A method for the separation and determination of ezetimibe and related substances by high performance liquid chromatography
A high-performance liquid chromatography and ezetimibe technology, which is applied in the field of high-performance liquid chromatography for separation and determination of ezetimibe and related substances, can solve the problem that impurities cannot be detected, degraded impurities cannot be separated, and impurities cannot be completely separated and detected. problems, to achieve the effect of improving efficiency, accurate test results, and precise quality control.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0068] Embodiment 1 Determination of related substances of ezetimibe crude drug
[0069] Take appropriate amount of ezetimibe, related substance 1, related substance 2, related substance 3, related substance 4, related substance 5, and related substance 6, and use 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10) Dissolve and prepare a systemic suitability solution containing 200 μg of ezetimibe per 1 ml, 2 μg of each of related substance 1, related substance 2, related substance 3, related substance 4, related substance 5 and related substance 6.
[0070] Take ezetimibe reference substance, dissolve it with 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10) and prepare a reference substance solution containing 0.2μg per 1ml.
[0071] Take an appropriate amount of ezetimibe raw material, dissolve it with 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10), and prepare a test solution containing 200 μg of ezetimibe per 1 ml.
[0072] Take sy...
Embodiment 2
[0080] Example 2 Determination of Related Substances in Ezetimibe Preparations (Ezetimibe Tablets)
[0081] Take appropriate amount of ezetimibe, related substance 1, related substance 2, related substance 3, related substance 4, related substance 5, and related substance 6, and use 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10) Dissolve and prepare a systemic suitability solution containing 200 μg of ezetimibe per 1 ml, 2 μg of each of related substance 1, related substance 2, related substance 3, related substance 4, related substance 5 and related substance 6.
[0082] Take ezetimibe reference substance, dissolve it with 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10) and prepare a reference substance solution containing 0.4μg per 1ml.
[0083] Grind ezetimibe tablets finely, take an appropriate amount, and use 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10) to sonicate for 30 minutes (shake at any time), and prepare a supply co...
Embodiment 3
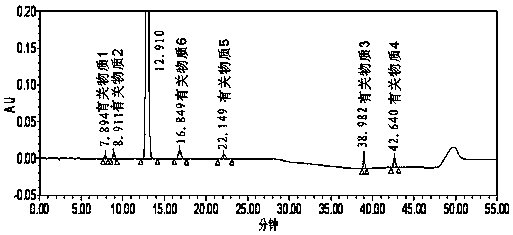
[0103] Example 3 Detection of related substances after ezetimibe alkali degradation
[0104] Chromatographic column: phenylsilane bonded silica gel as filler (Phenomenex Luna Phenyl Hexyl 250mm×4.6mm, 5μm);
[0105] Mobile phase: 0.1% phosphoric acid solution: acetonitrile: methanol (48:42:10);
[0106] Detection wavelength: 232nm;
[0107] Injection volume: 30μl;
[0108] Detection time: 25 min (isocratic elution part of the method of the present invention).
[0109] Take an appropriate amount of ezetimibe, put it in a measuring bottle, add 10ml of 0.01mol / L ethanol-made sodium hydroxide solution to dissolve, seal it tightly, heat at 55°C for 15 minutes, take it out, immediately add 2ml of 0.1mol / L hydrochloric acid solution, and use 0.1% phosphoric acid solution-acetonitrile-methanol (48:42:10) is dissolved, is mixed with the need testing solution that every 1ml contains ezetimibe 200 μ g, gets this need testing solution and injects high performance liquid chromatograph, ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
| wavelength | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More