Kit for anxiolytic and hypnotic drug in serum by ultra-high performance liquid chromatography-tandem mass spectrometry technology
An ultra-high performance liquid chromatography and tandem mass spectrometry technology, applied in the field of blood testing, can solve the problems of tediousness, low sensitivity of diazepam, and long analysis time of a single sample
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0144] 1. Experimental materials and instruments
[0145] 1. Materials
[0146] The samples came from serum samples collected from the outpatient clinic of Wuhan Asia Heart Hospital in November 2019.
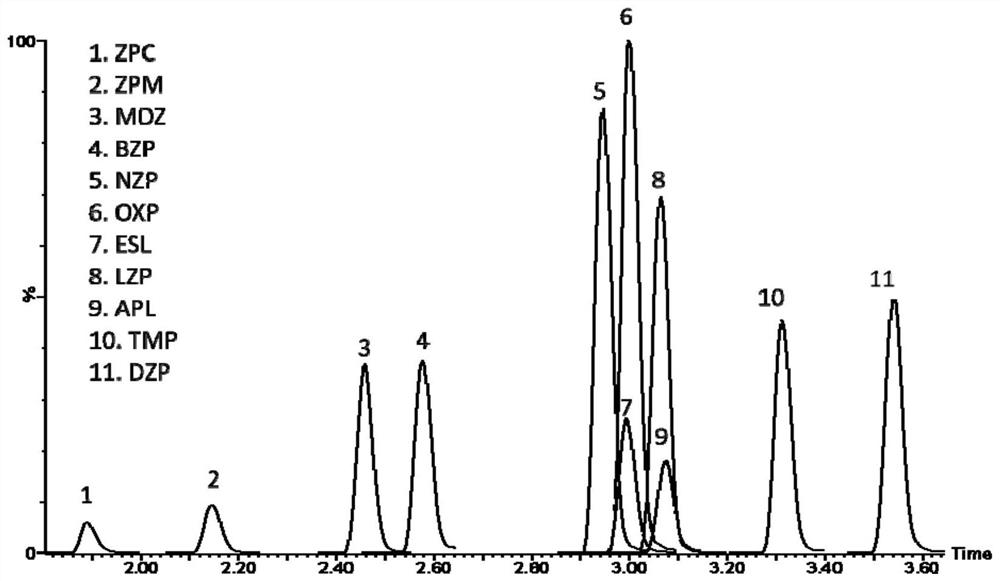
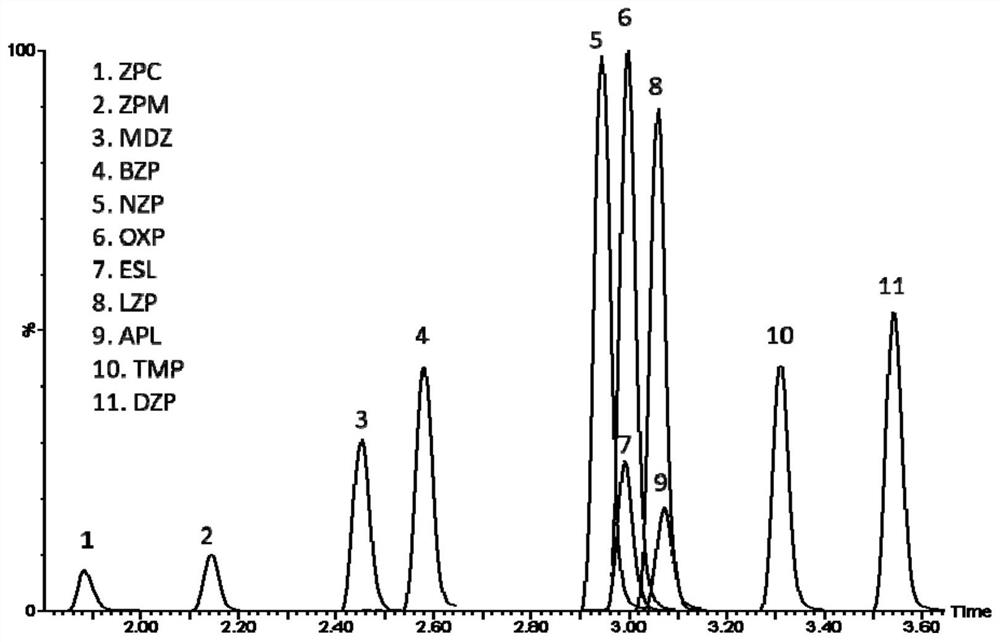
[0147] (1) Instruments: Xevo TQ-S triple quadrupole mass spectrometer (Waters Corporation); UPLC I-Class ultra-high performance liquid chromatography system (with automatic sampler, Waters Corporation); SCILOGEX D2012 high-speed desktop centrifuge (USA) ; ultrapure water instrument (ELGA LabWater, UK); multi-tube vortex mixer (Vortex genie2, US); adjustable pipette (Eppendorf 0.5-10 μL, 10-100 μL, 100-1000 μL); glassware, graduated cylinder, etc. . .
[0148] (2) Reagent consumables: MS grade methanol (Fisher, USA); MS grade acetonitrile (Fisher, USA); HPLC grade acetonitrile (Honeywell, USA); MS grade acetic acid (Fisher, USA); MS grade ammonium acetate (Fisher, USA) ); HPLC grade methanol (Honeywell, USA); column ACQUITY UPLC CSH C18 (2.1×50mm, 1.7 μm).
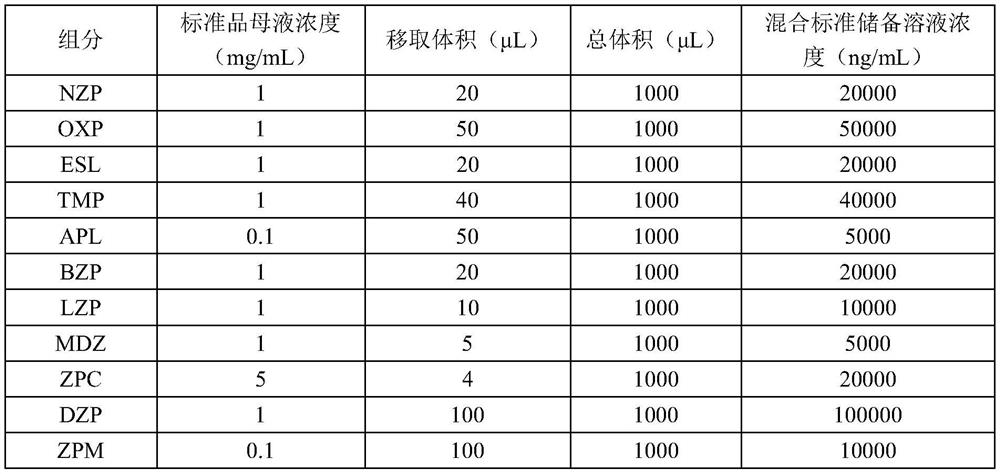
[0149] (3) Stand...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More