Method of detecting impurities in penehyclidine hydrochloride injection
A penehyclidine hydrochloride and a detection method are applied in the directions of measuring devices, instruments, scientific instruments, etc., and can solve the problems that the requirements of strict control of drug impurities cannot be met, the detection results can only be semi-quantitative, and the detection sensitivity is low. Achieve the effect of good accuracy, improved detection sensitivity, and high sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0044] Main instruments and chromatographic conditions:
[0045] TRACE GC MLTRA gas chromatograph, equipped with hydrogen flame ionization detector (FID), temperature 300°C, automatic sampler, Chromeleon chromatography workstation.
[0046] Chromatographic column: Rtx?-5 Amine (30m×0.25mm, 0.5μm)
[0047] Column temperature: the initial temperature is 90°C, keep for 1 minute, raise the temperature to 300°C at a rate of 8°C per minute, and keep for 8 minutes;
[0048] [0026] Column flow rate: 2. 5ml / min;
[0049] Inlet temperature: 190°C;
[0050] Split ratio: 4:1;
[0051] Injection volume: 2 μl.
[0052] Penehyclidine hydrochloride injection was provided by Chongqing Xianyang Pharmaceutical Technology Co., Ltd., specification 1ml:1mg, batch number 110900.
[0053] The impurity II reference substance was provided by Chongqing Xianyang Pharmaceutical Technology Co., Ltd., batch number 110601.
[0054] The impurity III reference substance was purchased from Sigma, batch...
Embodiment 2
[0061] Embodiment 2: Methodological verification test
[0062] According to the chromatographic conditions and operating methods of Example 1, the specificity, precision, linearity, detection limit and quantification limit, sample addition recovery rate and stability of the method were respectively verified by methodology, and the results were as follows:
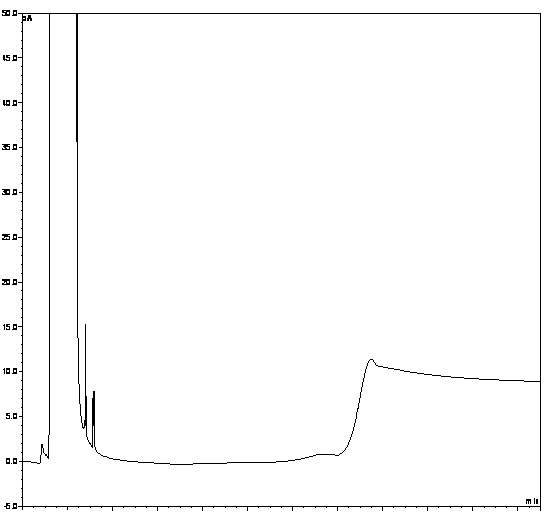
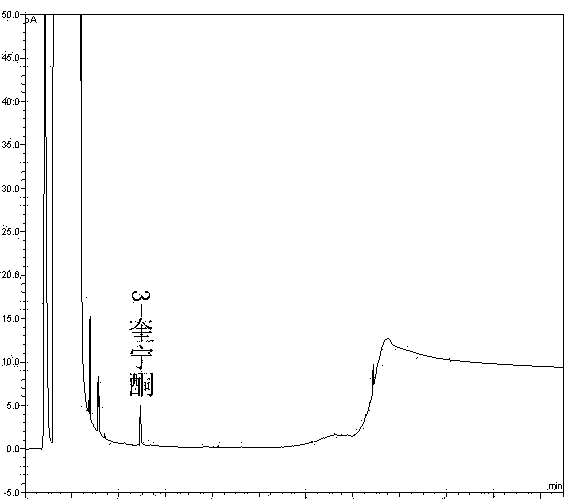
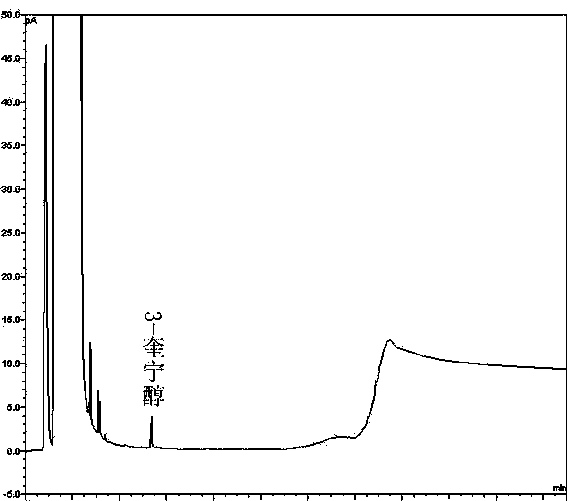
[0063] (1) Specificity:
[0064] Take the solvent DMF, the solution of the test product, the solution of the impurity reference substance II, the solution of the impurity reference substance III, the mixed solution of the test product solution and the solutions of the impurity reference substance II and III, and inject the samples in sequence. The results show that the solvent has peaks with retention times of 1.365min, 2.558min, 2.935min, and 3.326min, the retention time of impurity II is 5.121min, the retention time of impurity III is 5.543min, and the retention time of penehyclidine is 14.945min, indicating that the solv...
Embodiment 3
[0099] With reference to the method of Example 1, change the kind of dissolved reference substance acid solution, the results are shown in Table 6:
[0100] Table 6 The results of the investigation of the acid type and concentration of the dissolved reference substance
[0101]
[0102] The results showed that the impurities Ⅱ and Ⅲ were well separated by treating the reference substance with various dilute acids, and satisfactory results could be obtained. The product is the hydrochloride salt, so dilute hydrochloric acid is preferred.
[0103]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More