Use of R-enantiomer of N-propargyl-1-aminoindan, salts, compositions and uses thereof
a technology of npropargyl-1 and renantiomer, which is applied in the field of renantiomer of npropargyl-1aminoindan, salts, compositions, etc., can solve the problems of l-dopa and an inhibitor of both mao-a and mao-b, onset of disturbances in voluntary muscle control, and clinical response decline, so as to prevent nerve damage and prevent nerve damage
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
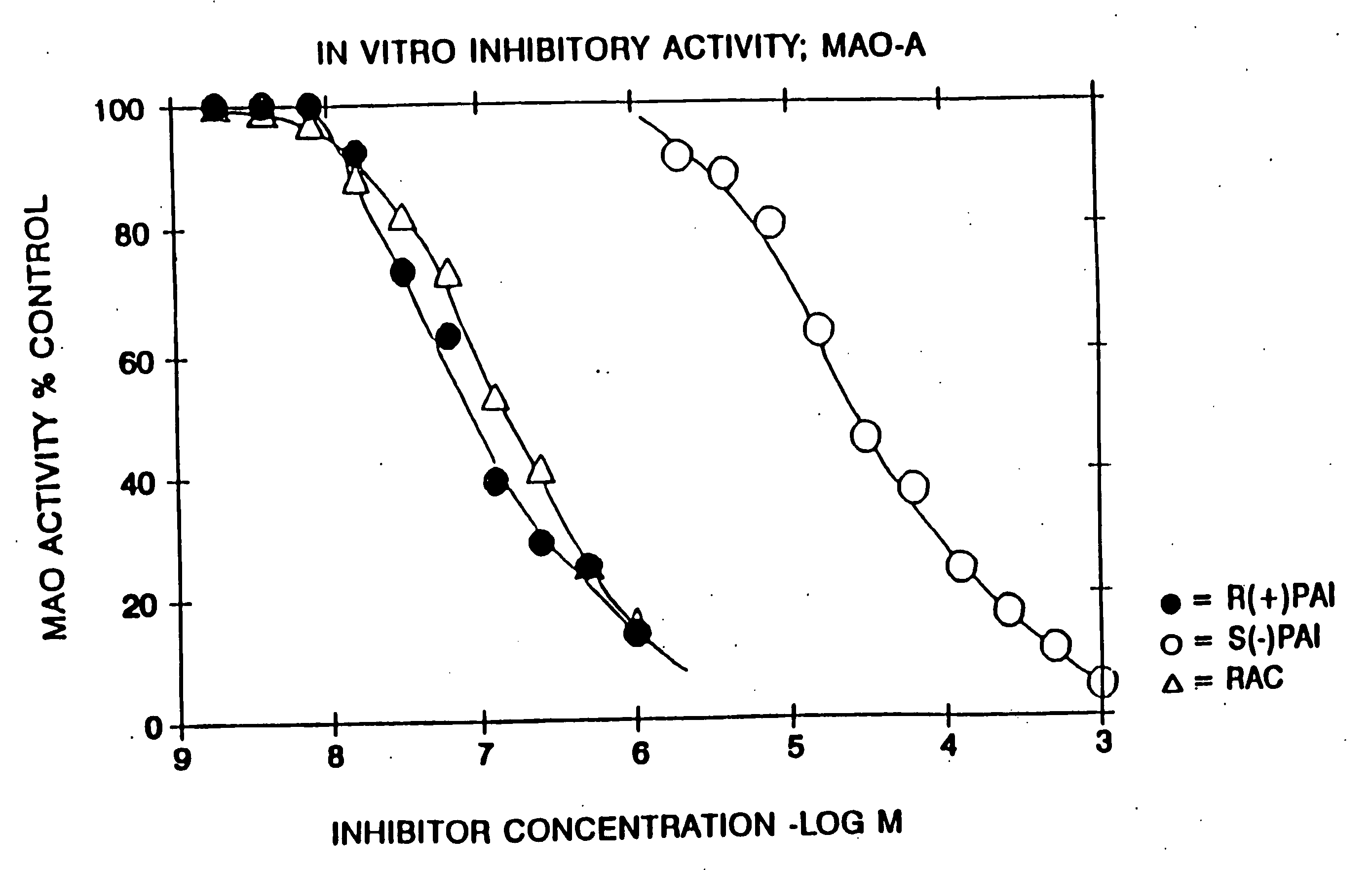
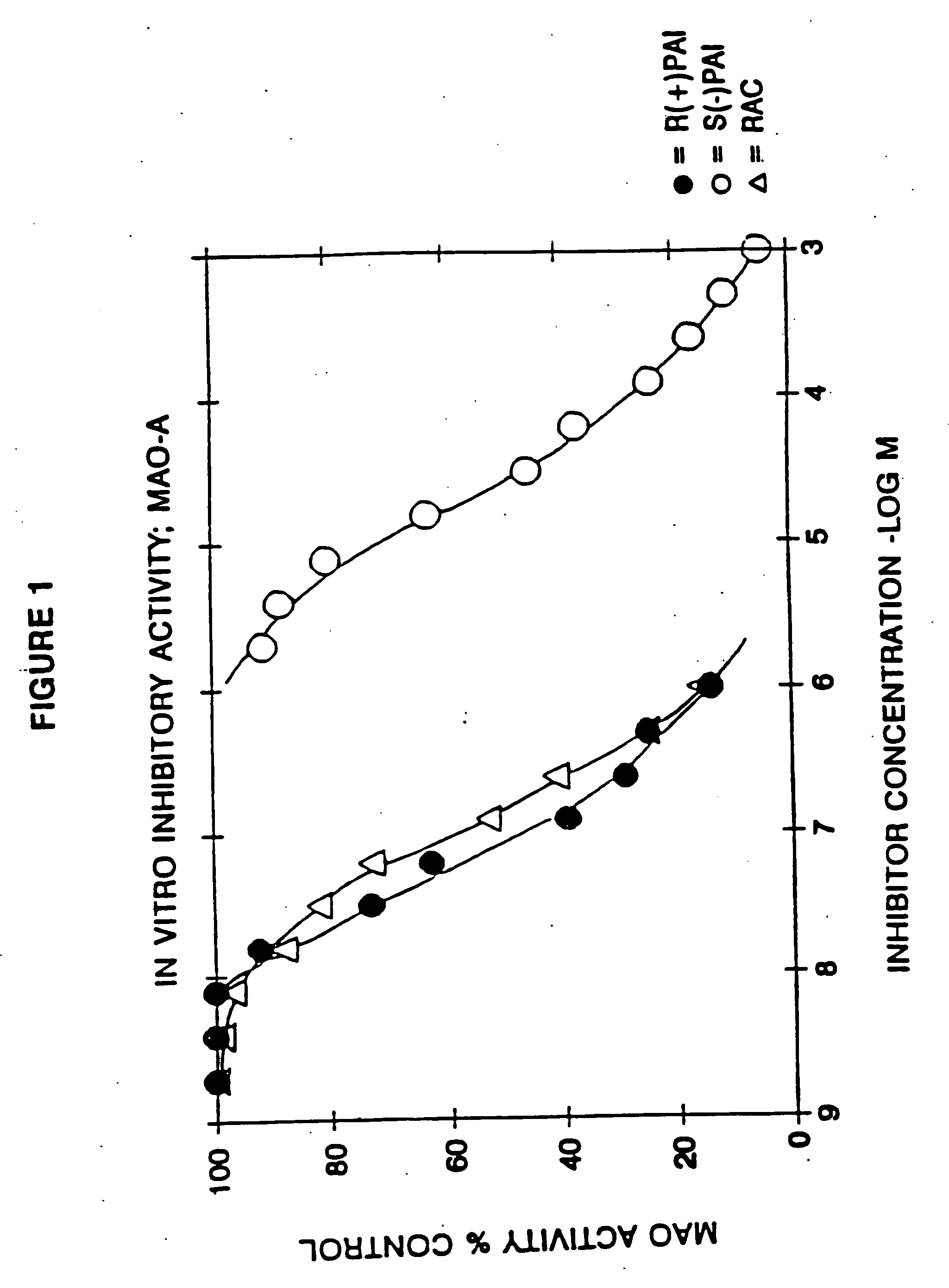
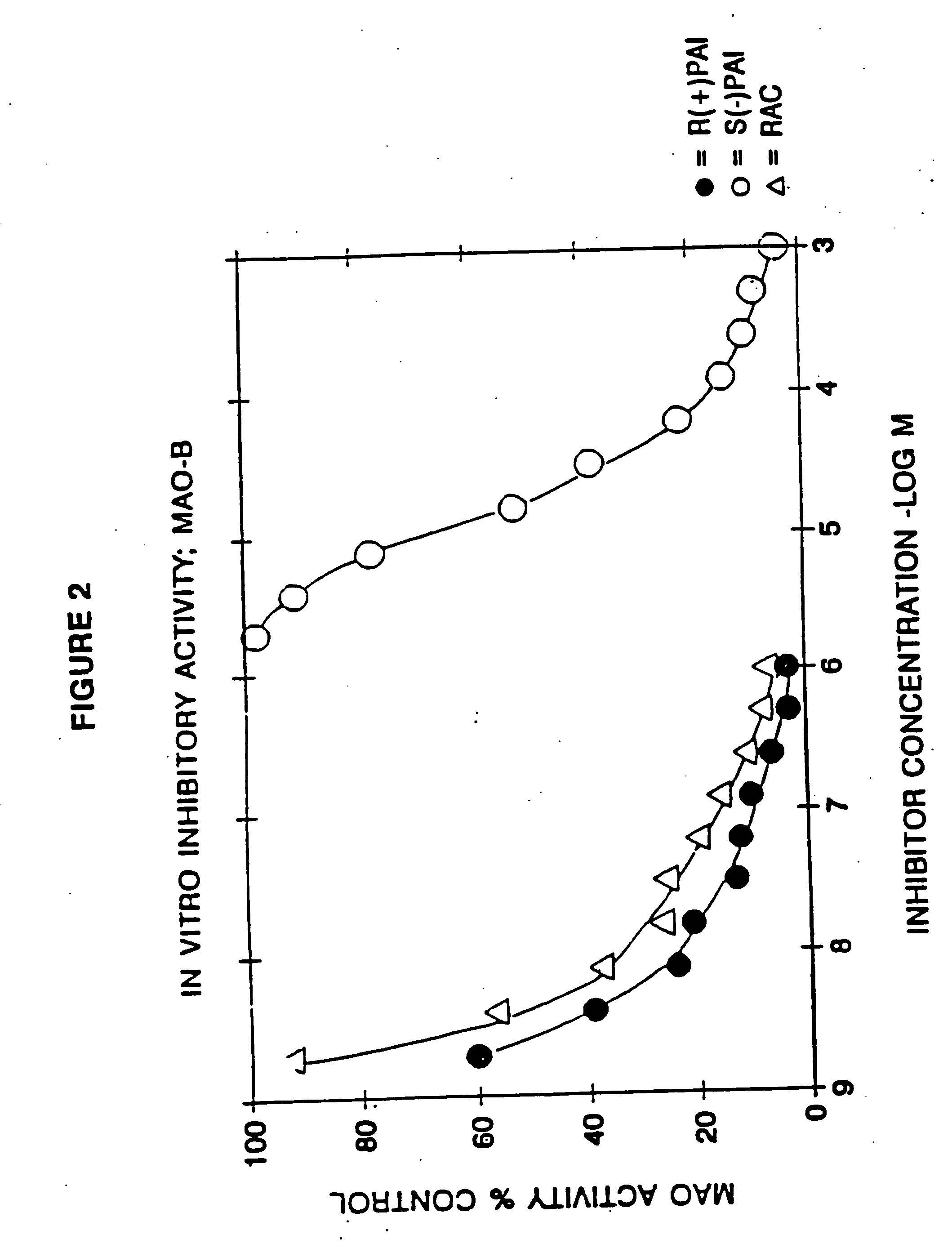
example 1
Racemic N-propargyl-1-aminoindan hydrochloride
[0147] 10.0 g of racemic 1-aminoindan and 10.4 g of potassium carbonate were added to 75 ml of acetonitrile. T*ee resulting suspension was heated to 60° C. and 4.5 g of propargyl chloride was added dropwise.
[0148] The mixture was stirred at 60° C. for 16 hours, whereafter most of the volatiles were removed by distillation in vacuo. The residue was partitioned between 10% aqueous sodium hydroxide and methylene chloride.
[0149] The organic phase was dried and the solvent removed by distillation. The residue was flash chromatographed on silica gel, eluting with 40% ethyl acetate / 60% hexane. The fractions containing the title compound as a free base were combined and the eluant replaced by ether. The ethereal solution was treated with gaseous HCl, the precipitate formed was isolated by suction filtration and recrystallized from isopropanol to yield 7.3 g of the title compound, m.p. 182-4° C.
[0150] Chromatographic and spectroscopic data we...
example 2
S-(−)-N-Propargyl-1-aminoindan hydrochloride
[0151] The title compound in free base form was isolated by resolving the racemic mixture of the free base of Example 1 on a Chiracel OJ (cellulose tris [p-methylbenzoate]) preparative HPLC column eluting with 10% isopropanol / 90% hexane and collecting the first eluted major peak. The resulting oil was converted to the title compound (hydrochloride) by treatment of a 10% diethyl ether solution of the oil with gaseous HCl, and the resulting precipitate was collected by suction filtration. [a]D−29.2° (1%, ethanol), m.p. 182-184° C. Other chromatographic and spectroscopic properties were identical with the hydrochloride salt of Example 1.
example 3
R-(+)-N-Propargyl-1-aminoindan hydrochloride
[0152] The title compound was prepared as in Example 2 above, except that the second eluted peak from the preparative HPLC was collected: [a]D+29.1° (0.8%, ethanol), m.p. 179-181° C. Other chromatographic and spectroscopic properties were identical with the hydrochloride salt of Example 1.
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More