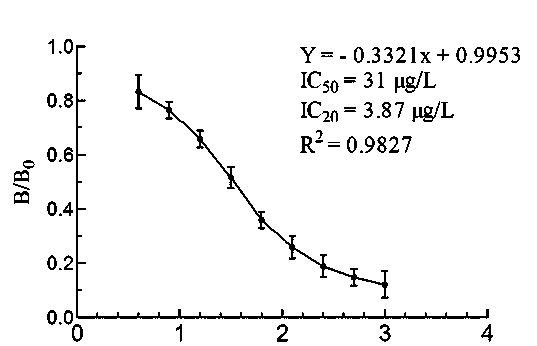Enzyme linked immunosorbent assay kit of sparfloxacin, establishing method and detecting method of enzyme linked immunosorbent assay kit
An enzyme-linked immunosorbent reagent, sparfloxacin technology, applied in the direction of measuring devices, instruments, scientific instruments, etc., can solve the problems of complicated operation process, limited application range, difficult to popularize and apply, etc., and achieve simple processing process, convenient use, Inexpensive effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0044] Example 1 Synthesis of Immunogen and Preparation of Monoclonal Antibody
[0045] 1.1 Reagents and instruments
[0046]Sparfloxacin was purchased from Sigma, N,N-dicyclohexylcarbodiimide and N-hydroxysuccinimide were purchased from Shanghai Chemical Reagent Company, bovine serum albumin, chicken ovalbumin, Freund's complete adjuvant and Freund's incomplete adjuvant from Pierce products.
[0047] Bio-Rad imark 680 microplate reader, AE260 electronic balance, purchased from Germany METTLER company; HI9321 acidity meter, American HANNA company; hand-held homogenizer, purchased from Germany IKA company; 93-3 time constant temperature two-way magnetic stirrer, purchased from Shanghai Yarong Biochemical Instrument Factory.
[0048] 1.2 Synthesis of sparfloxacin artificial antigen
[0049] (1) Weigh 3 mg of sparfloxacin, 3 mg of N-hydroxysuccinimide and 3 mg of N,N-dicyclohexylcarbodiimide, dissolve them in 1 mL of DMF solvent, and react at room temperature for 3 hours to ob...
Embodiment 2
[0060] Example 2 Establishment of immunoassay method
[0061] 2.1 ELISA method to determine the optimal coating concentration
[0062] Coat the enzyme with ovalbumin-sparfloxacin conjugate at a concentration of 10 μg / mL, 5 μg / mL, 2 μg / mL, 1 μg / mL, 0.5 μg / mL, and 0.25 μg / mL in an amount of 50 μL per well The target plate was coated at 4°C for 2 hours, washed 4 times, patted dry, added blocking solution, blocked at 4°C for 24 hours, washed 4 times, and patted dry. Join 1:8×10 4 50 μL / well of diluted antiserum was incubated at room temperature for 30 min, washed 4 times, and 50 μL / well of enzyme-labeled goat anti-mouse antibody was immediately added. React at room temperature for 30 minutes, wash 4 times, add chromogenic solution, 50 μL / well, chromogenic reaction at room temperature for 15 minutes, add 50 μL / well stop solution to terminate the reaction, and detect the A value with a microplate reader (450 nm). At the same time, set blank control wells (no antibody, only add it...
Embodiment 3
[0072] Example 3 Construction of the enzyme-linked immunosorbent assay kit for detecting sparfloxacin
[0073] Construct the ELISA kit of sparfloxacin to include the following components:
[0074] (1) ELISA plate coated with sparfloxacin artificially synthesized antigen;
[0075] (2) 2mL, 8×10 4 Times anti-sparfloxacin monoclonal antibody;
[0076] (3) Goat anti-mouse anti-antibody labeled with horseradish peroxidase;
[0077] (4) 6 bottles of sparfloxacin standard solution, the concentrations are 0μg / L, 10μg / L, 20μg / L, 30μg / L, 50μg / L, 100μg / L;
[0078] (5) Substrate chromogenic solution A is carbamide peroxide, and substrate chromogenic solution B is tetramethylbenzidine; chromogenic agent A and chromogenic agent B should be mixed in equal proportions.
[0079] (6) The washing solution is a phosphate buffer containing 0.05% Tween-20;
[0080] (7) The concentrated sample diluent is a phosphate buffered saline solution of 0.1% Tween-20;
[0081] (8) The stop solution is 2...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More