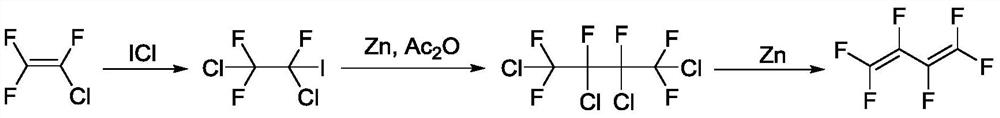
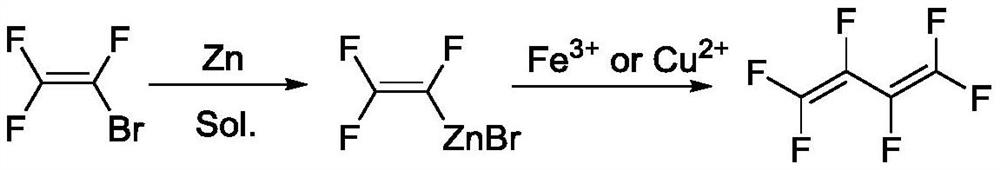
A kind of preparation method of hexafluoro-1,3-butadiene and its intermediate
A technology of butadiene and hexafluorobutane, which is applied in the field of synthesizing hexafluoro-1,3-butadiene and its intermediates by telomerization, can solve the problems of high equipment requirements and high process safety risks, and achieve improved The effect of selectivity, simple process and mild reaction conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0043] This embodiment provides a preparation method of hexafluoro-1,3-butadiene, the preparation method includes intermediate preparation steps and hexafluoro-1,3-butadiene preparation steps, specifically as follows:
[0044] A1. Preparation of 1,4-dibromo-2,3-dichloro-1,1,2,3,4,4-hexafluorobutane
[0045] Add 150g of acetonitrile, 69.1g (0.25mol) of 1,2-dibromo-1-chloro-1,2,2-trifluoroethane, 69.1g (0.25mol) of diphenylmethane peroxide, Acyl 1.8g (7.5mmol), with high-purity N 2 Purge for 10 minutes, then pour 34.8g (0.30mol) of chlorotrifluoroethylene from the steel cylinder into the reactor, heat up to 80°C under mechanical stirring (300-500r / min), the pressure of the reactor rises to about 0.5MPa, and keep warm for 12 hours to end the reaction.
[0046] The reaction solution was analyzed by GC-MS and calculated: the conversion rate of raw material 1,2-dibromo-1-chloro-1,2,2-trifluoroethane was 98.75%, and the conversion rate of intermediate 1,4-dibromo- The selectivity ...
Embodiment 2
[0051] The operation of this embodiment is the same as that of Embodiment 1, the difference is only in:
[0052] During the preparation of the intermediate, the type of initiator was changed, and the dibenzoyl peroxide in Example 1 was replaced with 1.1 g (7.5 mmol) of di-tert-butyl peroxide. The reaction results are shown in Table 1.
[0053] During the preparation of hexafluoro-1,3-butadiene, the type of catalyst was changed, and 2.5 g of zinc iodide was used instead of elemental iodine in Example 1 to obtain 89.20 g of the product. The results are shown in Table 3.
Embodiment 3
[0055] The operation of this embodiment is the same as that of Embodiment 1, the difference is only in:
[0056] During the preparation of the intermediate, the type of initiator was changed, and the dibenzoyl peroxide in Example 1 was replaced with 0.68 g (7.5 mmol) tert-butyl hydroperoxide; the reaction results are shown in Table 1.
[0057] During the preparation of hexafluoro-1,3-butadiene, the type of catalyst was changed, and 1.5 g of 1,2-dibromoethane was used instead of elemental iodine in Example 1 to obtain 95.0 g of the product. The results are shown in Table 3.
PUM
| Property | Measurement | Unit |
|---|---|---|
| boiling point | aaaaa | aaaaa |
| freezing point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More