Sulfur-bridged quinazoline-based tetradentate platinum complex phosphorescent material and preparation method thereof
A technology of quinazolines and platinum complexes, applied in the direction of luminescent materials, platinum group organic compounds, platinum organic compounds, etc., can solve the problems of low quantum efficiency, improve luminous efficiency, reduce self-quenching phenomenon, improve Solubility effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] The preparation of embodiment 1,2-(3-bromophenyl)-4-diphenylaminoquinazoline qba
[0030] Weigh 0.35g (1mmol) of 2-(3-bromophenyl)-4-chloroquinazoline, 0.08g (2mmol) of 60% sodium hydride, and 0.25g (1.5mmol) of diphenylamine in a two-necked bottle, and add Solvent dimethyl sulfoxide, react at 30°C for 10h. After the reaction was completed, the reaction solution was poured into water, a large amount of white precipitate was produced, suction filtered, column chromatography, separated by petroleum ether:ethyl acetate=20:1 developer chromatography, and 0.3g (0.67mmol) of white solid qba was obtained , the yield was 67%. 1H NMR (400MHz, CDCl3) δ7.93 (m, 8H), 7.46 (dd, J = 5.3, 3.2Hz, 4H), 7.12 (dd, J = 5.4, 3.1Hz, 4H), 5.84 (S, 2H) .
[0031]
Embodiment 2
[0032] Embodiment 2, the quinazoline S (qba) of ligand sulfur atom bridging 2 preparation of
[0033] Weigh 2-(3-bromophenyl)-4-diphenylaminoquinazoline qba 0.225g (0.5mmol), potassium phosphate 0.064g (0.3mmol), potassium thioacetate 0.029g (0.25mmol), bis( Dibenzylideneacetone)palladium 0.023g (0.025mmol), 1,1-bis(diphenylphosphino)ferrocene 0.02g (0.035mmol), add toluene 3ml, acetone 1.5ml mixed solvent under nitrogen protection, 110℃ Reaction 7h. After the reaction was completed, it was cooled to room temperature, extracted with dichloromethane, dried by adding anhydrous sodium sulfate, and separated by column chromatography with petroleum ether: ethyl acetate = 10:1 developer chromatography to obtain 0.1 g of white solid S(qba) 2 , the yield was 52%. 1H NMR (400MHz, CDCl3) δ = 8.38 (S, 2H), 8.11 (d, J = 7.6, 2H), 7.96 (d, J = 7.9, 2H), 7.65 (t, J = 7.7, 2H), 7.39 –7.28(m,17H),7.17(t,J=11.1,16H).
[0034]
Embodiment 3
[0035] Embodiment 3, complex S (qba) 2 Preparation of Pt
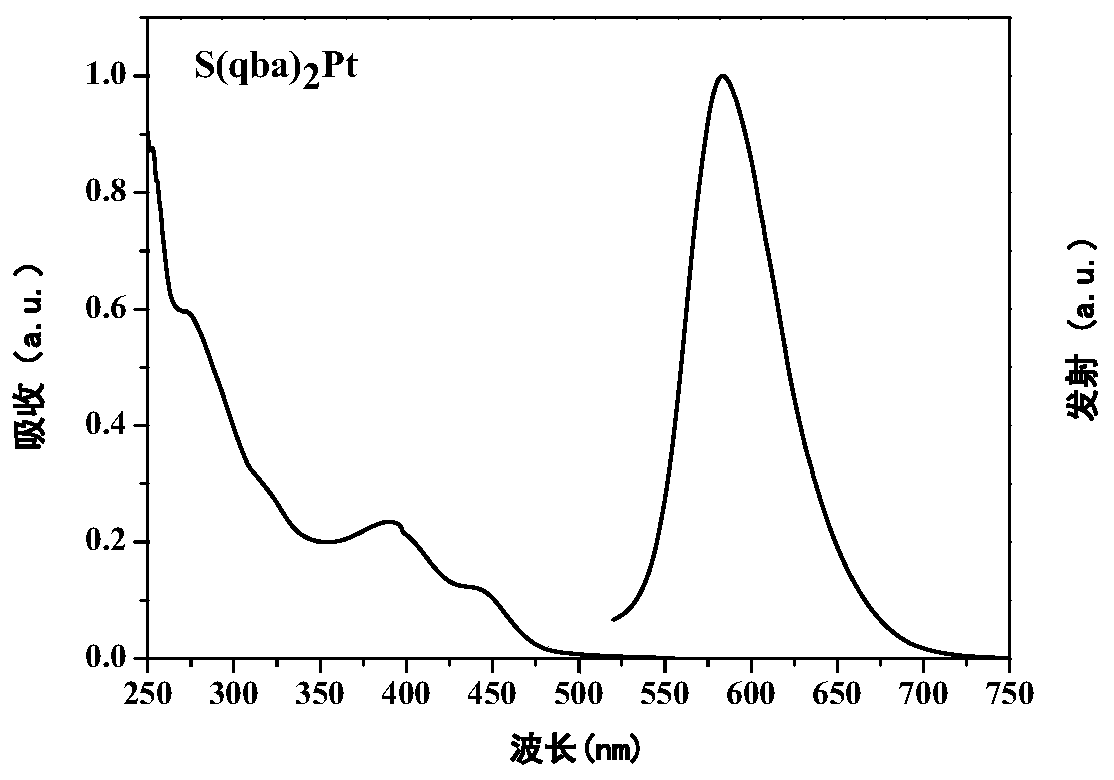
[0036] Weigh quinazoline S(qba) bridged by sulfur atom 2 0.077g (0.1mmol), 0.05g (0.1mmol) potassium tetrachloroplatinate, 4mg tetrabutylammonium bromide, 5ml acetic acid was added under nitrogen protection, stirred at room temperature for 12 hours, heated to 120°C for 70h. After the reaction was completed, cool to room temperature, add 15ml of deionized water, a precipitate precipitated out, filter with suction, dry, column chromatography, and use dichloromethane: ethyl acetate = 5:1 developer chromatography to obtain 0.032g orange-red solid S(qba) 2 Pt, 25% yield. 1H NMR (400MHz, CDCl3) δ=8.14–8.07(m,2H),7.47(t,J=7.7,4H),7.42–7.28(m,9H),7.09(t,J=7.6,1H),6.99 –6.93(m,2H).MALDI-TOF-MS(m / z):calcd for[M]+C 52 h 34 N 6 SPt: 969.37, found: 969.594.
[0037]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More