Method for determining content of bromoethane in Etazocine hydrobromide intermediate
A technology of etazocine hydrobromide and intermediates, which is applied in the field of drug analysis, can solve the problems of low recovery rate of bromoethane, does not meet detection requirements, etc., and achieves simple and convenient determination process, high accuracy, and precise sample introduction. good effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
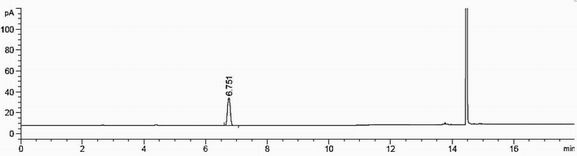
[0024] The mensuration of bromoethane content in the compound II of embodiment 1
[0025] 1) Chromatographic conditions:
[0026] Gas chromatograph: Agilent 7890A gas chromatograph (with headspace autosampler);
[0027] Detector: FID;
[0028] Gas chromatography column: DB-624 capillary column (30m×320μm, 1.8μm);
[0029] Carrier gas: nitrogen;
[0030] Carrier gas flow rate: 1.0ml / min;
[0031] Hydrogen flow rate: 30ml / min;
[0032] Air flow rate: 300ml / min;
[0033] Column oven temperature rise program: initial 40°C, keep for 10min, then raise the temperature to 200°C at a rate of 40°C per minute, keep for 4min;
[0034] Injection port temperature: 220°C;
[0035] Detector temperature: 260°C;
[0036] Split ratio: 10:1;
[0037] Headspace equilibrium temperature: 80°C.
[0038] Headspace equilibration time: 25min.
[0039] 2) Sample solution preparation:
[0040] Preparation of the reference substance solution: Accurately weigh about 10 mg of ethyl bromide, put i...
Embodiment 2
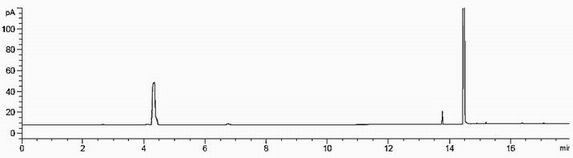
[0043] Embodiment 2 method verification experiment
[0044] 1) Chromatographic conditions
[0045] Instrument: Agilent 7890A gas chromatograph (additional Agilent 7891A headspace autosampler)
[0047] Chromatographic column: DB-624 capillary column (30m×320μm, 1.8μm)
[0048] Flow rate: 1.0ml / min
[0049] Hydrogen: 30ml / min
[0050] Air: 300ml / min
[0051] Carrier gas: nitrogen
[0052] Heating program: initially at 40°C, keep for 10 minutes, then raise the temperature to 200°C at a rate of 40°C per minute, and keep for 4 minutes.
[0053] Injector temperature: 220°C
[0054] Detector temperature: 260°C
[0055] Split ratio: 10:1
[0056] The headspace equilibration temperature was 80°C, and the equilibration time was 25 minutes.
[0057] 2) Preparation of solution
[0058] Blank solution: Accurately measure 1ml of N,N-dimethylformamide and 1ml of 48% hydrobromic acid, put it in a 20ml headspace bottle, seal it, shake well, and use it...
Embodiment 3
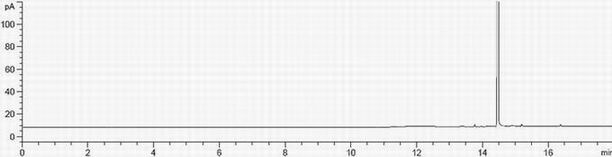
[0091] Embodiment 3 Different sample processing method recoveries difference
[0092] Investigate the recoveries of three kinds of samples prepared by untreated, treated with acetic acid and treated with hydrobromic acid to test the content of ethyl bromide.
[0093] 1) Chromatographic conditions:
[0094] Gas chromatograph: headspace autosampler;
[0095] Detector: FID;
[0096] Gas chromatography column: DB-624 capillary column (30m×320μm, 1.8μm);
[0097] Carrier gas: nitrogen;
[0098] Carrier gas flow rate: 1.0ml / min;
[0099] Hydrogen flow rate: 30ml / min;
[0100] Air flow rate: 300ml / min;
[0101] The temperature rise program of the column oven: the initial column temperature is 40°C and kept for 10 minutes, then the temperature is raised to 200°C at a rate of 40°C per minute, and kept for 4 minutes;
[0102] Injection port temperature: 220°C;
[0103] Detector temperature: 260°C;
[0104] Split ratio: 10:1;
[0105] Headspace equilibrium temperature: 80°C;
...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com