Peptides Having Activities of Insulin Like Growth Factor-1 and their Uses
a technology of growth factor and activity, applied in the field of peptides having insulin like growth factor1, can solve the problems of complex purification, time-consuming and cost-intensive refolding process, and the lik
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
preparation example 1
Tyr-Phe-Asn-Lys-Ala-Ala-Gly-Tyr-Gly-Ser-Ser-Ser-Arg-Arg (SEQ ID NO:1)
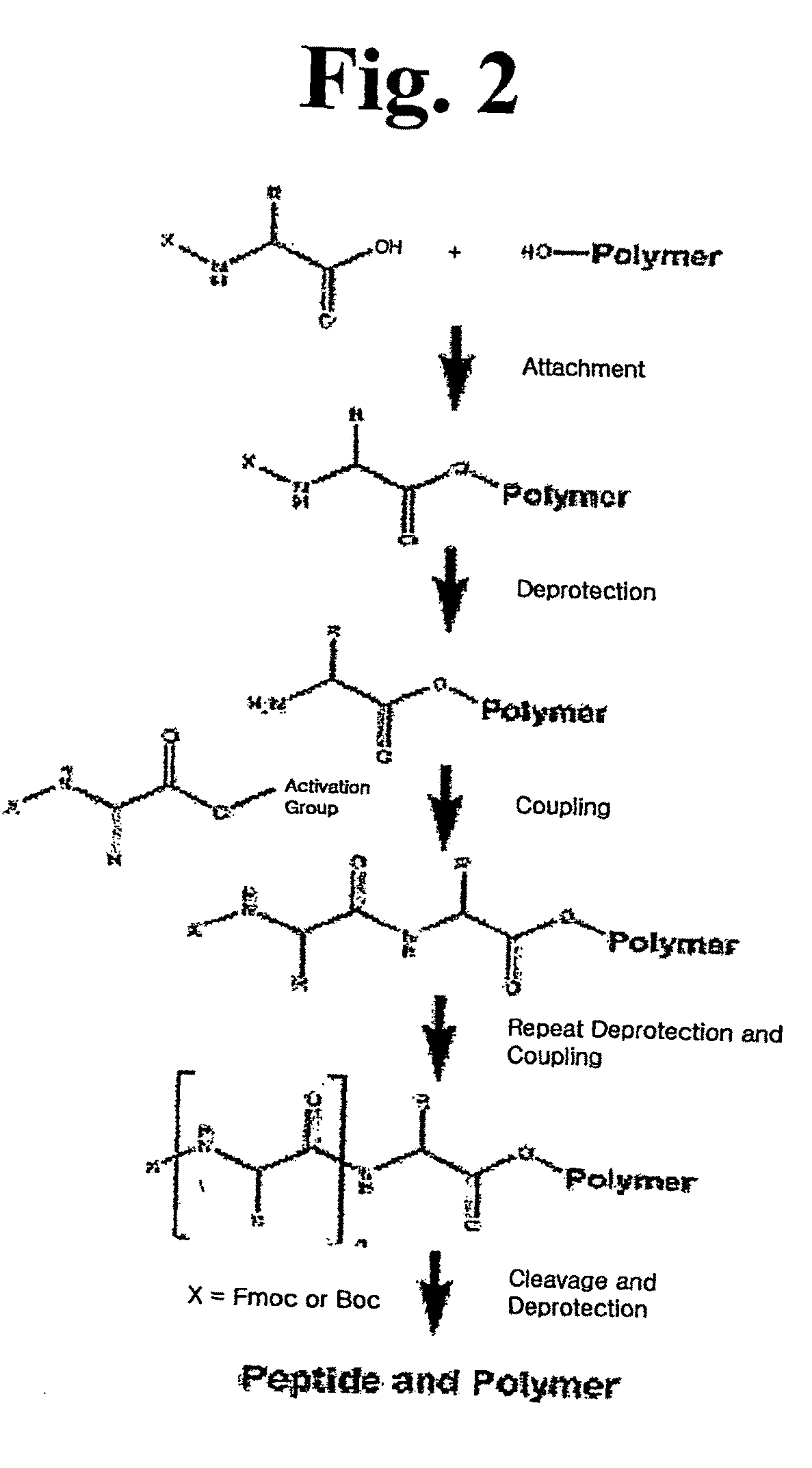
[0079]To 700 mg of chloro trityl chloride resin (CTL resin, Nova Biochem Cat No. 01-64-0021) introduced into a reactor were added 10 ml of methylene chloride (MC) and agitated for 3 min. After removing solution, 10 ml of dimethylformamide (DMF) were added to the resultant and then agitation was carried out for 3 min, after which the solvent was removed. 10 ml of dichloromethane solution were added to the reactor and 200 mmole of Fmoc-Arg(pbf)-OH and 400 mmole of diisopropyl ethylamine (DIEA) were then added to the reactor, after which the mixture was dissolved by agitation and reaction was then undertaken with agitating for 1 hr. After reaction, the resultant was washed and reacted for 10 min in methanol and DIEA (2:1) dissolved in DCM, followed by washing with excess DCM / DMF (1:1). Following the removal of the solvent, 10 ml of DMF were added to the reactor and agitated for 3 min. followed by removing the solvent...
preparation example 2
ptides
[0080]The peptides of SEQ ID NOs:2-5 were synthesized as processes described in Preparation Example 1. SEQ ID NO:2 (Gly-Tyr-Gly-Ser-Ser-Ser-Arg-Arg-Ala-Pro-Gln-Thr: GYGSSSRRAPQT) corresponds to amino acids 30-41 of IGF-1, SEQ ID NO:3 (Glu-Ser-Ser-Phe-Arg-Ser-Ser-Asp-Leu-Arg-Arg-Leu: ESSFRSSDLRRL) to amino acids 46-57 of IGF-1, SEQ ID NO:4 (Cys-Asp-Leu-Arg-Arg-Leu-Glu-Met-Tyr-Cys: CDLRRLEMYC) to amino acids 52-61 of IGF-1, and SEQ ID NO:5 (Arg-Arg-Leu-Glu-Met-Tyr-Cys-Ala-Pro-Leu-Lys-Pro: RRLEMYCAPLKP) to amino acids 55-66 of IGF-1.
[0081]The determined molecular weights of the peptides are summarized in Table 1.
TABLE 1Analyzed values(mass analyzer)SEQ IDAmino acidAnalyzedTheoreticalNOsequencevaluesvalues1GFYFNKAAGYGSSSRR1770.61767.92GYGSSSRRAPQT1270.881266.43ESSFRSSDLRRL1457.41452.64CDLRRLEMYC1304.81303.65RRLEMYCAPLKP1479.51476.8
preparation example 3
Leu-Arg-Arg-Leu-Glu-Met-Tyr-Cys (1,10 Cyclized CDLRRLEMYC)
[0082]To increase the stability of the peptide of SEQ ID NO:4 prepared in Preparation Example 2, the cyclization was carried out using Cys residues at the C- and N-terminal. 100 mg of the peptide of SEQ ID NO:4 (Cys-Asp-Leu-Arg-Arg-Leu-Glu-Met-Tyr-Cys-OH) were dissolved in 1 L of 10% DMSO / deprotection distilled water. After fixing pH to 8.0, the peptide solution was agitated for 8 hr under air to induce oxidation. By preparative chromatography, 40 mg of cyclized Cys-Asp-Leu-Arg-Arg-Leu-Glu-Met-Tyr-Cys-OH (1,10 cyclized). The molecular weight of the final cyclized peptide was determined as 1302.8 using a mass analyzer.
PUM
| Property | Measurement | Unit |
|---|---|---|
| total volume | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More