Thiazole Inner Salt Compounds, and Preparation Methods and Uses Thereof
a technology of thiazole inner salt and compound, which is applied in the field of pharmaceutical chemicals, can solve the problems of abnormal lipoprotein metabolism, reduced vascular elasticity, and reduced vascular compliance, and achieves stable pharmacokinetics, easy quality control, and equivalent activity.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 2
Preparation of 3-methylcarbonyloxy-4-methyl-thiazole inner salt (n=0)
[0101]10 g 3-carboxymethyl-4-methyl-thiazolium bromide white solid was dissolved in 50 mL distilled water, added then with 7.31 g 1,2-epoxypropane, stirred at room temperature for 12 h, after the end of reaction, the reaction solution was extracted with 30 mL of dichloromethane, for 3 times, dichloromethane layer was discarded; the water layer was evaporated at a reduced pressure to obtain a light yellow oily substance. A defined amount of acetone was added into the oily substance, and light yellow particles were obtained via precipitation; recrystallization was performed with ethanol-ethyl ether system (wherein the most preferable recrystallization ratio was: 1 g of yellow particles was heated and dissolved in 4.5 mL ethanol, then added with 2 mL ethyl ether), to obtain white crystal 5.15 g, yield 78%, mP=169° C.
[0102]MS: 158 [M+H]+, 315 [2M+H]+, 472 [3M+H]+; 1H-NMR (400 MHz, DMSO-d6), 2.41 (d, 3H), 4.76 (s, 2H), ...
example 3
Preparation of 3-methylcarbonyloxy-4-methyl-thiazole inner salt monohydrate (n=1)
[0107]2 g of the 3-methylcarbonyloxy-4-methyl-thiazole inner salt (n=0) as prepared in Example 2 was dissolved at 20° C. in a mixture solvent of 100 mL methanol and 1 mL water, after complete dissolution, 300 mL ethyl acetate solution was added slowly; after mixing homogeneously, standing was performed at 5° C. for 12 h, the precipitated crystal was 3-methylcarbonyloxy-4-methyl-thiazole inner salt monohydrate (n=1).
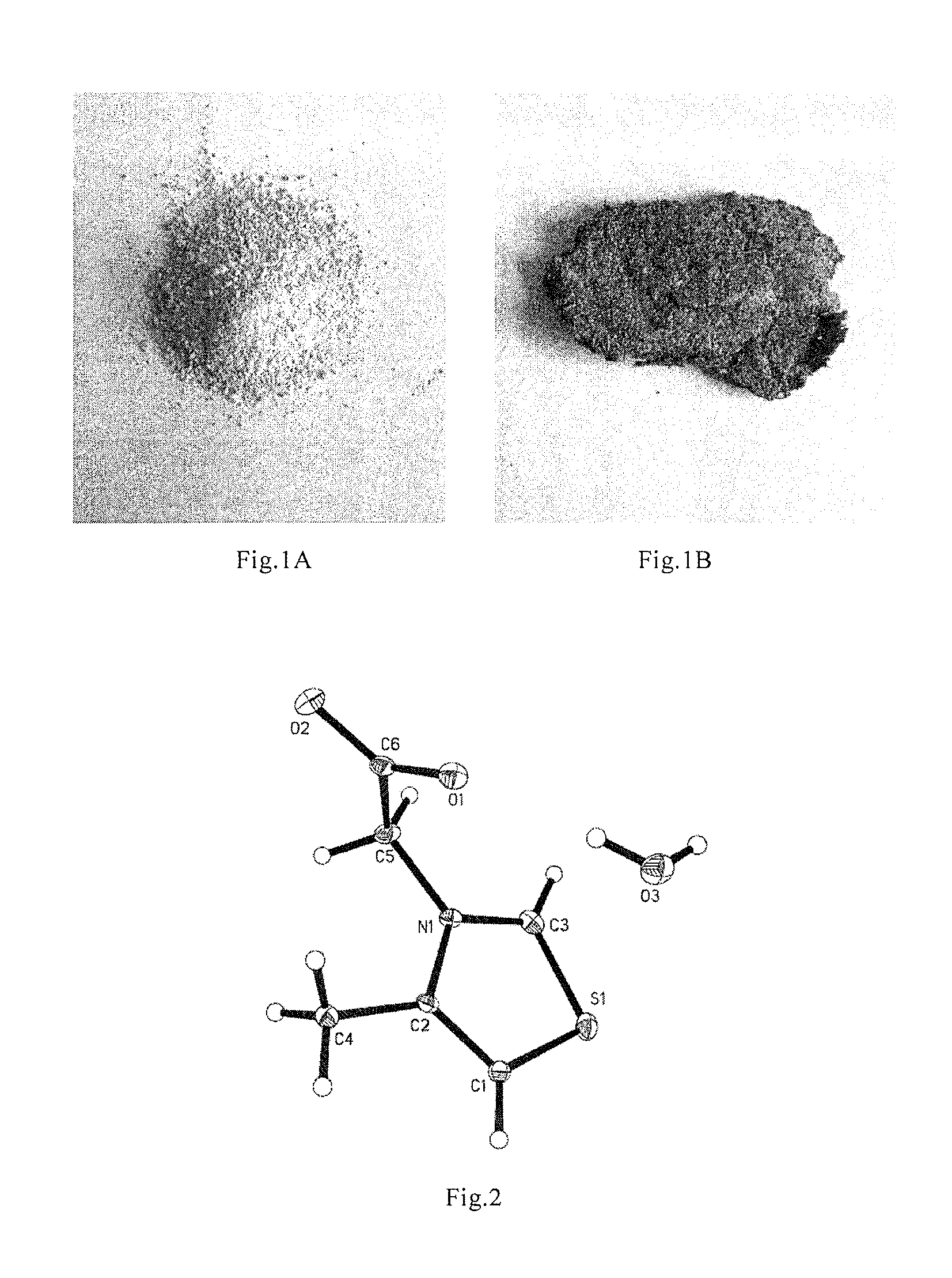
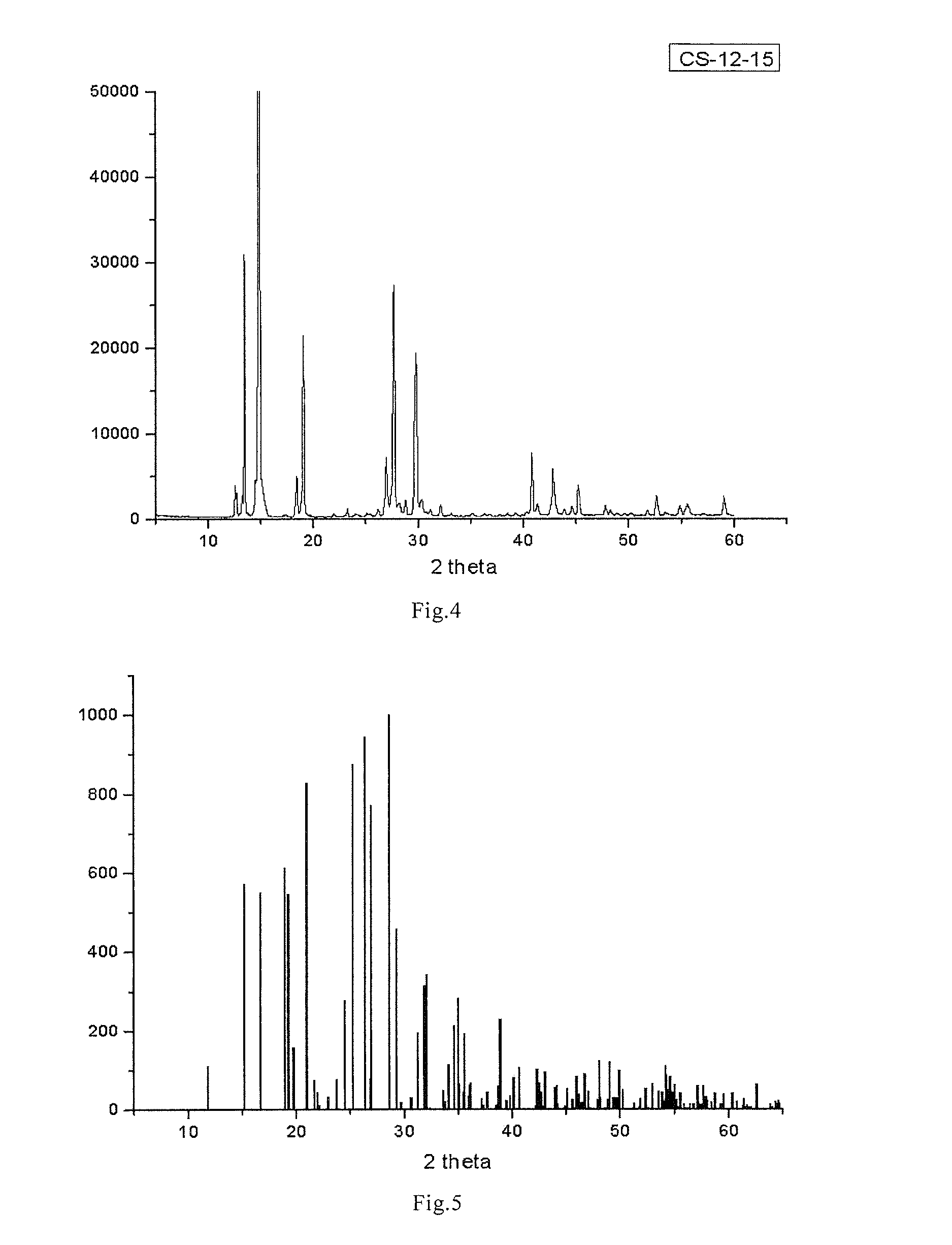
[0108]Test of Crystal Structure Determination with X-Ray Single Crystal Diffraction:
[0109]2 mg of 3-methylcarbonyloxy-4-methyl-thiazole inner salt white crystal was added with 0.1 mL of anhydrous methanol, after the particles were dissolved, 0.6 mL ethyl acetate was added drop-wisely, standing was carried out until crystal particles grew slowly to form monocrystals (3-methylcarbonyloxy-4-methyl-thiazole inner salt monohydrate, n=1). Crystal structure was determined with X-single crystal diffr...
example 4
[0113]Three batches of sample (prepared according to Example 2) were taken according to the Chinese Pharmacopoeia, 2010 Edition, packaged like packaging for sale (high-density polyethylene bags were used for drug packaging), placed under conditions of RT 40° C., RH 75% (NaCl saturated solution) to carry out accelerated test, after 1, 2, 3, 6 months, sampled for observation of the compound of Formula I and the compound of Formula A, compared to the data of the 0th day, and the results were shown in Table 1 and Table 2.
[0114]One batches of sample as prepared according to Example 3 were taken, packaged like packaging for sale (high-density polyethylene bags were used for drug packaging), placed under conditions of RT 40° C., RH 75% (NaCl saturated solution), after 1, 2, 3 months, sampled and compared to the data of the 0th day, and the results were shown in Table 3.
[0115]Three batches of sample of the compound of Formula A were taken according to the Chinese Pharmacopoeia...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Mass | aaaaa | aaaaa |
| Volume | aaaaa | aaaaa |
| Chemical properties | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More