Immunochromatography time resolution fluorescence kit for synchronously detecting mixed pollution of five types of fungi toxins fungaltoxin of aflatoxin and the like and application
A time-resolved fluorescence and aflatoxin technology, applied in fluorescence/phosphorescence, analytical materials, measurement devices, etc., can solve problems such as high requirements, poor reproducibility, and complicated sample pretreatment process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
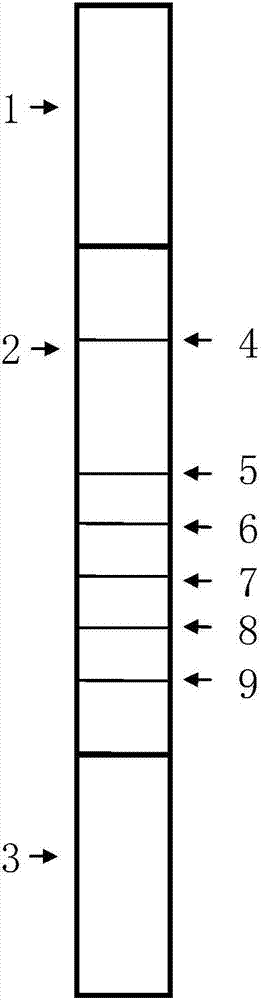
Image
Examples
Embodiment 1
[0040] The acquisition of embodiment 1 aflatoxin B1 monoclonal antibody
[0041] The aflatoxin universal monoclonal antibody is secreted and produced by the hybridoma cell line 3G1 with the deposit number CCTCC NO.C201014. Specifically, it is pre-prepared according to the method reported in the patent with the authorization number ZL201210117614.9. The preparation method is: the obtained hybrid The tumor cell line 3G1 was injected into BALB / c mice pre-treated with Freund's incomplete adjuvant, the ascites of the mice was collected, purified and processed to obtain aflatoxin B1 monoclonal antibody. Among them, the purification method is the octanoic acid-ammonium sulfate method, and the specific operation is: filter the mouse ascites with double-layer filter paper, centrifuge the filtered ascites at 4°C, 12000r / min for more than 15min, absorb the supernatant, and dilute the supernatant with 4 times the volume Mix acetate buffer solution, slowly add n-octanoic acid while stirrin...
Embodiment 2
[0043] Example 2 Obtaining of Ochratoxin A Monoclonal Antibody
[0044] Ochratoxin A monoclonal antibody is secreted and produced by the hybridoma cell line 1H2 with the deposit number CCTCC NO.C201329. Specifically, it is prepared in advance according to the method reported in the patent application number 201310115921.8. The preparation method is: hybridoma cell line 1H2 Inject into the abdomen of BALB / c mice treated with Freund's incomplete adjuvant in advance, collect the ascites of the mice, and purify to obtain ochratoxin A monoclonal antibody. The purification method is caprylic acid-ammonium sulfate method, and the specific steps are: filter mouse ascites with double-layer filter paper, centrifuge at 12,000 r / min for more than 15 minutes at 4°C, absorb the supernatant, and mix the obtained ascites supernatant with 4 times the volume of vinegar Mix salt buffer solution, add n-octanoic acid slowly under stirring, the volume of n-octanoic acid required per ml of ascites i...
Embodiment 3
[0046] Example 3 Obtaining of Zearalenone Monoclonal Antibody
[0047]The zearalenone monoclonal antibody is secreted and produced by the hybridoma cell line 2D3 with the deposit number CCTCC NO.C201328. Specifically, it is prepared in advance according to the method reported in the patent application number 201310115825.3. The preparation method is as follows: the hybridoma cell line 2D3 was injected into the abdomen of BALB / c mice pre-treated with Freund's incomplete adjuvant, the ascites of the mice was collected, and purified to obtain the zearalenone monoclonal antibody; the purification method is caprylic acid-ammonium sulfate The specific steps are as follows: filter mouse ascites with double-layer filter paper, centrifuge at 12,000 r / min for more than 15 minutes at 4°C, absorb the supernatant, mix the obtained ascites supernatant with 4 times the volume of acetate buffer, and slowly add n-octanoic acid, the volume of n-octanoic acid required per milliliter of ascites i...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Sensitivity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More