Luteinizing hormone (LH) quantitative determination kit, and making method and detection method thereof
A technology for the quantitative determination of luteinizing hormone, which is applied in the direction of chemiluminescence/bioluminescence, and analysis by making materials undergo chemical reactions, can solve the problems of unfavorable high-throughput automatic detection, poor specificity, and low sensitivity. Achieve the effect of optimizing the chemiluminescence enhancement system, small variation and high sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0054] The configuration of various buffers is as follows:
[0055] 1. Tris salt pH8.0 buffer
[0056] Tris: 12.12g, sodium chloride 5.82g, add to 1L of purified water, stir well until completely dissolved, adjust the final pH to 7.5 with hydrochloric acid.
[0057] 2. Preparation of calibrator buffer
[0058] Add 0.01g of tetracycline and 0.1g of neomycin sulfate to 1L of healthy male serum, fully dissolve and process through 0.22μm filter membrane to obtain.
[0059] 3. Anti-reagent buffer
[0060] Tris: 12.12mg~60.57mg, tetracycline: 0.01g~0.05g, sheep serum: 1g~5g, newborn bovine serum 3g~10g, horse serum 1g~5g, add 1L purified water, stir well until completely dissolved;
[0061] 4. Magnetic particle buffer
[0062] Tris: 12.12mg, sodium chloride 5.82mg, methyl cellulose ether 50g, add to 1L of purified water, stir well until completely dissolved.
[0063] 5. Luminescence substrate buffer
[0064] Tris12.12g~121.14g, sodium chloride 5.82g, lucigenin 0.03g, add to 1L...
Embodiment 2
[0067] Example 2: Preparation of the Quantitative Assay Kit for Luteinizing Hormone LH
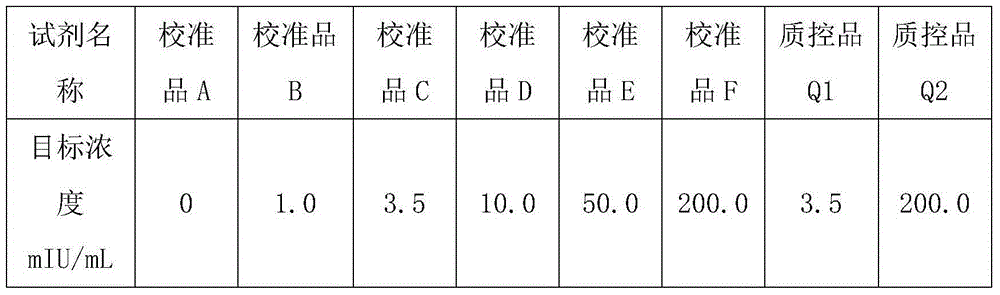
[0068] 1. Preparation of calibrators and quality controls
[0069] First: Dissolve luteinizing hormone LH with standard buffer solution, and prepare calibration products and quality control products with target concentrations as shown in Table 1; the luteinizing hormone LH used in this example is purchased from Fitzgerald, a well-known manufacturer at home and abroad .
[0070] Table 1: Preparation of calibrators and controls
[0071]
[0072] 2. The preparation method of the anti-reagent is as follows:
[0073] (1) Fluorescein isothiocyanate is coupled with luteinizing hormone LH antibody to obtain fluorescein isothiocyanate-labeled luteinizing hormone LH-coated antibody:
[0074] First, use the anti-reagent buffer to prepare the fluorescein isothiocyanate solution with a concentration of 2.5 mg / mL, and the mass ratio of luteinizing hormone LH to fluorescein isothiocyanate is 1:1.3 ...
Embodiment 3
[0086] Embodiment 3: Steps of detecting luteinizing hormone LH with luteinizing hormone LH quantitative assay kit
[0087] The method for detecting luteinizing hormone LH by using this luteinizing hormone LH quantitative assay kit, the method comprises steps:
[0088] (1) Take three test tubes and add 30 μL luteinizing hormone LH calibrator, 30 μL luteinizing hormone LH quality control, and 30 μL sample to be tested;
[0089] (2) Add 60 μL of anti-reagent to each test tube, cover the test tube with a plastic film, shake the test tube gently for 30 s, and place it in a water bath at 37°C for 15 minutes;
[0090] (3) Add 30 μL of magnetic particle reagent to each test tube, cover the test tube with plastic film, shake the test tube gently for 30 seconds, and place it in a water bath at 37°C for 5 minutes;
[0091] (4) Precipitate the test tube on the magnetic separator for 2 minutes, slowly invert the test tube and the magnetic separator, pour out the supernatant, put the inver...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More