 1.36 parts of stone powder, 1.8 parts of calcium hydrogen phosphate, lysine 0.28 part of acid sulfate, 0.01 part of threonine, 0.2 part of baking soda, 0.45 part of table salt, 0.1 part of choline chloride, 0.5 part of premix;
[0025] Among them, every 1000g premix consists of the following components: 35% ascorbyl phosphate 18.5g, 500,000 U / g vitamin A acetate 2.5g, vitamin D 3 0.73g, 50% vitamin E acetate 33.0g, sodium bisulfite menadione 1.97g, thiamine nitrate 0.31g, 80% spray-dried riboflavin 1.25g, pyridoxine hydrochloride 0.62g, 1% vitamin B 12 0.52g, 80% folic acid 0.62g, niacin 5.19g, D-calcium pantothenate 3.53g, 2% D-biotin 9.03g, copper sulfate pentahydrate 6.72g, ferrous sulfate monohydrate 37.4g, zinc sulfate monohydrate 32.3 g, 25.0 g of manganese sulfate ...
Embodiment 2
[0031] The compound feed for pregnant sows of this embodiment mainly consists of the following components in parts by weight: 44 parts of corn, 6.3 parts of soybean meal, 20 parts of wheat, 25 parts of bran, 1.35 parts of stone powder, 1.75 parts of calcium hydrogen phosphate, lysine 0.33 parts of acid sulfate, 0.02 parts of threonine, 0.2 parts of baking soda, 0.45 parts of table salt, 0.1 parts of choline chloride, 0.5 parts of premix;
[0032] Among them, every 1000g premix consists of the following components: 35% ascorbyl phosphate 18.5g, 500,000 U / g vitamin A acetate 2.5g, vitamin D 3 0.73g, 50% vitamin E acetate 33.0g, sodium bisulfite menadione 1.97g, thiamine nitrate 0.31g, 80% spray-dried riboflavin 1.25g, pyridoxine hydrochloride 0.62g, 1% vitamin B 120.52g, 80% folic acid 0.62g, niacin 5.19g, D-calcium pantothenate 3.53g, 2% D-biotin 9.03g, copper sulfate pentahydrate 6.72g, ferrous sulfate monohydrate 37.4g, zinc sulfate monohydrate 32.3 g, 25.0 g of manganese su...
Embodiment 3
[0038] The compound feed for pregnant sows of this embodiment mainly consists of the following components in parts by weight: 35 parts of corn, 5.3 parts of soybean meal, 30 parts of wheat, 25 parts of bran, 1.33 parts of stone powder, 1.70 parts of calcium hydrogen phosphate, lysine 0.38 part of acid sulfate, 0.04 part of threonine, 0.2 part of baking soda, 0.45 part of table salt, 0.1 part of choline chloride, 0.5 part of premix;
[0039] Among them, every 1000g premix consists of the following components: 35% ascorbyl phosphate 18.5g, 500,000 U / g vitamin A acetate 2.5g, vitamin D 3 0.73g, 50% vitamin E acetate 33.0g, sodium bisulfite menadione 1.97g, thiamine nitrate 0.31g, 80% spray-dried riboflavin 1.25g, pyridoxine hydrochloride 0.62g, 1% vitamin B 12 0.52g, 80% folic acid 0.62g, niacin 5.19g, D-calcium pantothenate 3.53g, 2% D-biotin 9.03g, copper sulfate pentahydrate 6.72g, ferrous sulfate monohydrate 37.4g, zinc sulfate monohydrate 32.3 g, 25.0 g of manganese sulfate...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Granularity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More