Choline M receptor antagonist aclidinium bromide and preparation method thereof
A technology of receptor antagonist and aclidinium bromide, which is applied in the field of respiratory system drug aclidinium bromide and its preparation, can solve the problems of high production cost, high production cost and long reaction route of aclidinium bromide, and achieve easy The effect of industrialized production, low cost and short reaction route
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
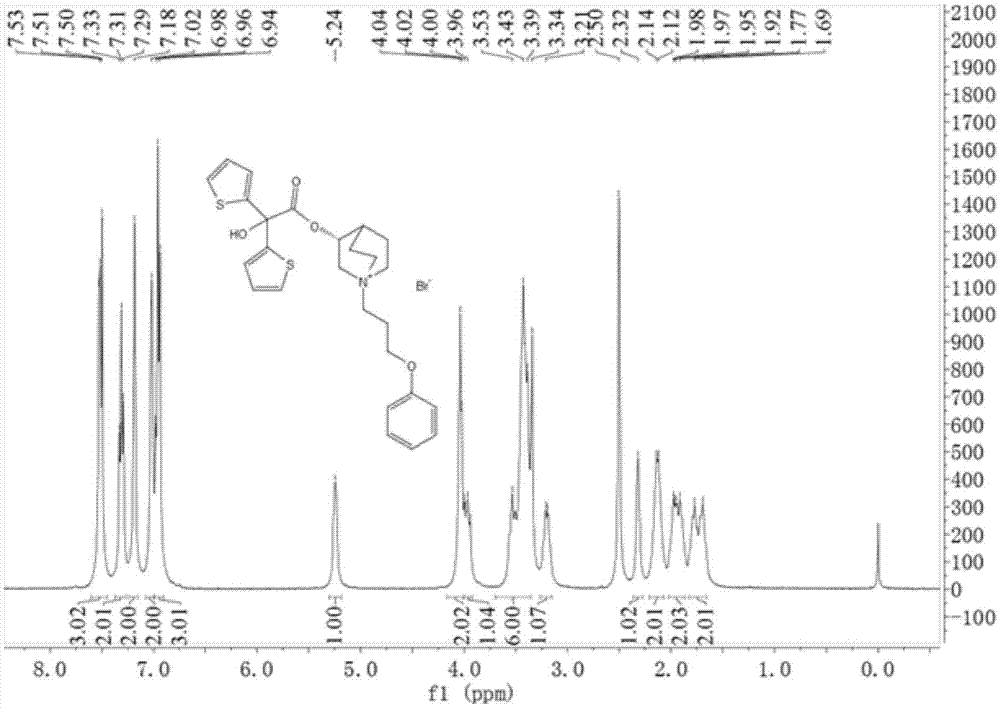
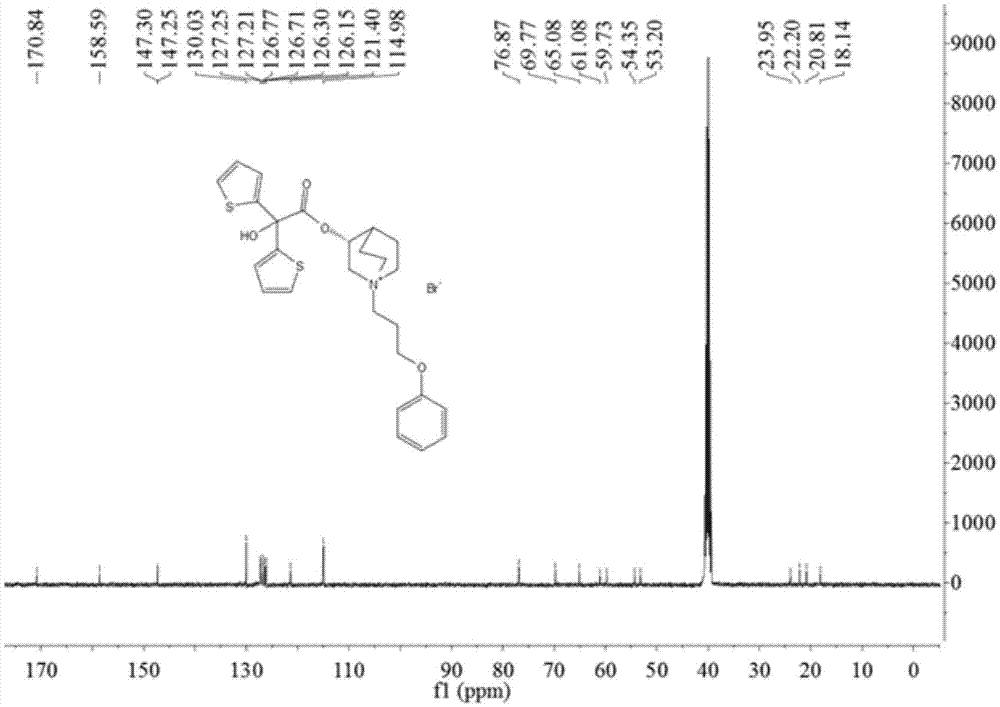
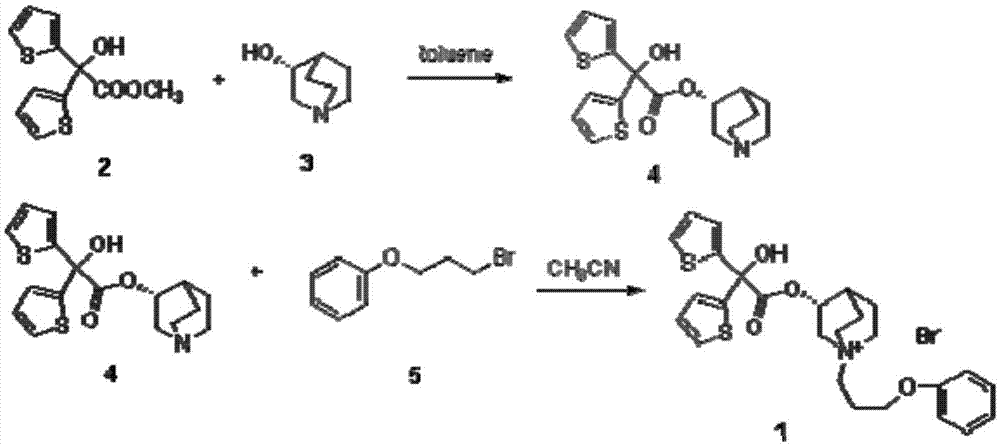
[0041] Add 3-quinine alcohol (38.1g, 0.3mol) and 600mL anhydrous toluene into a 1000mL three-necked flask, add 60% sodium hydride (13.2g, 0.33mol) in batches, stir at room temperature for 15min, then add 2-hydroxyl -2,2-bis(2-thienyl)acetic acid methyl ester (76.2g, 0.03mol), reflux reaction for 6h. After the reaction is complete, add 250 mL of water and stir for 20 min, separate the toluene layer, dry over anhydrous sodium sulfate and spin off the solvent to obtain the crude product. Ethanol was recrystallized to obtain 54.5 g of white solid, yield: 52.1%. Mp: 173~175℃. IR:3435,3075,2946,2876,2735,1732,1637,1566,1423,1354,1320,1232,1132,1085,997,837,784,689. 1 H-NMR (400MHz, DMSO-d 6 )δ:1.24(m,2H),1.54-1.58(m,3H),1.91(m,1H),2.45(m,1H),2.58-2.60(m,3H),3.07-3.36(m,1H) ,4.80-4.82(m,1H),6.69-7.02(m,2H),7.11-7.12(m,2H),7.36(m,1H),7.49-7.51(m,2H).
Embodiment 2
[0043] 2-Hydroxy-2,2-di(2-thiophene) acetic acid-3-quinyl ester (15.0 g, 0.043 mol), L-tartaric acid (6.45 g, 0.043 mol), 180mL of absolute ethanol was heated to reflux for 30min, and the reaction solution was clarified. After cooling to room temperature, a white solid precipitated out. Suction filtration yielded 8.4 g of white solid, with a yield of 39.1%. [α] D 25 =+116.1°(c=0.8g / 100mL, H 2 O). 1 HNMR (400MHz, DMSO-d 6 )δ: 1.08(d, J=6.4Hz, 2H), 1.65(dd, J=66.5, 27.4Hz, 4H), 2.13(s, 1H), 2.93(t, J=33.3Hz, 5H), 3.41( dd,J=19.3,12.0Hz,2H),4.06(s,2H),5.04(s,1H),7.01(s,2H),7.14(s,2H),7.50(s,2H),7.73(s ,2H).
[0044] Add tartrate prepared in the previous step and 5 mL of ethanol to a 50 mL eggplant-shaped bottle, add sodium hydroxide solution to adjust the pH to 7-8, stir at room temperature for 30 min, cool, and extract with chloroform. The solvent was removed to obtain 5.1 g of white solid R-2-hydroxy-2,2-bis(2-thienyl)acetic acid-3-quinyl ester, Mp: 175-178°C, the yiel...
Embodiment 3
[0046] Add 3-quinine alcohol (25.4g, 0.2mol) and 600mL anhydrous toluene to a 1000mL three-necked flask, add potassium tert-butoxide (23.5g, 0.21mol) in batches, stir at room temperature for 15min, then add 2-hydroxyl -2,2-bis(2-thienyl)acetic acid methyl ester (50.8g, 0.2mol), reflux reaction for 6h. After the reaction was completed, add 300 mL of water and stir for 20 min, separate the toluene layer, dry over anhydrous sodium sulfate, evaporate the solvent to obtain a crude product. Recrystallization from ethanol gave 39.1 g of white solid quinine-3-yl 2,2-bis(2-thienyl)-2-hydroxyacetate, yield: 56.1%. Mp: 173~175℃. Prepared, yield 58.0%, mp: 172~175°C.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap