Electrolyte for efficiently regulating and controlling dissolution behavior of magnesium anode and preparation method of electrolyte
A technology of magnesium anode and electrolyte, which is applied in the field of electrolyte and its preparation for efficiently regulating the dissolution behavior of magnesium anode, can solve the problems of voltage hysteresis, poor corrosion resistance of anode, etc., and achieve discharge specific capacity improvement, performance improvement, and discharge performance improvement. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0038] In the electrolyte for efficiently regulating the dissolution behavior of magnesium anode, the concentration of sodium chloride is 0.62M, and the concentration of 5-sulfosalicylic acid is 0.01M;
[0039] Mix sodium chloride and 5-sulfosalicylic acid to prepare a mixture;
[0040] Add deionized water to the mixture, and then stir all the materials until a transparent solution is formed; when adding deionized water, place the mixture in a container, and pour the deionized water along the inner wall of the container; the stirring speed is 100~150rpm;
[0041] The pH value of the transparent solution is adjusted with sodium hydroxide or hydrochloric acid, and the pH value is monitored by a pH-conductivity meter until the pH value is 7.0±0.2, and an electrolyte solution for efficiently regulating the dissolution behavior of magnesium anode is prepared.
[0042] The method of use is:
[0043] The magnesium anode is placed in an electrolyte that efficiently regulates the disso...
Embodiment 2
[0047] In the electrolyte for efficiently regulating the dissolution behavior of magnesium anode, the concentration of sodium chloride is 0.62M, and the concentration of 5-sulfosalicylic acid is 0.05M;
[0048] The preparation method is the same as in Example 1;
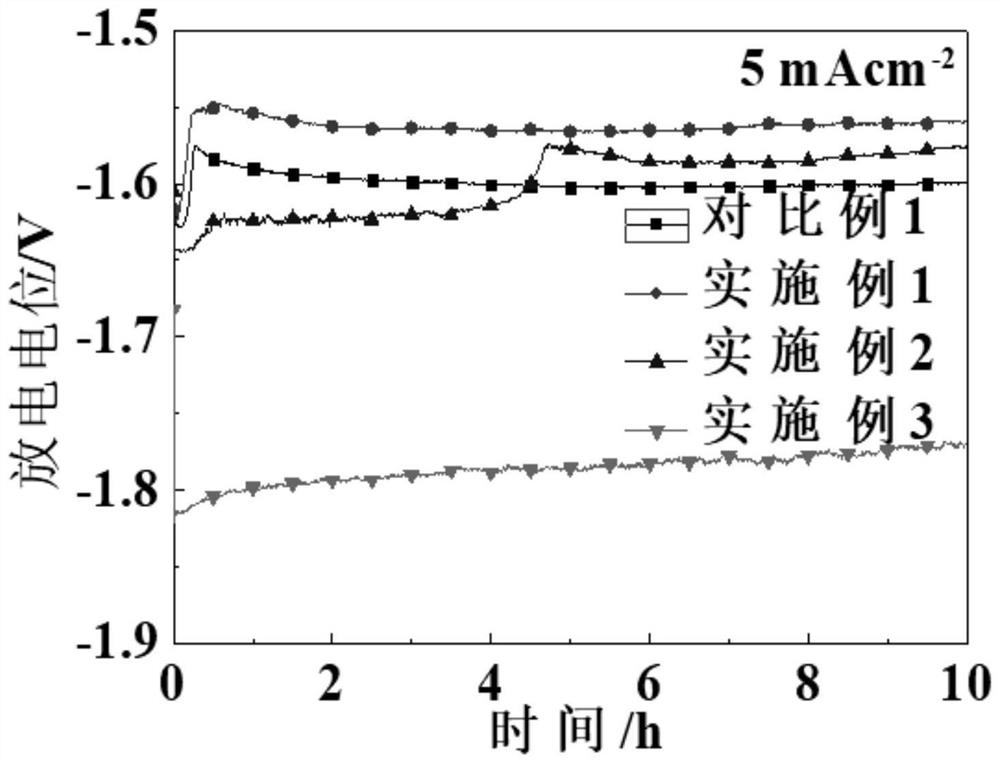
[0049] Using method and testing method are the same as Example 1, and the hydrogen evolution amount after soaking for two days is as follows: figure 1 The magnesium anode is chemically active, and the total amount of hydrogen evolution is higher than that of Comparative Example 1, which indicates that it is helpful to increase the electrochemical activity of the magnesium anode, increase the contact between the anode and the electrolyte, and promote the dissolution of the anode, so as to provide lower the discharge potential; by Figures 2 to 4 It can be seen that when the current density is 2.5, 5 mA cm -2 , the average discharge potential of the anode was significantly lower than that of Comparative Example 1; th...
Embodiment 3
[0051] In the electrolyte for efficiently regulating the dissolution behavior of magnesium anode, the concentration of sodium chloride is 0.62M, and the concentration of 5-sulfosalicylic acid is 0.1M;
[0052] The preparation method is the same as in Example 1;
[0053] The method of use and the test method are the same as in Example 1, and the hydrogen evolution amount after soaking for two days reaches the highest, which is 27mL / cm 2 ; This shows that the magnesium anode has the fastest dissolution rate in the electrolyte, the strongest electrochemical activity, and the greatest discharge potential; the rapid anode dissolution ability determines that the deposition of discharge products on the anode surface is extremely difficult; the anode during the discharge process Mg(OH) 2 The probability of occurrence is the smallest, which is convenient for the contact between the anode and the electrolyte, and provides a low and stable discharge potential; the discharge potential of ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More