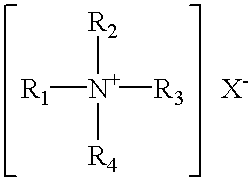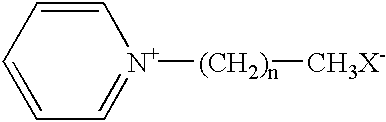Patents
Literature
347 results about "Sulfosalicylic acid" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
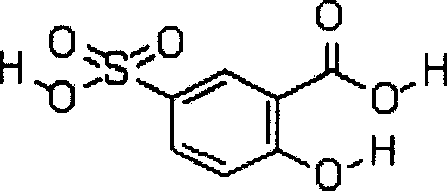
Sulfosalicylic acid is used in urine tests to determine urine protein content. The chemical causes the precipitation of dissolved proteins, which is measured from the degree of turbidity. It is also used for integral colour anodizing.
Reagent for measurement of leukocytes and hemoglobin concentration in blood
InactiveUSRE38131E1Measure the leukocyte countMeasurement stabilitySamplingBiological testingMethemoglobinWhite blood cell
A reagent for measurement of leukocytes and hemoglobin concentration in the blood includes a cationic surfactant in an amount sufficient to lyse erythrocytes and denature hemoglobin, at least one of the following hemoglobin stabilizers:(a) sulfosalicylic acid, or its salt, in an amount effective for promoting the conversion of hemoglobin into methemoglobin,(b) 0.2 to 10.0 g / L of a water-soluble chelating agent having a nitrogen atom and a carboxyl group, and(c) piperazine, or its salt, in an amount effective for promoting the conversion of hemoglobin into methemoglobin, anda buffer for maintaining pH at 4 to 6.
Owner:SYSMEX CORP
Rare earth modified surface treating agent for copper and copper alloys and preparation method thereof
ActiveCN102071415AImprove corrosion resistanceShort passivation timeMetallic material coating processesRare earthSulfosalicylic acid
The invention discloses a rare earth modified surface treating agent for copper and copper alloys and a preparation method thereof. The surface treating agent per liter comprises the following raw materials by weight: 1-3g of benzotriazole, 1-5g of sodium molybdate, 0.1-0.3g of sodium silicate, 0.3-0.6g of rare earth salt, 10-20g of complexing agent, 6-10g of sulfosalicylic acid, 0.2-0.6g of surfactant, a small amount of pH regulator and the balance water. The surface treating agent of the invention can be used for passivation at the room temperature, have short passivation time and be green and environmentally friendly; the passivated copper or copper alloy surface has good corrosion resistance; and the treating agent has simple treatment technology and is convenient for actual production.
Owner:斯特凯新材料(上海)有限公司
Environment-friendly cleaning agent
InactiveCN103525591AImprove residence reaction timeLess materialOrganic non-surface-active detergent compositionsPhosphoric acidSulfosalicylic acid
Owner:LIUZHOU XINLEI TECH
Preparation process of doxycycline hydrochloride
ActiveCN101786971AHigh yieldMaterial excellenceOrganic compound preparationSulfonic acids salts preparationHydrogenAdjuvant
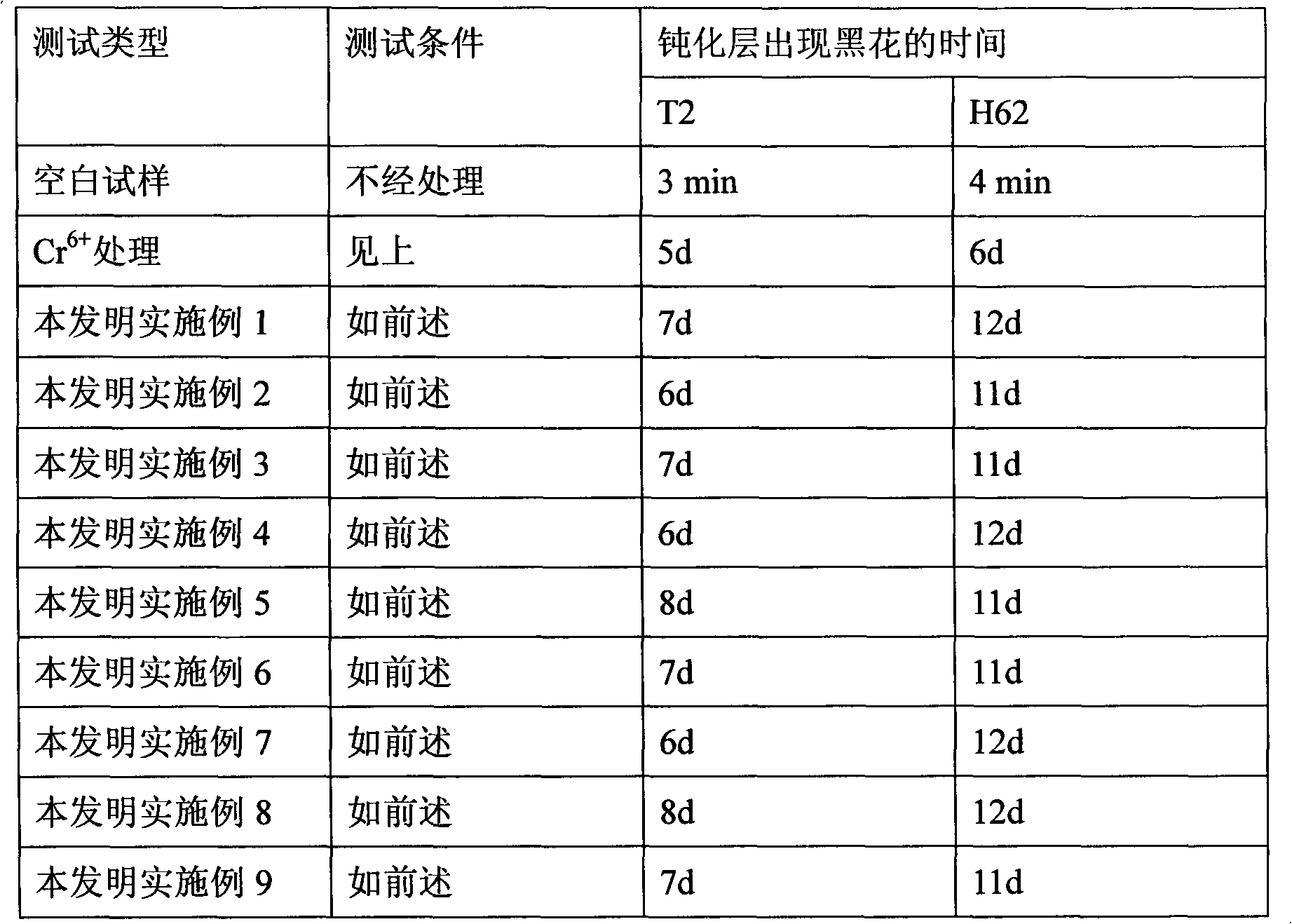
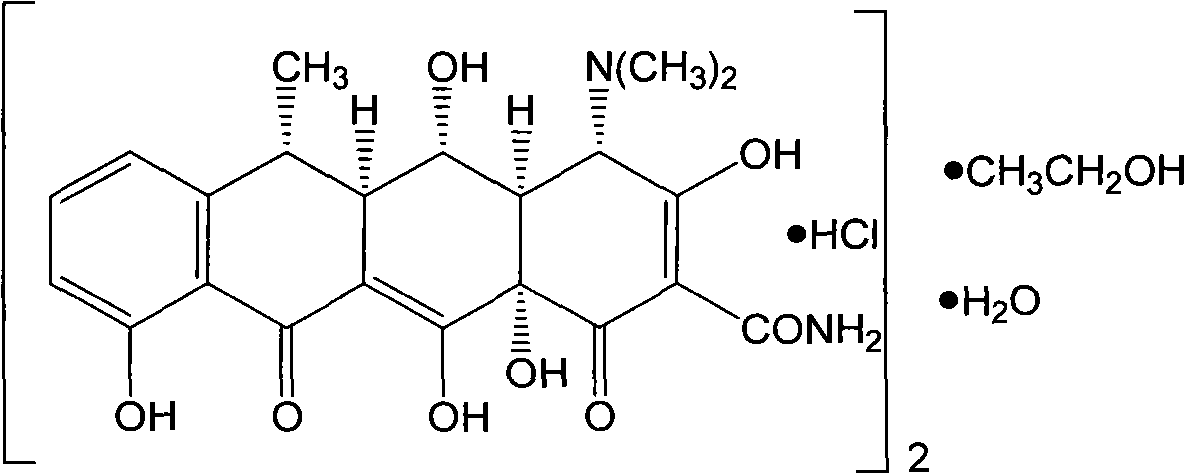
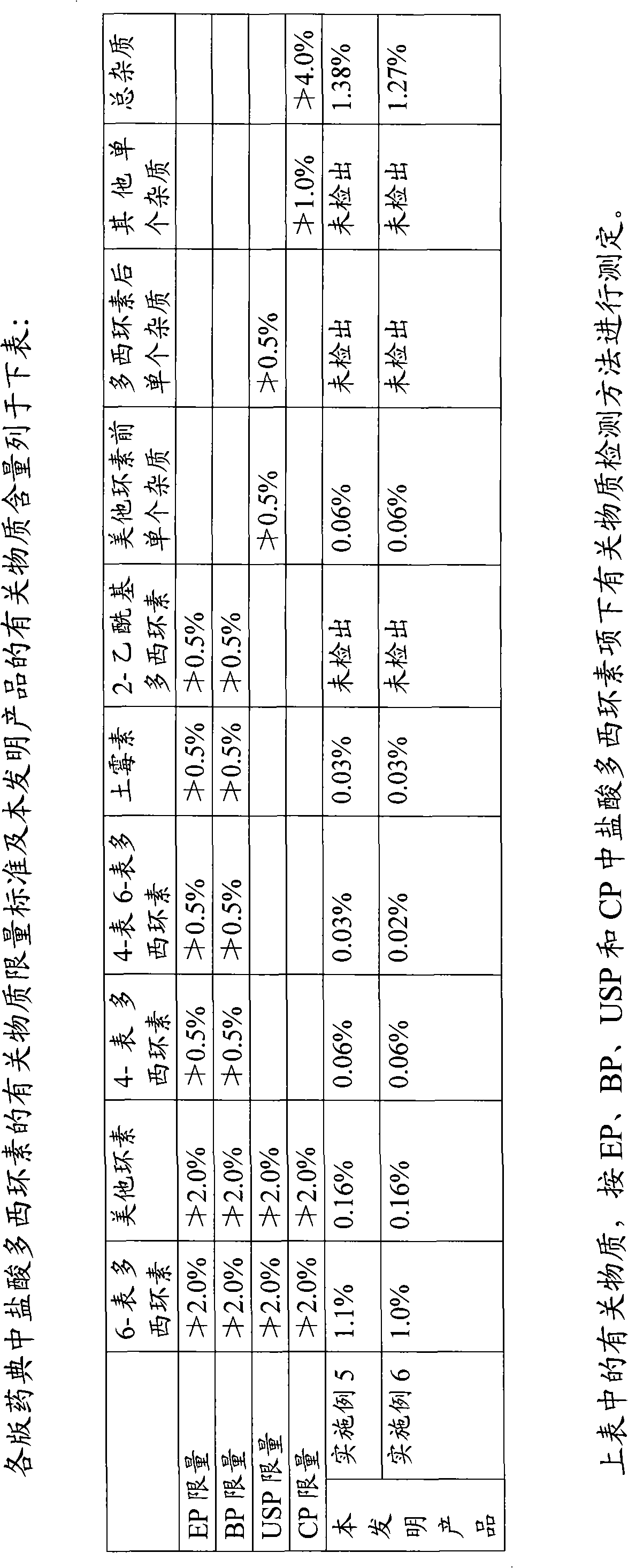
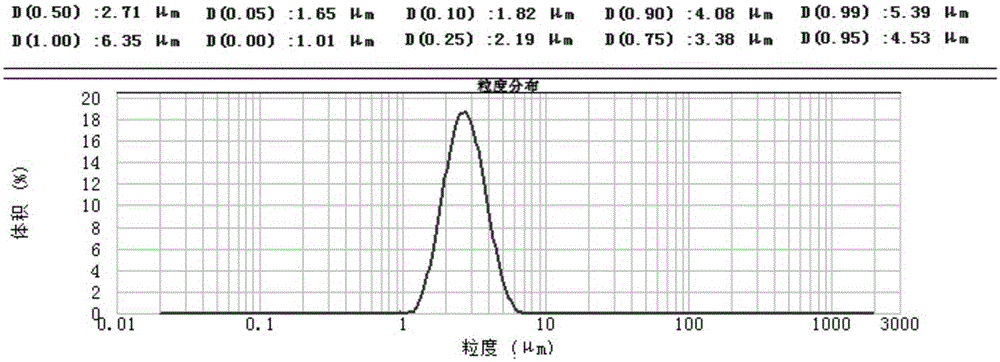
The invention provides a preparation process of doxycycline hydrochloride. In the preparation process, 5-hydroxy-6-demethyl-6-desoxy-6-methylene-11-chlorotetracycline is pressurized and hydrogenated with hydrogen in the presence of catalyst-palladium / carbon (Pd / C) and adjuvant and then reacts with 5-sulfosalicylic acid to obtain alpha-6-doxycycline 5-sulfosalicylate; then alpha-6-doxycycline base is generated after basification; and finally the doxycycline hydrochloride is generated through salt transformation. The preparation process of the doxycycline hydrochloride has the advantages that the productivity and stereoselectivity are improved, the content of related substances in the finished product is reduced, and the high-quality finished product is obtained.
Owner:扬州联博药业有限公司
Nickel, cobalt and aluminum hydroxide and preparation method thereof and cathode material for lithium ion cell
InactiveCN105206827AGood spherical particle sizeHigh tap densityCell electrodesSecondary cellsEthylene diamineNickel salt
The invention provides a preparation method for nickel, cobalt and aluminum hydroxide. The preparation method comprises the steps that a nickel salt solution and a cobalt salt solution are mixed to obtain a first mixed solution; an aluminum salt solution and a complexing agent are mixed to obtain a second mixed solution; under the action of a precipitant, a coprecipitation reaction is performed on the first mixed solution and the second mixed solution to obtain the nickel, cobalt and aluminum hydroxide, the complexing agent is prepared from aqueous ammonia, ethylene diamine tetraacetic acid and sulfosalicylic acid or glycine, the temperature of the coprecipitation reaction is set at 40 DEG C-65 DEG C, and the pH value of the coprecipitation reaction is set at 10.5-12. By controlling the material adding order, selection of the complexing agent and the temperature and the pH value of the coprecipitation reaction, the prepared nickel, cobalt and aluminum hydroxide has the better spherical appearance and is concentrated in particle size and high in tap density. The invention further provides the nickel, cobalt and aluminum hydroxide prepared through the method and a cathode material for a lithium ion cell.
Owner:NINGBO RONBAY LITHIUM BATTERY MATERIAL CO LTD
Method for measuring content of ferrous iron and ferric iron in ferrous phosphate lithium anode material
InactiveCN102323228AQuick responseResponse speed is efficientColor/spectral properties measurementsPhosphatePhenanthroline
The invention discloses a method for measuring the content of ferrous iron and ferric iron in a ferrous phosphate lithium anode material, which is used for carrying out sample treatment and sample measurement under a protective atmosphere and comprises the following steps: measuring the content of the ferrous iron: dissolving the ferrous phosphate lithium anode material by using acid; then, when the pH of a solution is regulated to 2-9, adding phenanthroline to produce an orange complex with the ferrous iron; and carrying out a sample measurement process of measuring the absorbance of the orange complex by using a spectralphotometer, and measuring the content of the ferric iron: dissolving the ferrous phosphate lithium anode material by using the acid; when the pH of the solution is 1-3, adding sulfosalicylic acid to produce a purple complex with the ferric iron; and measuring the absorbance of the purple complex by using the spectralphotometer. The invention also discloses a protective atmosphere device designed to test the method for measuring the content of the ferrous iron and the ferric iron, which comprises an admission pipe, an exhaust pipe, a box body, an operating space and operating gloves, wherein the operating space is filled with inert gases, such as argon, helium, neon, nitrogen and carbon dioxide.
Owner:NANOCHEM SYST SUZHOU
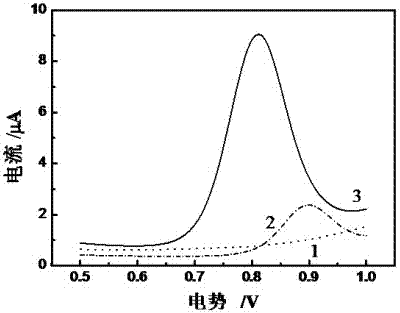
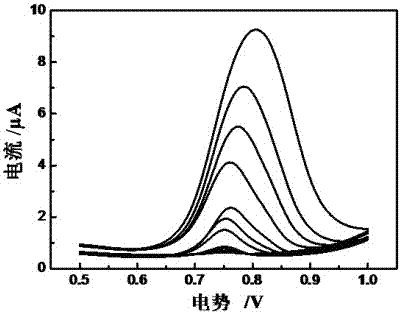
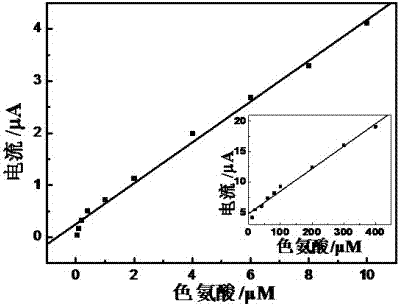
Method for preparing electropolymerized sulfosalicylic acid modified glassy carbon electrode and application of glassy carbon electrode in measurement of tryptophan
InactiveCN102507686AIncreased current responseHigh sensitivityMaterial electrochemical variablesPhosphateSalicylic acid
The invention relates to a method for preparing an electropolymerized sulfosalicylic acid modified glassy carbon electrode, and use of the glassy carbon electrode as a tryptophan electrochemical sensor in high-sensitivity measurement of tryptophan in compound amino acid injection, and belongs to the technical field of electrochemical analysis and detection. The invention mainly aims to prepare the electropolymerized sulfosalicylic acid modified glassy carbon electrode to perform sensitive quantitative analysis and measurement on the tryptophan by a differential pulse voltammetry. Experimental results show that: the modified electrode has an obvious sensitization effect on the tryptophan in a phosphate buffer solution at the concentration of 0.1mol / L and the pH of 3.5, and under the optimal conditions, the differential pulse voltammetry is adopted for measurement, and the tryptophan at the concentration in a range of 5.0*10<-8>-4.0*10<-4> and peak current of the tryptophan have a good linear relation, and the detection limit is 4.0*10<-8>. When the method is used for measuring the tryptophan content in a medicine, results are satisfactory.
Owner:SHANGHAI UNIV
Western blot kit for detecting antibody of autoimmune disease and preparation method thereof
ActiveCN103105489AOvercome the cumbersome operation of individual detection one by oneImprove accuracyMaterial analysisAntigenAnti-mitochondrial antibody
The invention provides a western blot kit for detecting the antibody of autoimmune disease and a preparation method of the western blot kit, and relates to a western blot kit for detecting related antibodies of various autoimmune diseases, aiming at overcoming the technical defect that a western blot product is unavailable for testing and screening various autoimmune diseases in the prior art. The nitrocellulose membrane or the nylon membrane contains at least two parallel detection lines coated by at least two of ten natural antigens or recombinant antigens, i.e. dsDNA (deoxyribonucleic acid), Sm / RNP (ribonucleoprotein), CCP (critical compression pressure), SSA (sulfosalicylic acid), SSB (single-strand binding protein), GAD (glutamic acid decarboxylase), ICA (islet cell antibody), IA-2A (islet cell), TG (triglyceride) and AMA-M2 (anti-mitochondrial antibody), a high-concentration quality control band, a median-concentration quality control band and a low-concentration quality control band. The deficiency of the detection sensitivity and the specificity of the single autoantibody can be overcome, the operating complexity for independently detecting the related autoantibody of various diseases one by one can be overcome, and the detection efficiency and the result judging accuracy degree can be greatly improved.
Owner:SHENZHEN YHLO BIOTECH
Preparation method of composite acids doped with conductive polyaniline
The preparation method for conductive polyaniline with doped-composite acid is characterized in that, adding the aniline monomer, dopant, solvent, oxidant, and acidic solution into reactor to mix rapidly and form emulsion; controlling strictly the reaction condition; then, adding demulsifying agent, clearing, filtering, baking, and grinding to solid powder; adjusting the composite proportion between sulfosalicylic acid and type of other assist doped acid to control effectively the conductivity. This product can be used in antistatic material, EM shielding material and electronic element package.
Owner:CHONGQING UNIV
Preparation method for dendritic-nanometer-structure polyaniline air-sensitive sensor
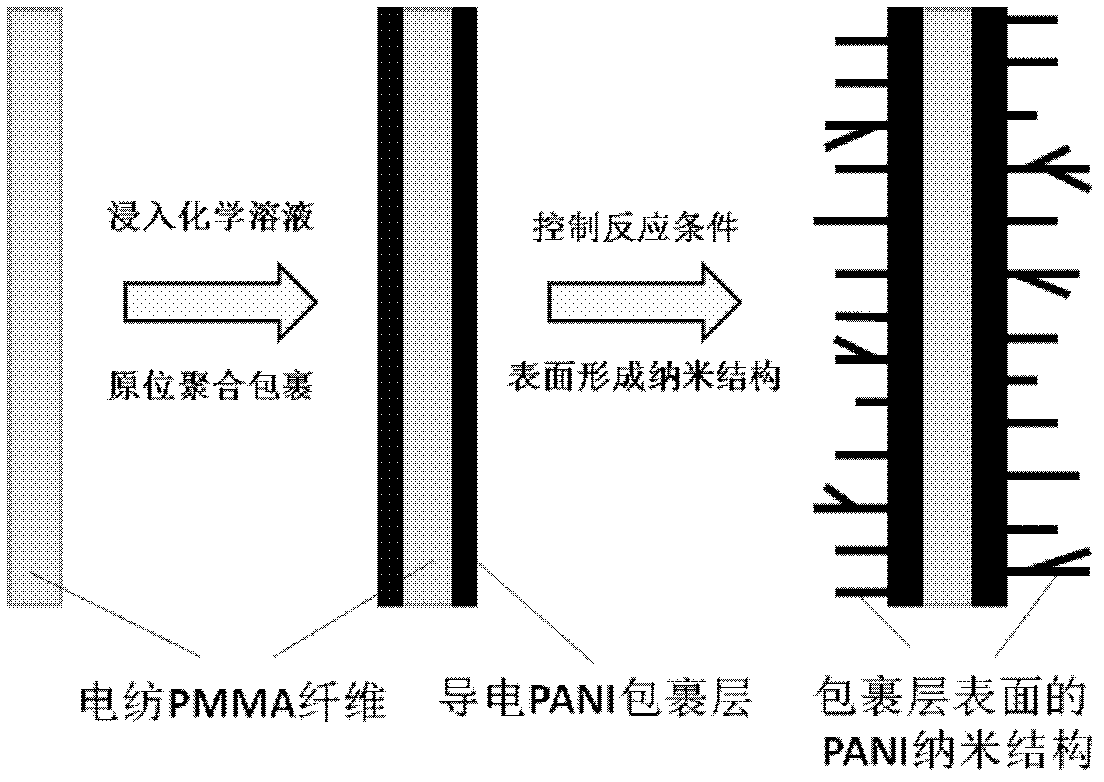
InactiveCN102561042AHigh sensitivityImprove response speedFibre typesMaterial resistanceFiberIn situ polymerization
The invention belongs to the technical field of sensors and relates to a preparation method for a dendritic-nanometer-structure polyaniline air-sensitive sensor. The preparation method comprises the steps of preparing polymethyl methacrylate (PMMA) nanometer fiber (namely an insulation high polymer material) by an electrostatic spinning method, immersing the PMMA nanometer fiber serving as a reaction template or a basement into polyaniline reaction solution doped with sulfosalicylic acid, wrapping a layer of conduction polyaniline on the surface of the PMMA nanometer fiber by in-situ polymerization, and forming a dendritic nanometer structure in self assembly on the surface of the polyaniline wrapping layer by controlling reaction time, polymerization temperature, and a reaction condition of molar ratio between an aniline monometer and a doping agent, so as to obtain coaxial-structure polyaniline fiber, wherein the contact area between the coaxial-structure polyaniline fiber and gas to be tested is large. The dendritic-nanometer-structure polyaniline air-sensitive sensor is simple in structure, small in power consumption, simple in preparation method, low in cost, good in mechanical property, and low in possibility of breaking; and as the polyaniline wrapping the outer layer of the coaxial polyaniline fiber has a coral-antenna-shaped dendritic structure, the surface of the sensor, which is in contact with the gas, is enlarger, so that the sensitivity and response speed of the sensor are high.
Owner:QINGDAO UNIV
Rare earth modified brushing electroplating silver plating solution and preparing technology and use method of solution
ActiveCN106283134AUniform and dense appearanceBright appearanceN dimethylformamideHexamethylenetetramine
The invention provides a rare earth modified brushing electroplating silver plating solution. The rare earth modified brushing electroplating silver plating solution comprises following components including, by mass concentration, 10 g / L to 30 g / L of silver salt, 0.1 g / L to 2 g / L of modifying agent rare earth cerous nitrate, 50 g / L to 200 g / L of coordination agent sulfosalicylic acid, 50 g / L to 200 g / L of imidazole, 0.01 g / L to 1 g / L of protection agent hexamethylenetetramine, 0.01 g / L to 1 g / L of addition agent N,N-dimethylformamide, 1 g / L to 30 g / L of polyethylene glycol, 10 g / L to 50 g / L of ammonium hydroxide, 10 g / L to 40 g / L of stabilizing agent ammonium acetate and 5 g / L to 30 g / L of potassium pyrophosphate. The pH value of the solution is adjusted to range from 6.8 to 7.2 through dilute nitric acid or dilute sodium hydroxide. The invention further provides a preparing technology and use method of the rare earth modified brushing electroplating silver plating solution. The harmfulless silver salt, a rare earth modification agent, a coordination agent, a protection agent and the like are mixed for forming the solution, the solution is a neutral cyanide-free silver plating solution, raw materials are easily obtained, and the preparing technology is simple. A product is applied to a copper or copper alloy surface to be subject to brushing electroplating, the method is simple and easy to operate, and a plating is stable.
Owner:STATE GRID CORP OF CHINA +1
Chemical polishing method of stainless steel by high-temperature oxidation
InactiveCN103046053AIncrease loading capacityFast and effective mechanical peeling actionCavitationPhosphoric acid
The invention discloses a chemical polishing method of stainless steel by high-temperature oxidation, and belongs to the field of surface treatment of stainless steel parts. The method comprises the following steps: using phosphoric acid, sulfuric acid and nitric acid as main materials; supplementing additives: polyethylene glycol, sulfosalicylic acid and nicotinic acid which are used to prepare a chemical polishing liquor; introducing an ultrasonic cleaning instrument in the chemical polishing process; and polishing the surface of a stainless steel part within a short time by means of cavitation of ultrasonic wave in the polishing liquor. The chemical polishing method provided by the invention is high in polishing efficiency, less in pollution and strong in operability. Polished parts are bright and white in surface, and uniform and meticulous. The method is especially suitable for polishing treatment of stainless steel parts with precision screws, high precision sizes, deep holes, blind holes, superfine holes and complex structures.
Owner:中国兵器工业第二一三研究所
Detection method for trace protein of Chinese medicinal injection
InactiveCN101865926AIncreased sensitivityImprove featuresMaterial analysis by observing effect on chemical indicatorBiological testingSpecific adsorptionOrganic solvent
The invention discloses a detection method for trace protein of a Chinese medicinal injection, which is used for detecting the protein impurities of the Chinese medicinal injection so as to control the quality of the Chinese medicinal injection. The detection method is characterized by comprising the following steps of: 1) applying a Chinese medicinal injection sample on a protein specific adsorption film; 2) washing the sample application film with organic solvent-containing solution to wash away the natural color of dots of the Chinese medicinal injection; and 3) subsequently performing coomassie brilliant blue staining on the sample application film. If the protein contained in the Chinese medicinal injection is no less than 12mu g / ml, stable and differentiable blue can be displayed on the film. The detection sensitivity of the method is higher than that of a sulfosalicylic acid precipitation method, and the detection method has high anti-interference capacity and is suitable for the detection of the trace protein of all the Chinese medicinal injections.
Owner:YUNNAN UNIV OF TRADITIONAL CHINESE MEDICINE
Edible oil peroxide value measurement combined reagent and detection method
InactiveCN103245660AReduce typesReduce distractionsMaterial analysis by observing effect on chemical indicatorOil and greaseSulfosalicylic acid
The invention relates to measurement of peroxide values, and in particular relates to an edible oil peroxide value measurement combined reagent and a detection method. The edible oil peroxide value measurement combined reagent comprises a polarity organic solvent, a reducing agent, a color development reagent and a color development stabilizing agent, wherein the polarity organic solvent comprises ethanol with the mass concentration of 237-710 g / L in percentage by mass; the reducing agent comprises ferrous sulfate with the mass concentration of 1-20 g / L in percentage by weight; the color development reagent is a sulfosalicylic acid solution with the mass concentration of 300-1,000 g / L; and the color development stabilizing agent is a pyridoxamine dihydrochloride solution with the mass concentration of 20-800 g / L. By a colorimetric analysis method, the operation is quick and simple, and interference caused by heating on a detection result is reduced; the types of the needed detection reagents is few, and the reagents are convenient to combine and carry; and the edible oil peroxide value measurement combined reagent can develop and detect a plurality of oil samples at the same time, is uniform in developing, is low in toxicity and even non-toxic, and is safe and environmentally-friendly.
Owner:ZHEJIANG UNIV
Method for plating zinc and nickel on neodymium-iron-boron material
InactiveCN104233408AImprove corrosion resistanceReduce magnetic lossElectrogalvanizationSulfosalicylic acid
The invention relates to a method for plating zinc and nickel on a neodymium-iron-boron material, and belongs to the field of alloy plating processes. The method comprises the following steps of: (1) firstly carrying out roll finish chamfer treatment on the neodymium-iron-boron material by adopting vibrating type roll finish; (2) placing the neodymium-iron-boron material into a deoiling solution and carrying out ultrasonic deoiling; (3) then carrying out derusting treatment by adopting a nitric acid solution with the volume fraction of 10%; (4) carrying out hole sealing treatment by using a low-temperature particule plugging agent; (5) then carrying out activating treatment under the condition of room temperature by using a sulfosalicylic acid solution; (6) plating the neodymium-iron-boron material subjected to the treatment; (7) carrying out polishing treatment on the plated neodymium-iron-boron material, and then carrying out zinc-nickel alloy plating layer passivating treatment; (8) finally carrying out subsequent impregnation treatment by adopting passivated impregnating resin. The method for plating zinc and nickel on the neodymium-iron-boron material, which is disclosed by the invention, greatly enhances the corrosion-resistant capacity of a neodymium-iron-boron surface after the neodymium-iron-boron surface is treated and reduces the magnetic loss through the improvement of a process and has the advantages of simple process and easiness for popularization and use.
Owner:XIAN SANWEI SECURITY TECH
Method for promoting fast germination and enhancing salt tolerance of rice seeds in seashore saline land
InactiveCN105075453APromote germinationImprove salt resistanceGerminating apparatusWater immersionSulfosalicylic acid
The invention discloses a method for promoting fast germination and enhancing salt tolerance of rice seeds in seashore saline land. The method includes the steps that firstly, an aqueous hydrogen peroxide solution with the mass fraction being 10%-15% is used for soaking the rice seeds for 20-30 minutes; then the rice seeds are soaked by water for 8-10 hours, and then a NaCl aqueous solution of 2-5 mmol / L is used for soaking the rice seeds subjected to water immersion for 15-30 minutes; afterwards, the rice seeds are soaked for 1-2 hours by an aqueous solution including proline of 20-45 mmol / L and sulfosalicylic acid of 0.5-2 mmol / L, and then the rice seeds are soaked for 1-2 hours by an aqueous gibberellin solution of 70-200 mg / L; then the soaked rice seeds are germinated, and the germinated rice seeds are sown in a seashore saline land field. By the adoption of the method, the rice seeds are subjected to long-termed salt stress treatment, and then the mixed solution prepared from the proline and the sulfosalicylic acid and gibberellin are used for soaking the seeds, so that the germination capacity and salt tolerance of the rice seeds are improved quickly, and the bottleneck problem that the emergence rate of direct seeding rice seeds in the seashore saline land is low is solved. The method has great practical application value.
Owner:QINGDAO AGRI UNIV
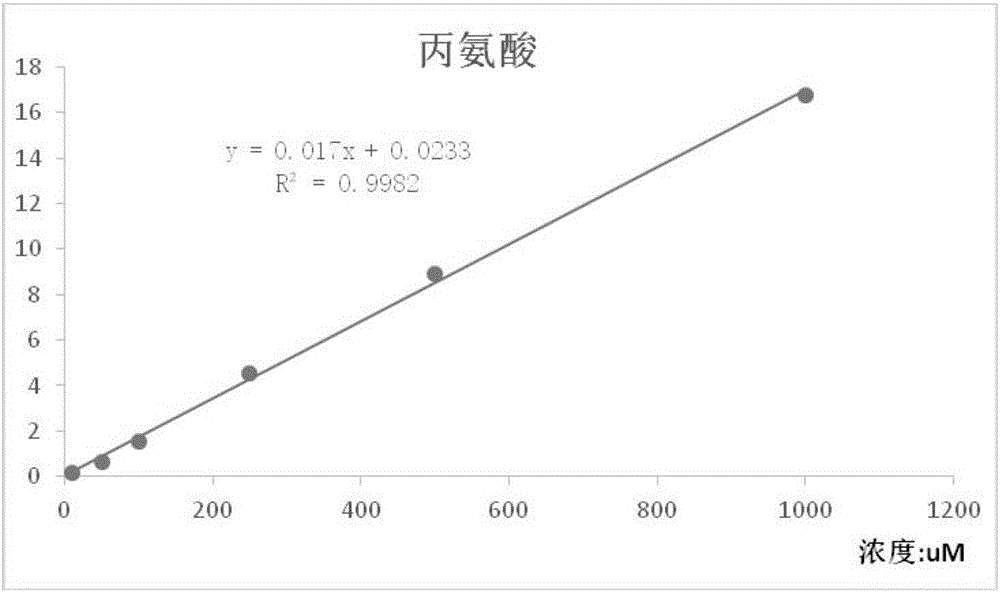
Liquid mass spectrometry method for detecting various amino acids in human blood plasma in underivatized mode
InactiveCN106442758AEasy to handleShort analysis timeComponent separationPerfluoroheptanoic acidGradient elution
The invention discloses a liquid mass spectrometry method for detecting various amino acids in human blood plasma in an underivatized mode. The method includes the following steps that firstly, the human blood plasma is obtained and subjected to deproteinization with sulfosalicylic acid, and supernate is obtained; secondly, the supernate is mixed with an internal standard solution containing a volatile ionized reagent, LC-MS / MS is used for analysis, wherein according to chromatographic conditions, 1 mM perfluoroheptanoic acid aqueous solution serves as a mobile phase A, a 1 mM perfluoroheptanoic acid acetonitrile solution serves as a mobile phase B, and gradient elution is conducted; according to mass spectrum conditions, multi-ion reaction monitoring of positive ion electrospray ionization is conducted. A volatile ionized reagent sample is added to a sample and mobile phase system, analysis time is short, analysis efficiency is improved, operation is easy, and accuracy of experiment results is improved. LC-MS / MS is used for detecting twenty amino acids in the human blood plasma, specificity, sensitivity and flux are high, operation is easy, sample processing is easy, detection time is short, and the method is particularly suitable for clinical application and popularization.
Owner:陈大为
Chromium-free passivating agent for galvanized plate with organosilane as main component and preparation process thereof
ActiveCN102732869AImprove corrosion resistanceImprove coating adhesionMetallic material coating processesSodium molybdateSalicylic acid
Owner:合肥华清高科表面技术股份有限公司
Method for preparing conductive non-woven fabric by using in-situ polymerization method
ActiveCN102251396AImprove stabilityStable resistanceFibre treatmentIn situ polymerizationPolypyrrole
The invention relates to a method for preparing conductive non-woven fabric by using an in-situ polymerization method, comprising the following steps of: impregnating the non-woven fabric in a solution consisting of pyrrole, anthraquinone-2-sodium sulfonate or anthraquinone-2,7-sulfonic acid sodium salt and water; dropping a solution consisting of ferric nitrate, 5-sulfosalicylic acid and water into the mixed solution in which the non-woven fabric is impregnated; in the presence of the oxidation of the ferric nitrate, subjecting the pyrrole to in-situ polymerization to synthesize polypyrrole;and forming a conductive layer on the surface of the non-woven fabric through adsorption precipitation of the polypyrrole. The preparation method disclosed by the invention has the advantages of simpleness, low cost, no pollution and mild reaction condition and is suitable for large-scale industrial production. The conductive non-woven fabric prepared by using the preparation method has favorableconductivity, electromagnetic shielding performance, flexible characteristics, and the like.
Owner:武汉飞帛丝科技有限公司
Method for extracting lanthanum from coal ash
ActiveCN105969994ASimple methodEasy to operateProcess efficiency improvementRare-earth elementFiltration
The invention provides a method for extracting lanthanum from coal ash. The method comprises the following steps that 1, coal ash is acidified and dissolved through hydrochloric acid to be prepared into an acidic leaching solution, particularly, the coal ash and the hydrochloric acid are mixed and reacted, then, filtration is carried out, and the acidic leaching solution is obtained; 2, sulfosalicylic acid is added into the acidic leaching solution prepared in the step 1 for a complex reaction, then, an extraction agent is added for extracting and separating the rare earth element in the acidic leaching solution, and hydrochloric acid is added into the extraction phase for reextracting and separating the rare earth element in the extraction phase, and a rare earth mixed solution is obtained; and 3, resin is extracted for separation to obtain a lanthanum solution, particularly, the impregnating resin is prepared, the rare earth mixed solution obtained in the step 2 is used for extracting the resin to separate lanthanum, a leacheate is used for leaching the impregnating resin with lanthanum adsorbed, and a lanthanum solution is obtained. The method for extracting the lanthanum from the coal ash is simple and easy to operate, lanthanum can be extracted from coal ash, and the extraction rate and extraction purity are high.
Owner:CHINA SHENHUA ENERGY CO LTD +1
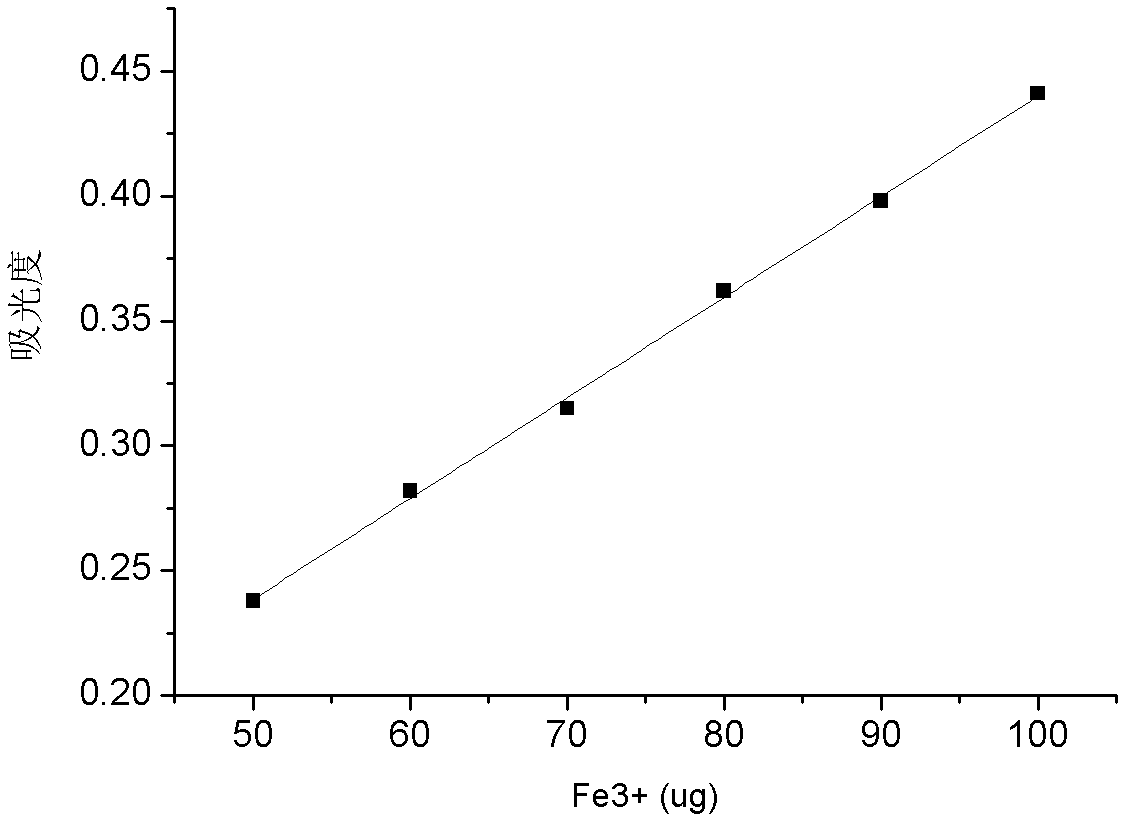
Ferric ion stabilizing capability measurement method of acidification ferric ion stabilizer
ActiveCN102519894AEasy to operateEvaluation of the ability to achieve stable iron ionsColor/spectral properties measurementsCentrifugationPhysical chemistry
The invention relates to a ferric ion stabilizing capability measurement method of an acidification ferric ion stabilizer, which comprises the steps that: excess standard ferric ion solution is added into a certain amount of ferric ion stabilizer, so complex reaction is generated between all the ferric ion stabilizer and the standard ferric ion solution, then the pH is adjusted to between 5-6, the redundant Fe<3+> is hydrolyzed to generate the Fe(OH)3 flocculent precipitate, the precipitate is removed through centrifugation, all the supernatant is collected, and in the acid medium, a sulfosalicylic acid spectrophotometric method is utilized to test the total ferric content which is stabilized by the ferric ion stabilizer. The invention has the following advantages that: 1, the adopted method is simple and quick to operate, and has good repeatability and high precision; 2, the problems of multiple repeated heating, repeated diluting, finish determination and the like of the liquid do not exist in the adopted method, and the personal factor is prevented from influencing the test result; and 3, the test precision of the ferric ion content is high, and the test of the ferric ion is benefited.
Owner:SHAANXI RES DESIGN INST OF PETROLEUM CHEM IND
Cleaning agent for development equipment and use method thereof
ActiveCN103046061AExtended service lifeReduce corrosionPhotosensitive material processingPhosphatePhosphoric acid
The invention discloses a cleaning agent for development equipment. The cleaning agent comprises 5-30 parts by weight of a main agent of and 3-15 parts by weight of an auxiliary agent, wherein the main agent consists of one or more of sulfonamic acid, sulfosalicylic acid, citric acid, phosphoric acid and glycollic acid; the auxiliary agent consists of one or more of oxalic acid, ammonium bifluoride, maleic anhydride and polyethylene phosphate. The cleaning agent consisting of the main agent and the auxiliary agent in weight ratio is configured into a cleaning solution to clean the development equipment, so that scales and crystals in the development equipment are effectively removed, meanwhile, the corrosion rate of the cleaning solution to the development equipment is reduced during cleaning, the development equipment is protected, and the service life of the development equipment is prolonged.
Owner:HUANGSHAN YINJIANG TECH
Titanate alloy steel anticorrosive coating and preparation method thereof
InactiveCN106147592AUniform particlesFine grainAnti-corrosive paintsEpoxy resin coatingsEpoxyPolyamide
The invention discloses a titanate alloy steel anticorrosive coating. The titanate alloy steel anticorrosive coating consists of the following raw materials in parts by weight: tetrabutyl titanate 4-6 parts, polysorbate 80 0.8-1 part, aniline 50-60 parts, pyrroles 30-40 parts, sulfosalicylic acid 7-9 parts, epoxy resin E51 3-4 parts, ammonium persulfate 1.3-2 parts, molybdic acid 4-6 parts, polyamide resin 3-5 parts, n-butyl alcohol 160-170 parts, butyl glycol ether 20-30 parts, polyadipate ethylene glycol 3-5 parts, zirconium ammonium carbonate 0.7-1 part, methyl stannum mercaptide 1-2 parts, kt101 titanate coupling agent 0.6-1 part, octyl and decyl glycerate 0.2-0.4 part, and aluminium triphosphate 1.6-2 parts. The titanate alloy steel anticorrosive coating is modified by the titanate coupling agent, and therefore, the surface hydrophobicity and the weather fastness of the finished coating are improved effectively.
Owner:TIANCHANG YINHU PAINT
Tin plating electrolyte
The invention discloses tin plating electrolyte. The tin plating electrolyte comprises the following components 60 to 90g / L of tin methane sulfonate, 10 to 30g / L of tin sulfate, 110 to 140mg / L of p-cresolsulphonic acid, 60 to 120mg / L of sulfosalicylic acid, 30 to 50g / L of phosphinic acid, 45 to 70mg / L of 2,2-PySSPy, 15 to 25mg / L of 8-hydroxyquinoline, 20 to 30mg / L of catechins, 10 to 15g / L of 2,2-dihydroxy diethyl sulfide, 5 to 10mg / L of hexadecylpyridinium bromide, 8 to 12 g / L of dodecyl-phenol polyoxyethylene ether, 5 to 10g / L of ethylene glycol, 4 to 8g / L of benzalacetophenone, 5 to 10g / L of sodium hexametaphosphate, 4 to 8g / L of sodium dodecyl sulfate, and the balance of deionized water. According to the tin plating electrolyte, Sn2<+> can be effectively restrained to convert into Sn4 <+> in an electrolytic process by improving an electrolyte formula, and the stability of plating solution is improved; and meanwhile, the throwing power of the electrolyte is increased, and the luminance, the corrosion resistance and the welding performance of tin coatings are improved.
Owner:郎溪县金科金属有限公司
Environment-friendly derusting conditioning fluid for metal equipment of flue-curing barn
InactiveCN102703914AAchieving processing powerAchieve anti-rust one-time finishMetallic material coating processesEnvironmental resistanceZinc hydroxide
The invention relates to environment-friendly derusting conditioning fluid for metal equipment of a flue-curing barn. By the environment-friendly derusting conditioning fluid, the technical problem of poor derusting effect or high cost and the like in the conventional deruster is solved. According to the key point of the technical scheme, the environment-friendly derusting conditioning fluid is prepared by mixing and stirring the following raw materials in part by weight: 50 to 70 parts of deionized water, 20 to 30 parts of orthophosphoric acid, 1 to 3 parts of zinc hydroxide, 2 to 4 parts of manganous dihydrogen phosphate, 0.5 to 1.5 parts of sodium tripolyphosphate, 1 to 3 parts of sulfosalicylic acid, 1 to 3 parts of sodium nitrite, 2 to 4 parts of boric acid, 0.5 to 1.5 parts of sodium molybdate and 0.5 to 1.5 parts of polyvinyl alcohol. By the environment-friendly derusting conditioning fluid, rust melting and rust proofing are completed at one time, so that the labor production efficiency is improved greatly, and the cost of conditioning rust is reduced. The environment-friendly derusting conditioning fluid can be widely applied to a surface treatment process for steel devices.
Owner:湖南省烟草公司永州市公司
Microcrystalline phosphating solution for brake block hardware
InactiveCN101348906AImprove pollutionLittle pollutionMetallic material coating processesPhosphateNitrobenzene
The invention provides a pollution-free microcrystalline phosphating treatment solution for brake pad hardware, which is mainly made from the following raw materials in portion by weight: 40 to 50 portions of zinc nitrate, 22 to 30 portions of zinc dihydrogen phosphate, 30 to 40 portions of Mazhev salt (manganous dihydrogen phosphate), 1.5 to 2 portions of guanidine nitrate, 1.5 to 2 portions of sodium m-nitrobenzene sulfonate, 1 to 2 portions of sodium tripolyphosphate, 2 to 4 portions of tartaric acid, 2 to 3 portions of sulfosalicylic acid, 1.5 to 2.5 portions of citric acid and 1,000 to 1,200 portions of water. The solution is used for the blackening-phosphating treatment of the surface of iron hardware, and has the characteristics of fast phosphorization, good performances, little environmental pollution, low cost, etc. The treatment solution has the advantages of fine phosphatized crystals of phosphating films, uniform color in consistent black and good corrosion resistance. Especially, as the treatment solution chooses the raw materials with little environmental pollution and high efficiency, the treatment solution acquires excellent effects in both phosphatization and environmental protection through optimized combination and reasonable proportioning.
Owner:RUIYANG AUTOMOTIVE MATERIALS XIANTAO
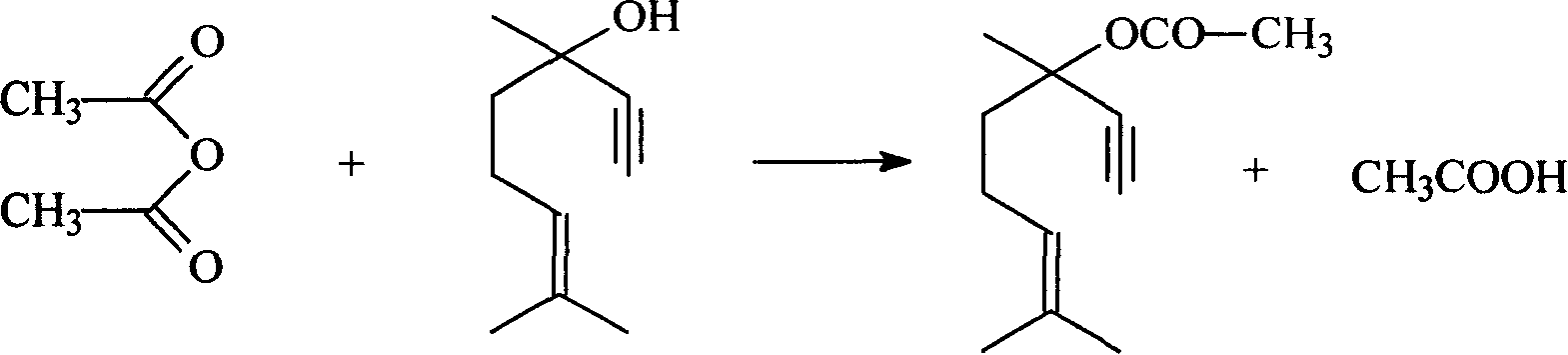
Method for preparing dehydrolinalyl acetate from dehydrolinalool
InactiveCN101209965AHigh reactivityShort reaction timeOrganic compound preparationCarboxylic acid esters preparationAcetic acidAcetic anhydride
The invention relates to a method for preparing dehydrolinalyl acetate from dehydrolinalool. The method comprises esterification between dehydrolinalool and acetic anhydride is carried out in the presence of catalyst so as to obtain dehydrolinalyl acetate. Molar ratio of raw material provided of dehydrolinalool to acetic anhydride is 1: (1-5); reaction temperature is 25-70 DEG C; reaction pressure is normal pressure; reaction time is 20-150min; the catalyst is sulfosalicylic acid, dosage of which is 0.05-5wt percentage of total amount of the raw material provided. Compared with the prior art, the invention has the advantages that reaction activity of the adopted esterification catalyst is high, which is reflected by moderate reaction temperature and short reaction time, raw material conversion and product selectivity, etc. are ideal and removal of the catalyst from the product is convenient.
Owner:SINOPEC SHANGHAI PETROCHEMICAL CO LTD
Etching solution composition for copper layer and titanium layer and method of preparing array substrate for liquid crystal display using the same
ActiveCN104911592AWon't change quicklyImprove stabilityNon-linear opticsLiquid-crystal displaySulfosalicylic acid
The invention discloses an etching solution composition for a copper layer and a titanium layer and a method of preparing an array substrate for a liquid crystal display using the same. The etching solution composition comprises persulphate and sulfosalicylic acid, so that the multiple metallic layers including copper layers and titanium layers can be uniformly etched in at a high speed in an environmental-friendly way. Excellent thermal control performance and time stability are ensured.
Owner:DONGWOO FINE CHEM CO LTD
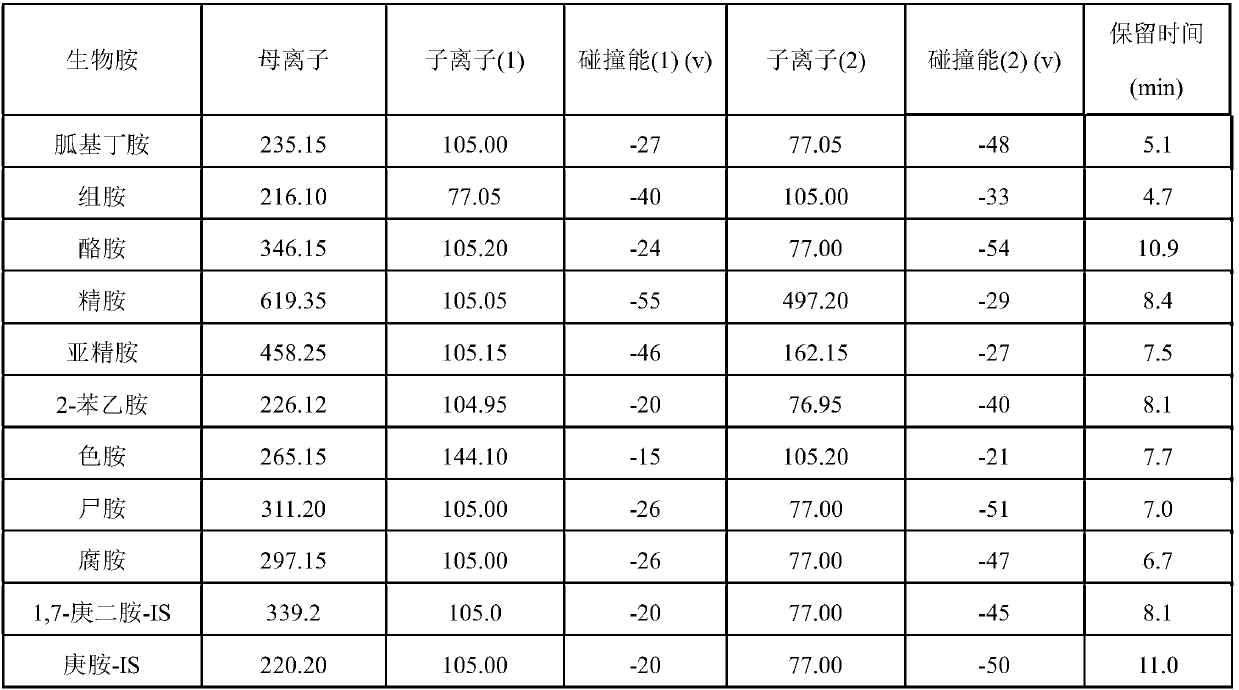
Rapid analysis method of biogenic amine in fish
ActiveCN107782834AReduce the impactHigh sensitivityComponent separationTryptamineSulfosalicylic acid
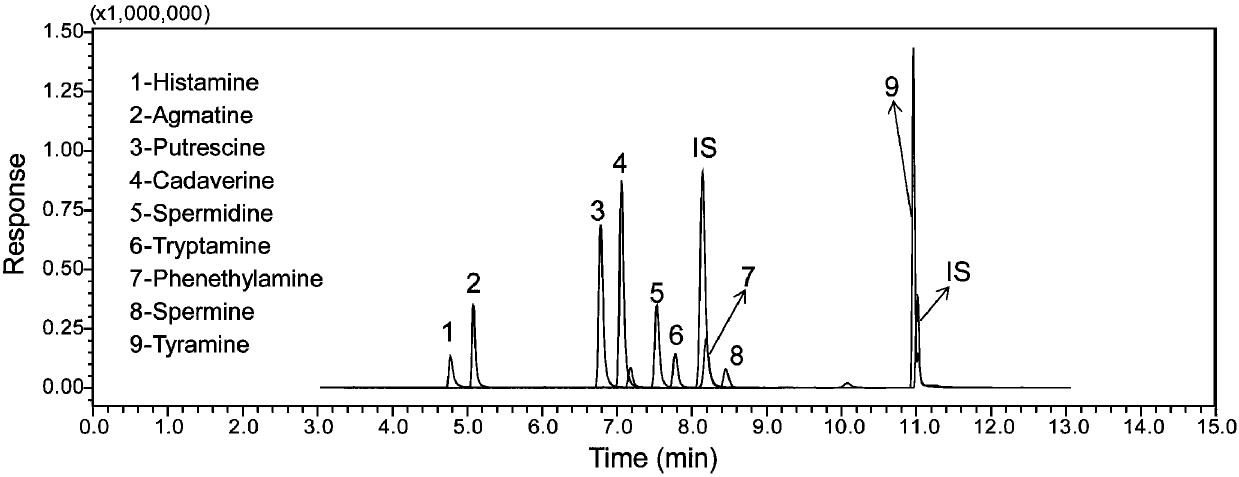
The invention discloses a rapid analysis method of biogenic amine in fish. The common biogenic amine (nine types of biogenic amine comprising agmatine, histamine, tyramine, spermine, spermidine, 2-phenylethylamine, tryptamine, cadaverine and putrescine) is analyzed in a targeted manner by taking 5-sulfosalicylic acid as an extraction solvent, adopting bead agitating and grinding as an extraction method, taking benzoyl chloride as a derivatization reagent and adopting an LC (liquid chromatogram)-triple tandem quadrupole mass spectrometry. The biogenic amine is detected in a multi-reactions monitoring manner by taking ion pairs with optimum response, which are obtained by optimization, as quantitative ion pairs and taking ion pairs with following response as auxiliary qualitative ion pairs.According to the method, the sample extraction time and the biogenic amine derivation time can be greatly shortened, so that the analysis time of the whole analysis process is greatly shortened. The analysis method has the advantages of being simple in sample pretreatment, rapid in derivation, high in detection sensitivity, high in repeatability and the like.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Clear fracturing fluid
ActiveCN102643637AEasy to make and useGood anti-expansion effectDrilling compositionFracturing fluidPotassium
The invention relates to clear fracturing fluid, which is used for fracturing treatment of a hypotonic and water-sensitive reservoir, and comprises the following components in percentage by weight: 0.5-5 percent of dimethyl octadecyl ethoxy quaternary ammonium nitrate; 0.2-2 percent of sulfosalicylic acid triethanolamine ester; 0.5-2 percent of potassium chloride; and the balance of water, wherein the weight percentage of the components is a hundred percent. A preparation method comprises the following steps of: adding clear water into a mixing liquid tank, adding the potassium chloride and the sulfosalicylic acid triethanolamine ester under a stirring condition, uniformly stirring, adding the dimethyl octadecyl ethoxy quaternary ammonium nitrate, and stirring for 15 minutes after adding the materials, so that the concentration or the viscosity of liquid in the upper part and the lower part of the tank is uniform. The clear fracturing fluid has the effects that: the clear fracturing fluid is suitable for fracturing the reservoir at the temperature of 10 DEG C-110 DEG C, the damage caused by residues of the fracturing liquid and reservoir water sensitivity can be reduced, and the crude output is improved.
Owner:PETROCHINA CO LTD