High efficiency biosynthesis method of caffeic acid with catechol as substrate
A biosynthesis and catechol technology, applied in the direction of microorganism-based methods, biochemical equipment and methods, microorganisms, etc., can solve the problems of low conversion rate of caffeic acid yield, unsatisfactory effect, and no significant increase in yield, etc., to achieve Inexpensive and easily mass-producible effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0033] The construction method of embodiment 1 recombinant escherichia coli
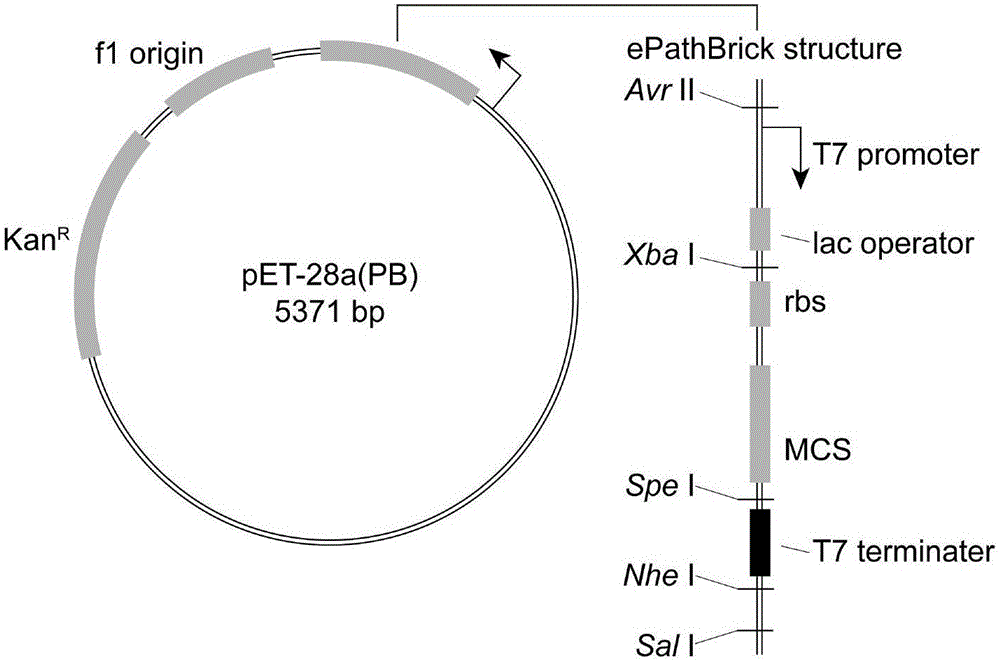
[0034]The genes CfTPL (SEQ ID NO.3) and TcXAL (SEQ ID NO.4) were optimized and synthesized by GenScript Biotechnology Co., Ltd., and cloned into pUC57-Simple. The recombinant plasmids were named pUC57-CfTPL and pUC57-TcXAL, respectively. pET28a(PB) is an ePathBrick expression vector constructed on the basis of pET-28a(+). The vector contains homologous enzymes Avr II, XbaI, Spe I, and Nhe I and downstream Sal I. Different co-expression vectors can be constructed through the ePathBrick strategy. expression structure. The structure of pET28a(PB) is as follows figure 2 , the DNA sequence is SEQ ID NO.5.
[0035] The recombinant vectors pUC57-CfTPL, pUC57-TcXAL and the expression vector pET28a(PB) were digested with restriction endonuclease Bam HI / HindIII respectively, and the cut products were separated by agarose gel electrophoresis, and the target genes CfTPL (1371bp), TcXAL (2070bp) and expressio...
Embodiment 2
[0042] Example 2 Analysis of recombinant Escherichia coli strCL-1 catalyzing the synthesis ability of levodopa
[0043] In order to verify the ability of the recombinant E. coli strCL-1 in Table 1 to catalyze the synthesis of levodopa from catechol, according to the transformation method in Example 1, the E. coli BL21 (DE3 ) as a blank control, with PBS (50mM, pH 7.0) as the reaction medium, reacted at 37°C and 220rpm, and took samples at specific time points to measure the synthesis of levodopa.
[0044] The results showed that levodopa was synthesized in the reaction system catalyzed by E.coli strCL-1, but levodopa was not detected in the blank control. This result verified the ability of CfTPL to catalyze the synthesis of levodopa from catechol, and the synthesis process could not proceed spontaneously in the absence of tyrosine benzene lyase. The chromatogram and mass spectrum of levodopa are as follows Figure 4 , wherein, A is the chromatogram of levodopa, B is the pri...
Embodiment 3
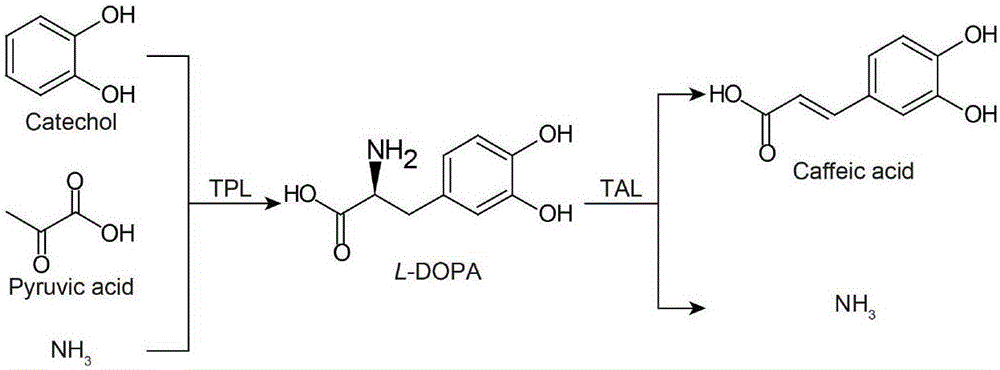
[0045] Example 3 Recombinant Escherichia coli catalyzes the ability to synthesize caffeic acid from catechol
[0046] In order to verify the ability of the recombinant Escherichia coli listed in Table 1 to transform catechol, pyruvate and ammonium chloride into caffeic acid, the collected bacteria were washed with 25 mL of PBS, centrifuged and resuspended in an equal volume of PBS (50 mM, pH 7.0 ), the cell concentration is OD 600 =18±1. Simultaneously add 0.65M NH 4 Cl, 50 mM sodium pyruvate and 50 mM catechol were used as substrates for the reaction, and the reaction was carried out on a constant temperature shaker at 37° C. and 220 rpm. Escherichia coli BL21(DE3) containing empty plasmid pET28a(PB) was used as blank control. Samples were taken at different time points for analysis to determine caffeic acid synthesis. Take the maximum production of caffeic acid in 0-10 hours as the corresponding maximum production of the strain.
[0047] The results showed that, except ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More