A kind of synthetic method of 1,2,3-triazole compound
A synthesis method and compound technology, applied in organic chemistry and other directions, can solve problems such as product structure limitations, achieve high reaction specificity, improve reaction yield and product quality, and have no harsh reaction conditions.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
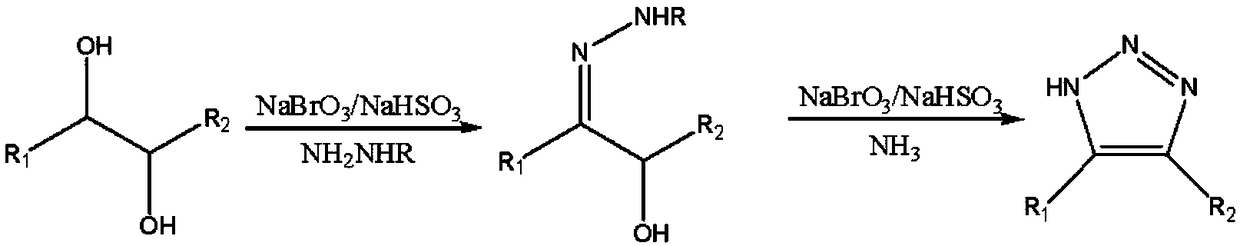
[0034] A synthesis method of a 1,2,3-triazole compound proposed by the present invention, the 1,2,3-triazole compound is 4,5-dimethyl-1H-1,2,3-triazole Azole, the synthetic method of described 4,5-dimethyl-1H-1,2,3-triazole comprises the following steps:
[0035] Synthesis of S1, 3-p-toluenesulfonylhydrazono-2-butanol
[0036] Dissolve 9.03g of 2,3-butanediol in 40ml of dichloroethane, slowly add 11.5g of sodium bromate and 7.81g of sodium bisulfite to the solution, raise the temperature to 40°C, and stir within 1 hour Add 19.57g p-toluenesulfonyl hydrazide in batches, and HPLC tracks and detects the reaction process until the peak area of 2,3-butanediol in HPLC is less than 0.2%, and adding a mass fraction of 10% sodium hydroxide aqueous solution to the reaction solution The pH value is 7, the reaction solution is concentrated to half volume and then cooled to room temperature, the precipitated inorganic salt is filtered, and the filtrate is distilled under reduced pressur...
Embodiment 2
[0042] A synthesis method of a 1,2,3-triazole compound proposed by the present invention, the 1,2,3-triazole compound is 4,5-dimethyl-1H-1,2,3-triazole Azole, the synthetic method of described 4,5-dimethyl-1H-1,2,3-triazole comprises the following steps:
[0043] Synthesis of S1, 3-benzenesulfonylhydrazono-2-butanol
[0044] Dissolve 9.02g of 2,3-butanediol in 40ml of acetonitrile, slowly add 12.6g of sodium bromate and 8.24g of sodium bisulfite to the solution, raise the temperature to 50°C, and add in batches within 1 hour under stirring 18.62g of benzenesulfonyl hydrazide, HPLC tracking and detection of the reaction process until the peak area of 2,3-butanediol in the HPLC is less than 0.2%, adding a mass fraction of 10% sodium hydroxide aqueous solution to the reaction solution until the pH value of the solution is 7 , the reaction solution was concentrated to half the volume and then cooled to room temperature, the precipitated inorganic salt was filtered, and the filt...
Embodiment 3
[0050] A synthesis method of a 1,2,3-triazole compound proposed by the present invention, the 1,2,3-triazole compound is 4,5-diphenyl-1H-1,2,3-triazole Azole, the synthetic method of described 4,5-diphenyl-1H-1,2,3-triazole comprises the following steps:
[0051] S1, Synthesis of 1,2-diphenyl-1-methanesulfonylhydrazono-2-ethanol
[0052] Dissolve 21.26g of 1,2-diphenyl-1,2-ethanediol in 40ml of carbon tetrachloride, slowly add 14.8g of sodium bromate and 9.62g of sodium bisulfite into the solution, and raise the temperature to 60°C. Under stirring condition, in 1 hour, add 12.5g methanesulfonyl hydrazide in batches, simultaneously HPLC tracks and detects reaction progress to the peak area of 1,2-diphenyl-1,2-ethanediol in HPLC is less than 0.2%, to The reaction solution is added with a mass fraction of 10% sodium hydroxide aqueous solution until the pH value of the solution is 7, the reaction solution is concentrated to half the volume and then cooled to room temperature, t...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com