Epitope vaccine for resisting A/B subgroup avian leucosis virus infection and preparation method and application of epitope vaccine
A technology of avian leukosis virus and epitope vaccine, which is applied in the field of immunology, can solve the problems of virulence recovery, long purification cycle, and unstable immune effect, and achieve the effects of strong specificity, avoiding immune failure, and efficient and safe vaccine protection
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] The screening synthesis of embodiment 1ALV-A / B multi-epitope gene
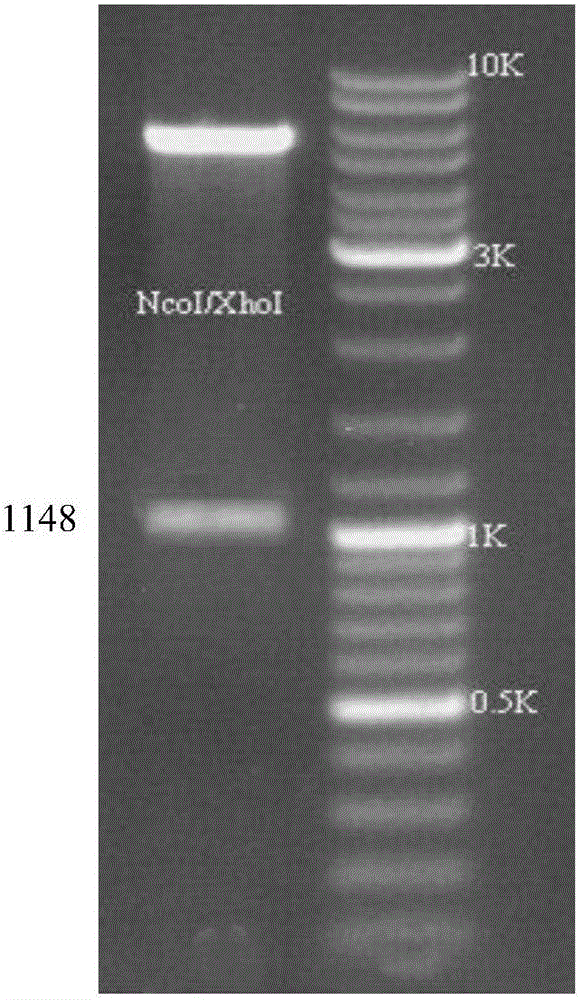
[0042] Search the ALV-A / B envelope gene sequence published on NCBI, and use bioinformatics methods to analyze and obtain 22 segments of the envelope antigen epitope, screen out the envelope epitope gene sequence representing the epidemic strains in China in recent years, and use the code Glycine and serine codons were concatenated to obtain a recombinant envelope gene sequence of 1148 bp, the gene sequence of which is shown in SEQ ID NO.1. The 5' end of the gene sequence contains an NcoI restriction site, and the 3' end contains an XhoI restriction site.
[0043] The above gene sequence was synthesized by Shanghai Sangon Bioengineering Co., Ltd., and the synthetic product cENV was further connected to the pUC57 vector by conventional techniques.
[0044] Synthetic product sequencing, its gene sequence is shown in SEQ ID NO.1;
[0045] Digest pET30a and the above-mentioned pUC-57 cloning vector linked ...
Embodiment 2
[0049] Example 2 Expression and purification of recombinant envelope protein
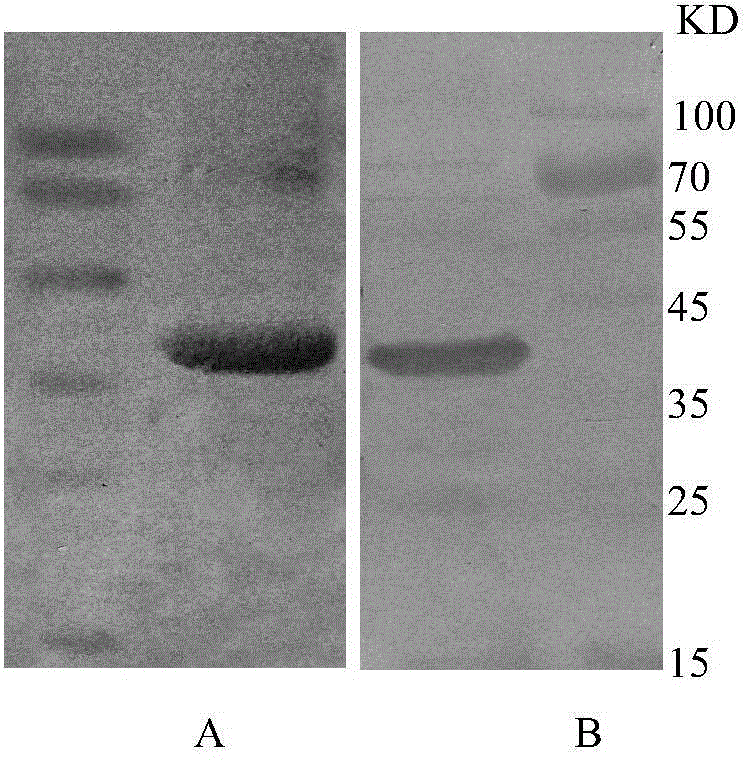
[0050] The positive BL21 bacteria containing the recombinant plasmid after sequencing were inoculated in LB liquid medium containing kanamycin (final concentration of 10 μg / ml) (commercially available conventional medium containing 1% (w / v) Tryptone, 0.5 %(w / v) Yeast Extract, 1% (w / v) NaCl, 0.1mg / ml Kanamycin), shake at 37°C (200rpm), culture to OD600=1.0, add IPTG with a concentration of 500mMol to the whole medium IPTG The final concentration was 1mmol / L, and the culture was continued at 37°C for 4h. At the same time, BL21 bacteria transformed with uninduced recombinant plasmids were set as a control.
[0051] SDS-PAGE analysis shows that the detection steps are as follows: collect and cultivate the engineering bacteria liquid, centrifuge at 5000rpm for 5min, discard the supernatant, and resuspend the precipitate with standard PBS to obtain a resuspension; add 3ml of standard PBS to 100ml of initi...
Embodiment 3
[0074] Activity detection of embodiment 3 recombinant protein
[0075] The above purified His-cENV protein was used to coat the ELISA plate, and the biological activity of the protein was detected by enzyme-linked immunosorbent assay. The chicken serum tested positive by the His-tag antibody and the IDEXX detection kit was used as the positive control, and the healthy SPF chicken serum was used as the negative control. HRP-labeled goat anti-mouse IgG and goat anti-chicken IgG enzyme-labeled secondary antibodies were used according to the instructions. The results showed that the ratio of the OD value of the positive control to the negative control was greater than 2.1, indicating that the protein had good biological activity and antigenicity.
[0076] The specific steps of ELISA are as follows:
[0077] 1) Coating: Take the purified recombinant protein, dilute it to the optimal concentration with physiological saline, and coat the reaction plate, 100 μl / well, overnight at 4°C...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More