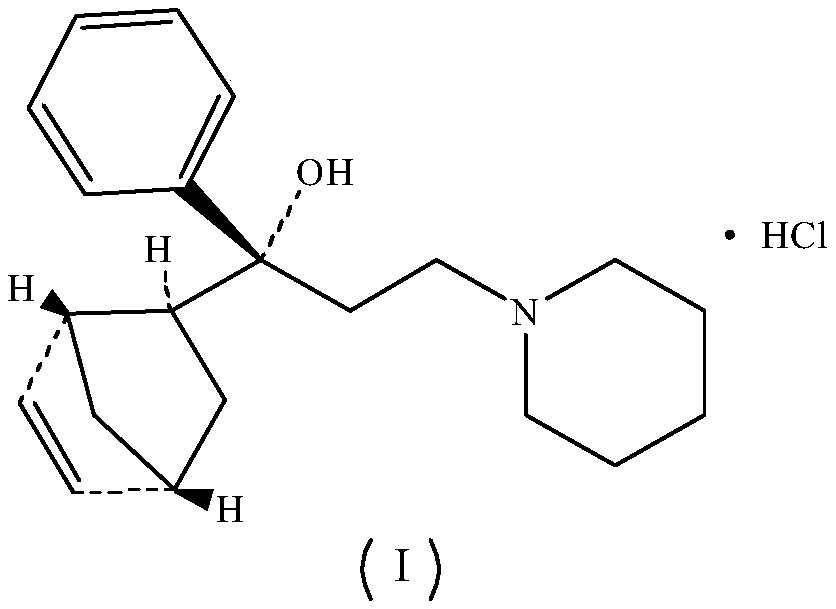
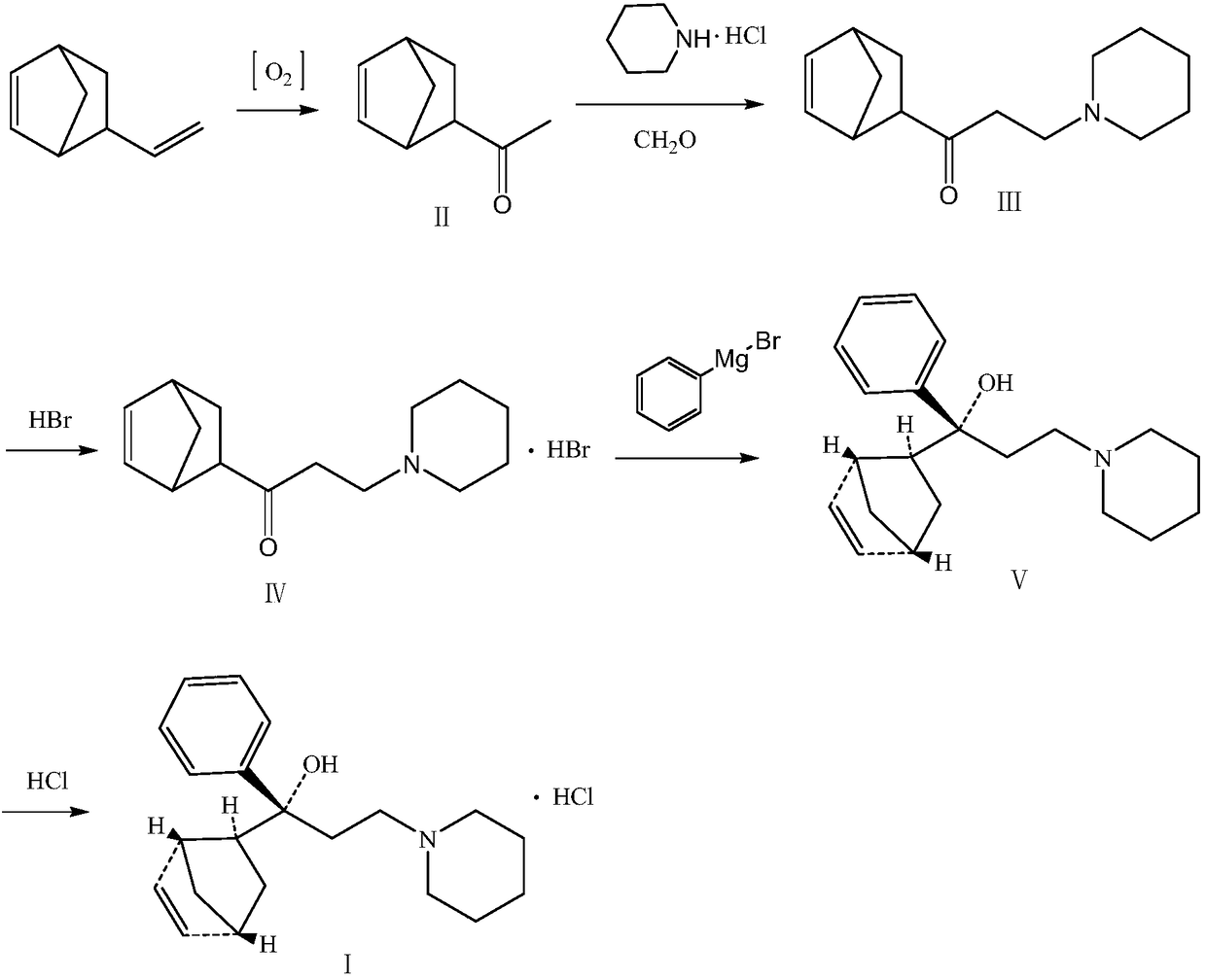
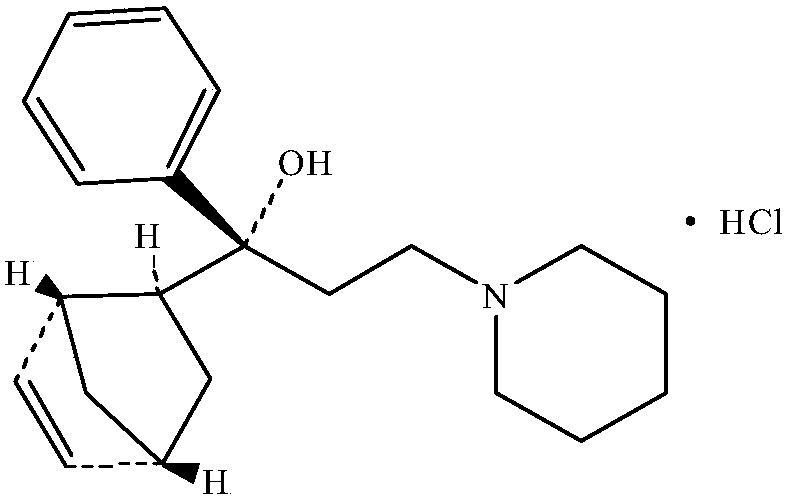
Preparation method of biperiden hydrochloride
A technology for biperiden hydrochloride and intermediates, which is applied in the field of drug synthesis technology, can solve the problems of high internal impurities, pollution, and low yield of hydrochloric acid, and achieve the effects of saving costs and reducing pollution
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0048] Embodiment one: the preparation of biperiden hydrochloride
[0049] a. Preparation of Intermediate II
[0050] 84g (0.7mol) of 5-ethylene-2-norbornene, 325ml of acetonitrile, 65ml of deionized water, 3.6g of palladium dichloride and 1.8g of copper chloride were added to the autoclave, and the 2 Displacing air, passing O 2, the temperature is maintained at 80-90°C, the pressure is at 15-16 atmospheres, react for 4 hours, GC identifies the reaction end point, the reaction is completed, cooled to room temperature, filtered with diatomaceous earth, and the filtrate is concentrated under reduced pressure to obtain 65.8g (0.483mol) Colorless oily liquid intermediate II, with a yield of 69.1% and a GC content of 99.1%, was directly used in the next step.
[0051] b. Preparation of Intermediate III
[0052] Glacial acetic acid 480ml, piperidine hydrochloride 60.8g (0.5mol), paraformaldehyde 14.5g (0.5mol), intermediate II 61.3g (0.45mol), heated to 95-98°C under stirring, an...
Embodiment 2
[0059] Embodiment two: the preparation of biperiden hydrochloride
[0060] a. Preparation of Intermediate II
[0061] 59g (0.5mol) of 5-ethylene-2-norbornene, 250ml of DMF, 44ml of deionized water, 2.5g of palladium dichloride and 1.25g of copper chloride were added to the autoclave and heated with N 2 Displacing air, passing O 2 , the temperature is maintained at 85-90°C, the pressure is at 15-17 atmospheres, react for 3h, GC identifies the reaction end point, the reaction is completed, cooled to room temperature, filtered with diatomaceous earth, and the filtrate is concentrated under reduced pressure to obtain 46g (0.338mol) of The color oily liquid intermediate II, with a yield of 69.7% and a GC content of 99.5%, was directly used in the next step.
[0062] b. Preparation of Intermediate III
[0063] 350ml of glacial acetic acid, 43g (0.35mol) of piperidine hydrochloride, 10.2g (0.35mol) of paraformaldehyde, 43g (0.315mol) of intermediate II, heated to 95-98°C under sti...
Embodiment 3
[0070] Embodiment three: the detection of biperiden hydrochloride
[0071] Instrument: Shimadzu GC-16A gas chromatograph, C-R3A chromatographic processor, poly(dimethyl)-(diphenyl)-(divinyl)siloxane R-50m×0.25mm fused silica glass column ;
[0072] Detector: hydrogen flame ion detector (FID); detection chamber, vaporization chamber temperature: 300°C; carrier gas: N240ml / min; H250ml / min; air 500ml / min; attenuation: 3; injection volume: 2μl; column temperature Follow the table below:
[0073]
[0074] Result: GC content is 99.8%, and the detected impurities are shown in the table below:
[0075]
[0076] The hydrochloric acid prepared by the invention has better quality than periden.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More