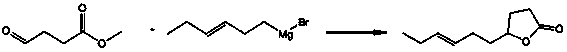Gamma-3-hexenyl-gamma-butyrolactone synthesis method
A synthetic method and technology of butyrolactone, which is applied in the direction of organic chemistry, can solve the problems of inconvenient source of raw materials and low yield, and achieve the effects of low production cost, easy operation and low price
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
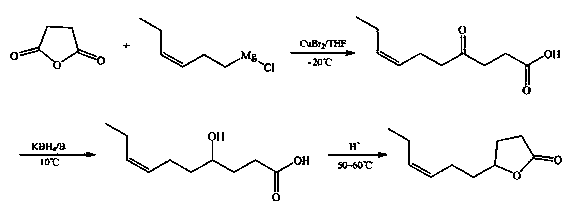
[0028] A kind of synthetic method of gamma-3-hexenyl-gamma-butyrolactone, specifically comprises the steps:
[0029] (1), Synthesis of 4-oxo-7-decenoic acid intermediate
[0030] In the reactor, add 100g (1.0mol) of succinic anhydride, 11g (0.05mol) of copper bromide and 600ml of tetrahydrofuran to obtain a mixed solution, and cool to -20°C;
[0031] Keep the temperature at -20°C, add a solution consisting of 172g (1.2mol) of 1-chloro-cis-3-hexene magnesium and 200ml tetrahydrofuran dropwise into the above mixture within 2 hours, and continue to control the temperature at - 20°C, carry out Grignard reaction for 2 hours, add 1N hydrochloric acid to the obtained reaction solution to adjust the pH=2, then filter, the obtained filtrate is extracted with chloroform, and the obtained organic layer is sequentially washed with a 10% mass percent concentration of sodium bisulfite aqueous solution 1. After washing with saturated brine, dry with anhydrous sodium sulfate, and distill the...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More