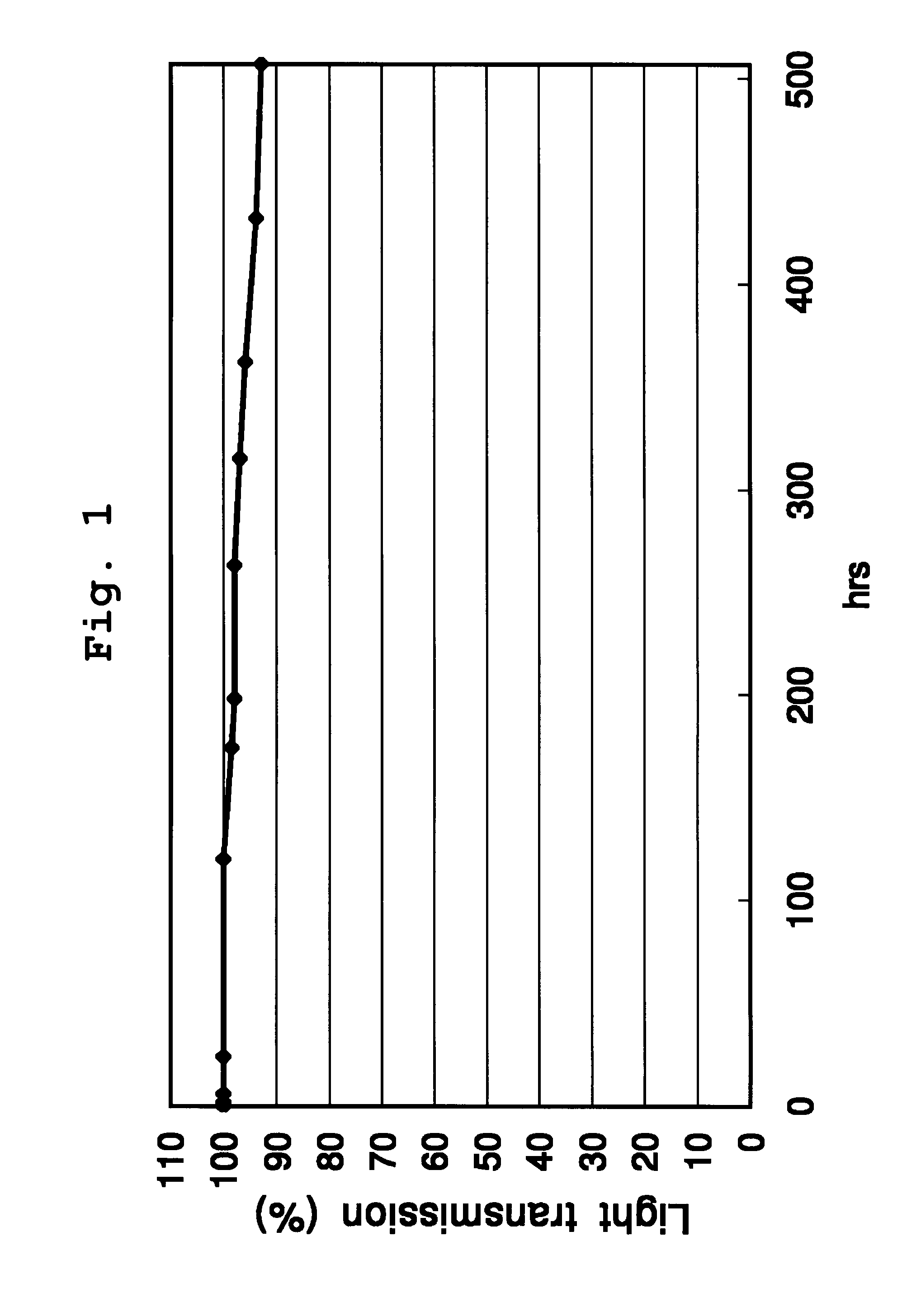
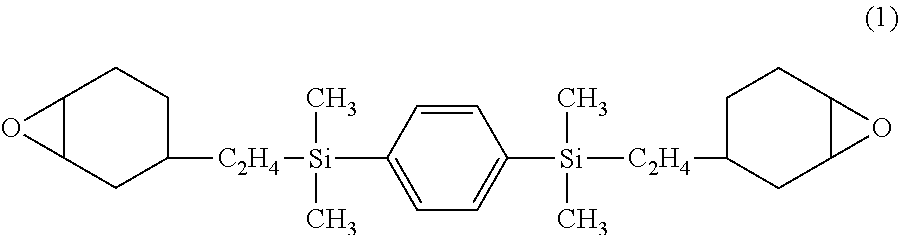
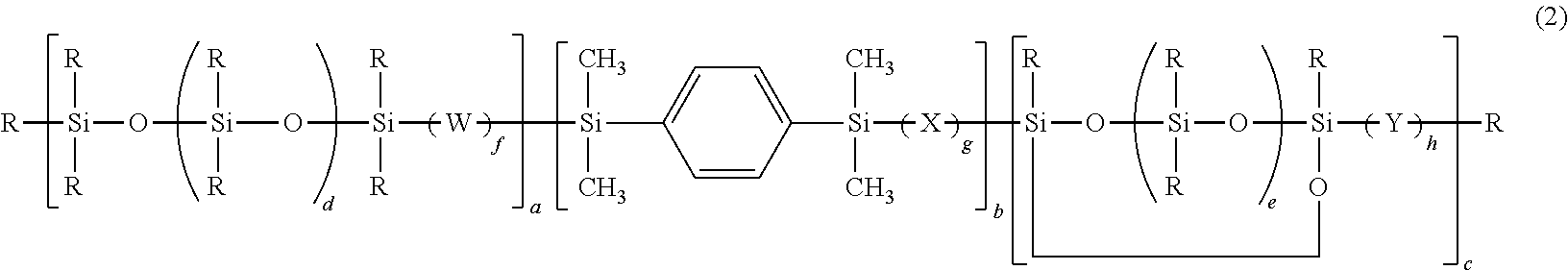
Silphenylene-containing photocurable composition, pattern formation method using same, and optical semiconductor element obtained using the method
a technology of photocurable composition and silphenylene, which is applied in the direction of photomechanical equipment, instruments, photosensitive materials, etc., can solve the problems of gas emission and discoloration of devices using a stronger light intensity, the inability of resins to undergo the inability to achieve microfabrication in the order of 10 m, etc., to achieve excellent transparency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
synthesis example 1
[0077]A flask fitted with a stirrer and a thermometer was charged with 49.7 g (0.4 mols) of 1,2-epoxy-4-vinylcyclohexane, 500 g of toluene and 0.5 g of a 2% by mass ethanol solution of chloroplatinic acid, and with the flask contents undergoing constant stirring, the flask was heated using an oil bath until the internal temperature reached 70° C. Subsequently, 38.9 g (0.2 mols) of 1,4-bis(dimethylsilyl)benzene was added dropwise to the flask over a period of 20 minutes. Following completion of the dropwise addition, the reaction mixture was stirred for 3 hours at 90° C. Following completion of the reaction, the reaction liquid was subjected to a distillation treatment under heating and reduced pressure (50° C., 1 mmHg) using a rotary evaporator until no further distillate was produced, thus yielding 85 g of a silphenylene (A-1) having both terminals modified with alicyclic epoxy groups that represents the component (A).
synthesis example 2
[0078]A flask fitted with a stirrer and a thermometer was charged with 49.7 g (0.4 mols) of 1,2-epoxy-4-vinylcyclohexane, 500 g of toluene and 0.5 g of a 2% by mass ethanol solution of chloroplatinic acid, and with the flask contents undergoing constant stirring, the flask was heated using an oil bath until the internal temperature reached 70° C. Subsequently, 65.1 g (0.1 mols) of an organosiloxane having an average structure represented by formula (7) shown below was added dropwise to the flask over a period of 20 minutes. Following completion of the dropwise addition, the reaction mixture was stirred for 3 hours at 90° C. Following completion of the reaction, the reaction liquid was subjected to a distillation treatment under heating and reduced pressure (50° C., 1 mmHg) using a rotary evaporator until no further distillate was produced, thus yielding 110 g of an epoxy-modified siloxane compound (B-1) having an average structure represented by formula (8) shown below.
synthesis example 3
[0079]A flask fitted with a stirrer and a thermometer was charged with 27.3 g (0.22 mols) of 1,2-epoxy-4-vinylcyclohexane, 16.8 g (0.09 mols) of 1,3-divinyl-1,1,3,3-tetramethylpropanedisiloxane, 500 g of toluene and 0.5 g of a 2% by mass ethanol solution of chloroplatinic acid, and with the flask contents undergoing constant stirring, the flask was heated using an oil bath until the internal temperature reached 70° C. Subsequently, 24.1 g (0.1 mols) of 1,3,5,7-tetramethylcyclotetrasiloxane was added dropwise to the flask over a period of 20 minutes. Following completion of the dropwise addition, the reaction mixture was stirred for 3 hours at 90° C. Following completion of the reaction, the reaction liquid was subjected to a distillation treatment under heating and reduced pressure (50° C., 1 mmHg) using a rotary evaporator until no further distillate was produced, thus yielding 65 g of an epoxy-modified siloxane compound (B-2) having an average structure represented by formula (9) ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Percent by mass | aaaaa | aaaaa |
| Nanoscale particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com