Pravastatin sodium pharmaceutical co-crystal and preparation method and application thereof
A technology of pravastatin sodium and drugs, which is applied in the field of pravastatin sodium drug co-crystal and its preparation, can solve problems such as inaccurate prediction, and achieve the effects of promoting kinetic advantages, improving mobility, and using less
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0043] Put an equimolar amount of pravastatin sodium and the following CCF molecules in an agate mortar, add methanol or water to it, and then grind, add methanol or water continuously during the grinding process, grind for 30 minutes, take it out, and place Dry in a vacuum oven at 60°C to obtain the pravastatin sodium drug co-crystal.
[0044] CCF molecules: amino acids: histidine, lysine, arginine, threonine, isoleucine, serine, methionine, valine, phenylalanine, aspartic acid; ureas: thiourea, Urea; adamantane derivatives: 1-adamantanamine hydrochloride, 1-adamantanecarboxylic acid; other small molecules: saccharin, malic acid.
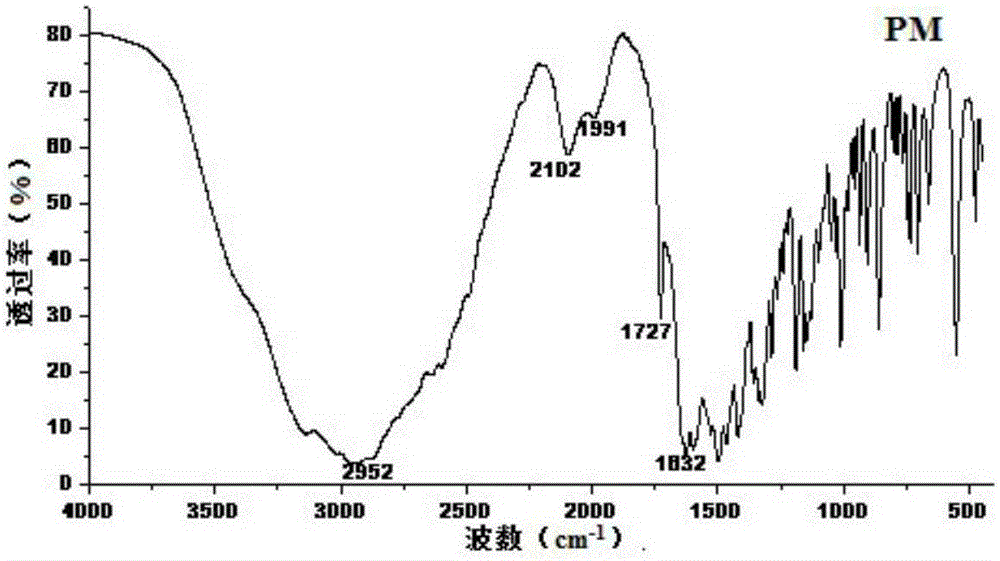
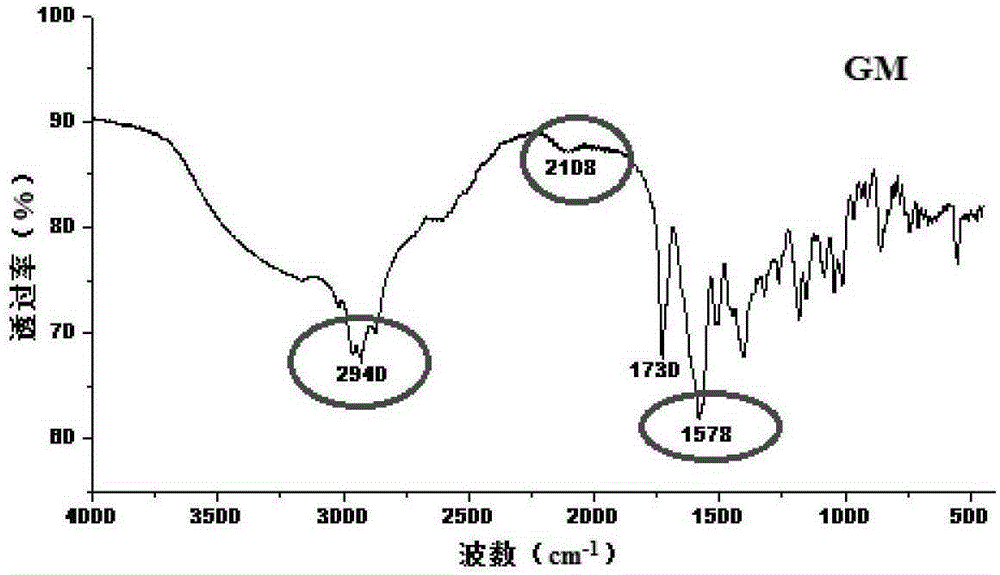
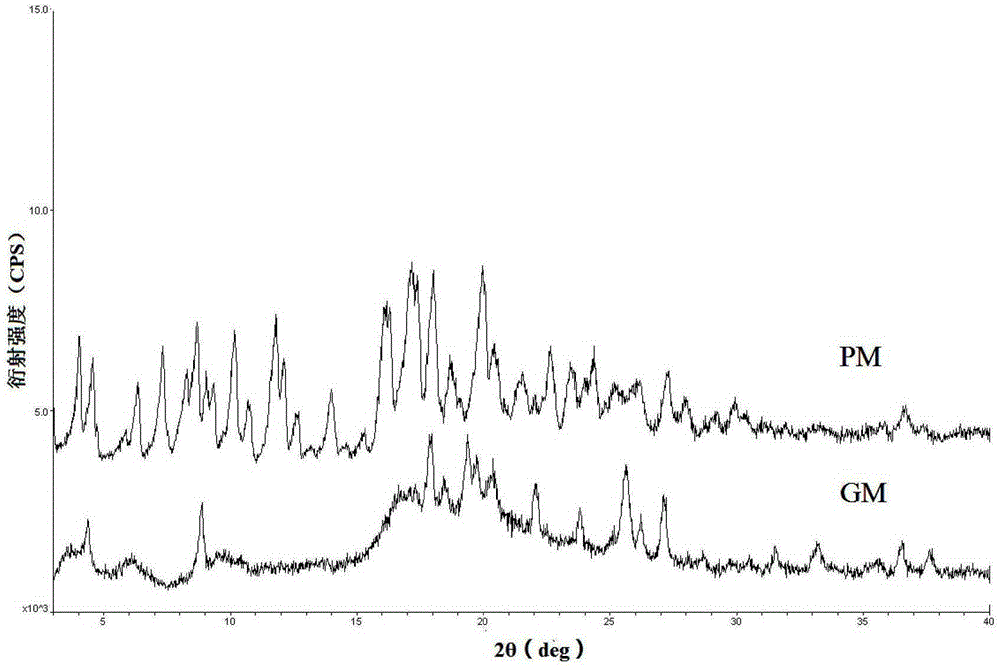
[0045] In order to confirm the formation of the co-crystal compound, the co-crystal mixture (GM) and the physical mixture (PM, obtained by mixing equimolar amounts of pravastatin sodium and CCF molecules) were compared by Fourier infrared spectroscopy (FT-IR). Investigate the stretching vibration frequency corresponding to -OH, -NH, -C=O at 2500~3...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More