Crystal forms of (r)-n-methylnaltrexone bromide and uses thereof
a technology of r-n-methylnaltrexone and crystal forms, which is applied in the field of crystal forms of r-n-methylnaltrexone bromide, can solve the problems of severe itching, nausea, vomiting, and severe itching, and the effectiveness of opioids for pain is often limited
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of Form a
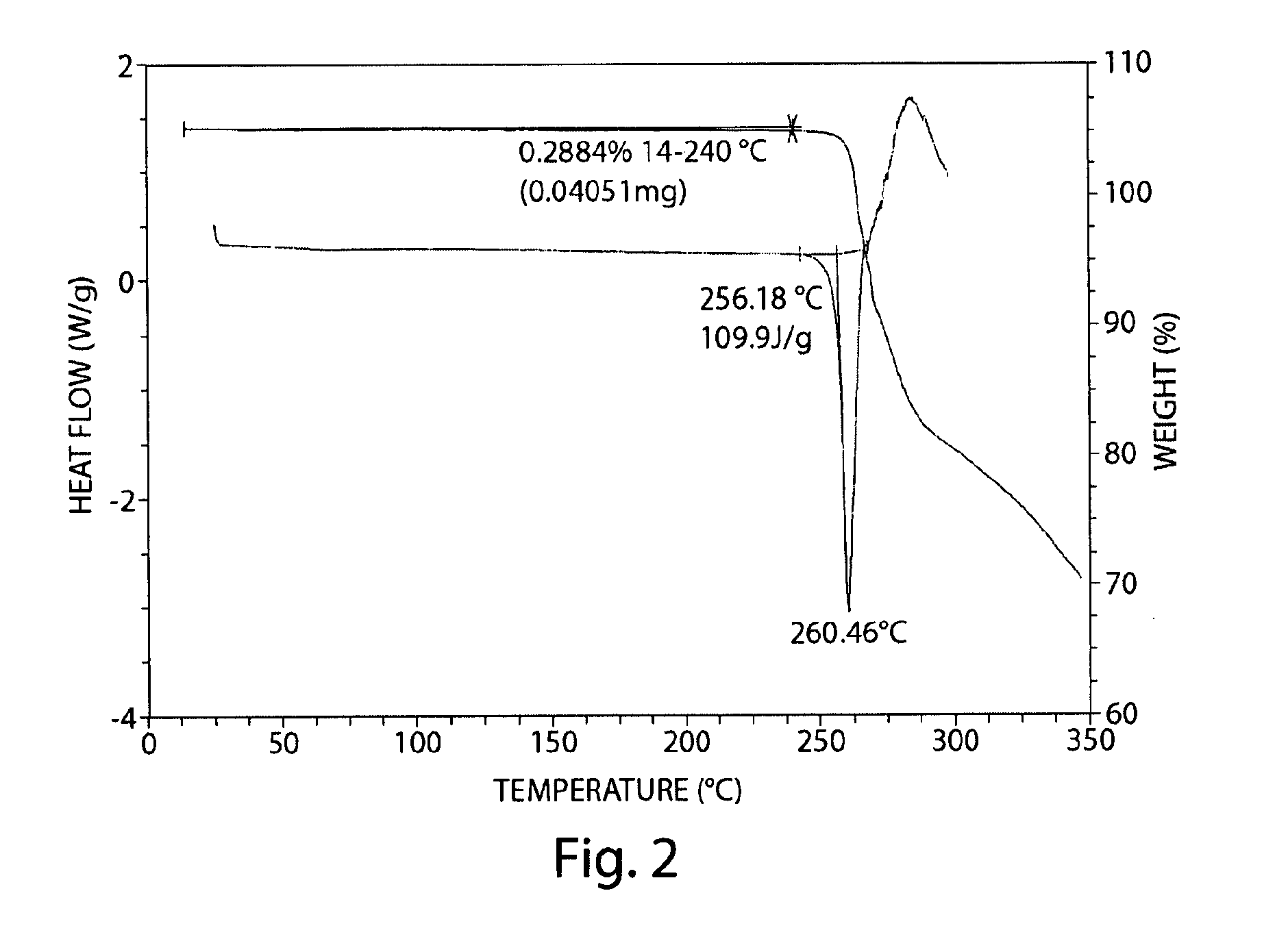
[0106]Compound 1 (54.7 mg) was dissolved in 2,2,2-trifluoroethanol (2 mL), and the solution was filtered through a 0.2 μm nylon filter into a 20-mL vial. The 20-mL vial was placed inside a 100-mL glass jar containing 5 mL of ethyl acetate. The 20-mL vial was left uncapped and the jar was capped to allow vapor diffusion to occur. After four days, single crystals were observed in solution.
[0107]The monoclinic cell parameters and calculated volume at 150° K are: a=7.9013(3)Å, b=12.7337(9)Å, c=9.4247(7)Å, ≈90.00°, β=98.868(4)°, γ=90.00°, V=936.91(10) Å3, wherein each value is ±1.5. For Compound 1, Form A, the formula weight is 435.35 g / mol with Z=2 resulting in a calculated density is 1.543 g cm−3. The space group was determined to be P21 (no. 4), this is a chiral space group. A single crystal of Form A of Compound 1 was indexed at room temperature and the unit cell parameters are summarized in Table 5, below.
TABLE 5Unit Cell Data for Form AForm AForm A150°K DataRT ...
example 2
Preparation of Form B
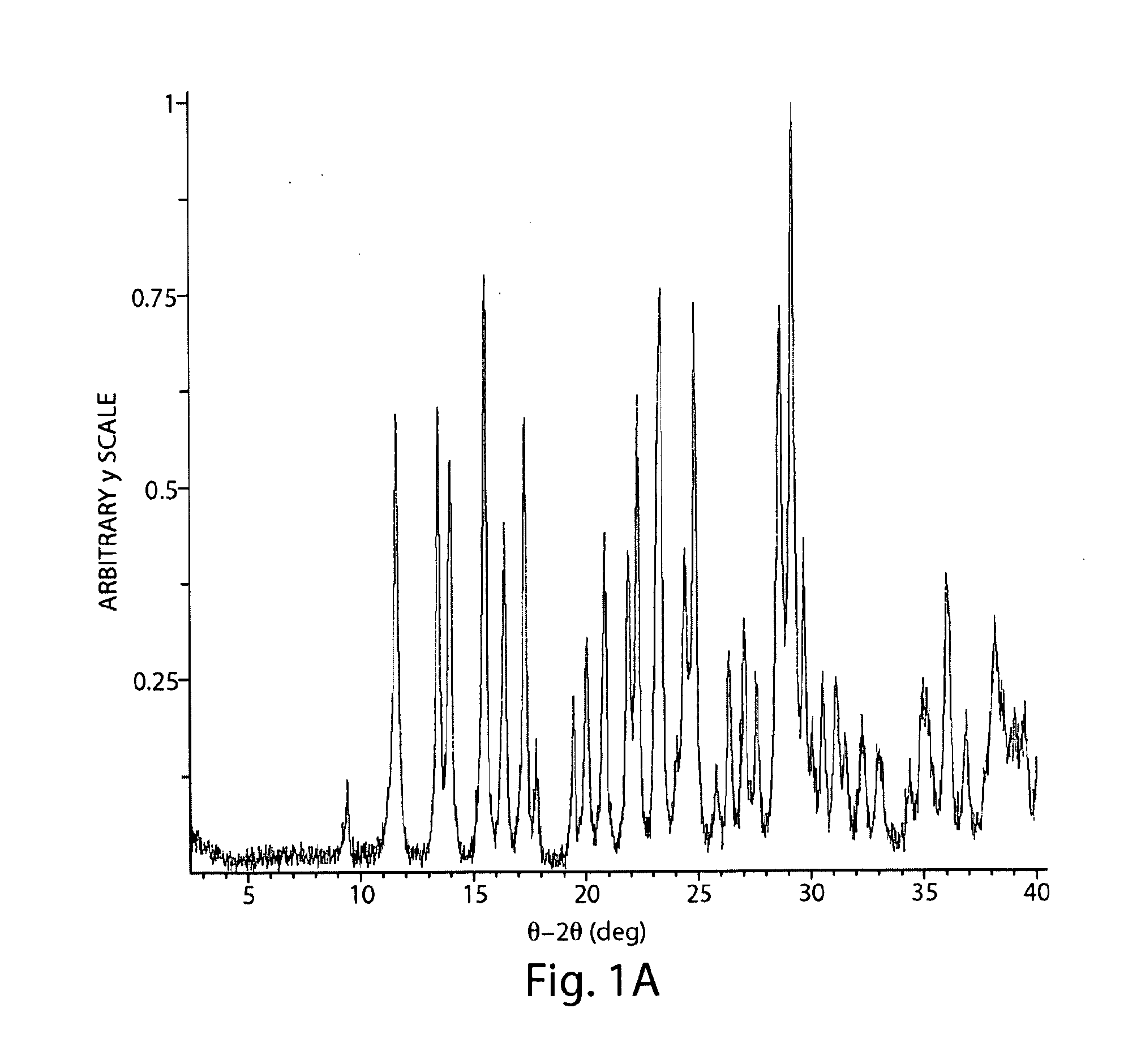
[0108]Compound 1 (52.8 mg) was dissolved in methanol (35 mL) with sonication to obtain a clear solution. The solution was filtered through a 0.2 μm nylon filter (Whatman) and evaporated using a rotary evaporator (ambient temperature bath used). The sample was left on the rotary evaporator for approximately 1 hour after the sample was visually dry. White solid containing birefringent spherulites of needles and blades resulted. An XRPD pattern of Form A+peaks was observed when the evaporation was carried out at approx. 45° C.
example 3
Preparation of Form C
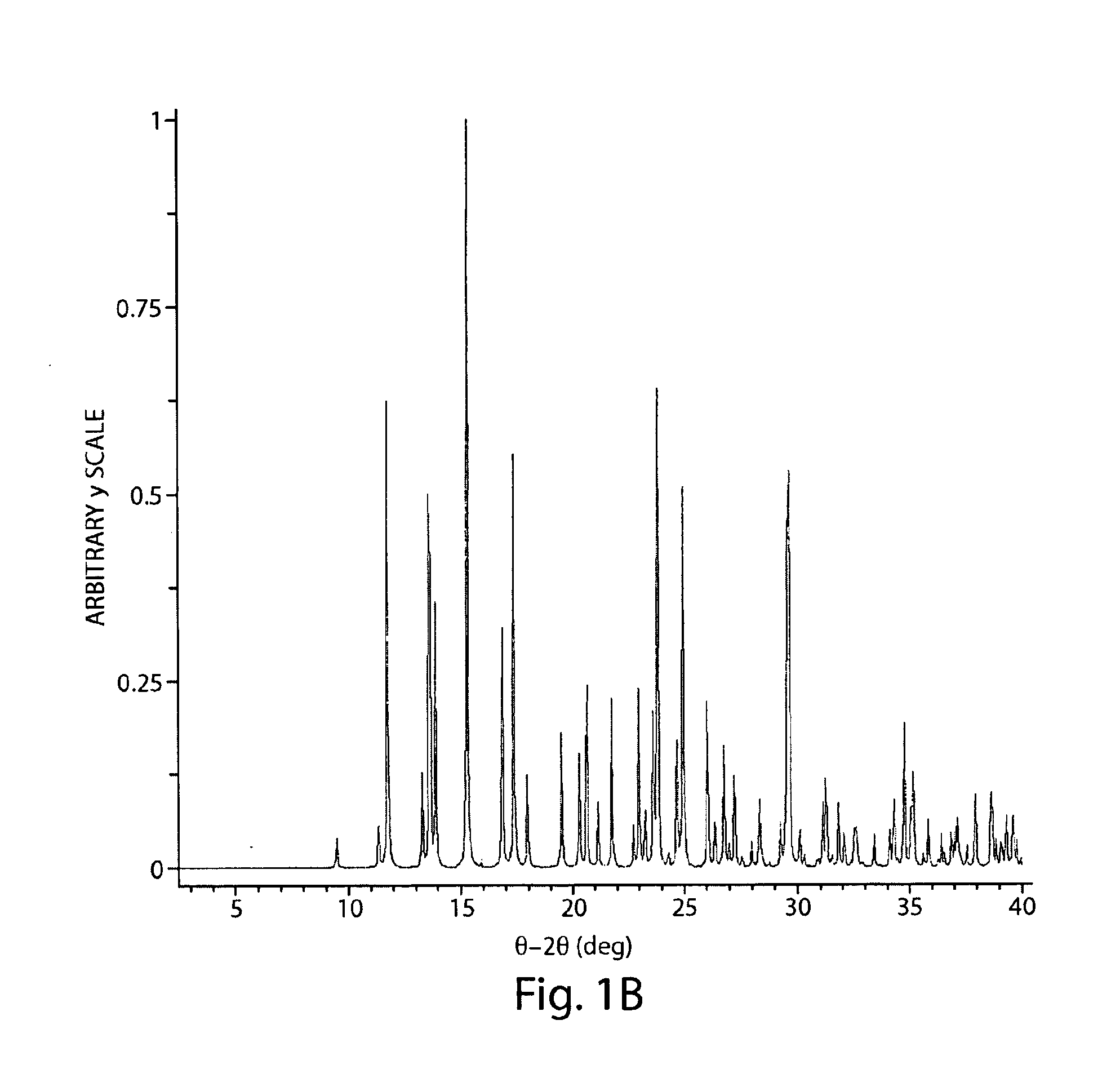
[0109]Compound 1 (321.5 mg) was dissolved in TFE (4.28 mL) with sonication. A clear solution resulted. The solution was filtered through a 0.2 μm nylon filter (Whatman) into a clean 20-mL vial. A 500 μL aliquot of the filtered solution was dispensed into a 1-dram vial. Into this vial, aliquots (500 μL) of 1-propanol were dispensed with stirring until a total of 3 mL had been added. A clear solution resulted. The solution was allowed to stand at ambient conditions for approximately 1 hour. The sample was found to contain a very small amount of white precipitate suspended in solution. It was then placed in a refrigerator. A clear solution containing colorless specks of solid resulted after 5 days. The solution was drawn off with a pipette and discarded, and the solid was allowed to air dry at ambient conditions overnight. The sample containing birefringent pentagonal plates was submitted for single crystal X-ray analysis.
[0110]The monoclinic cell parameters and ca...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More