Madecassoside liposomal gel for local external application and preparation method thereof
The technology of madecassoside and liposome gel is applied in the field of pharmaceutical preparations and skin care products, which can solve the problems of increased skin dosage, strong fluidity, waste of drugs, etc., so as to be beneficial to wound healing without irritation. Effect, good fit effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0044](1) Preparation of madecassoside liposome solution: weigh 0.50 g of egg yolk lecithin, 0.10 g of cholesterol, and 0.1 g of VE acetate and dissolve it in 10 mL of dichloromethane to prepare an organic solution; remove the organic solvent by rotary evaporation To form a film, add 20 mg / mL madecassoside solution into the film, add an appropriate amount of glass beads and stir at 50°C for 1 hour, sonicate with a probe for 30 minutes, and pass through a 0.22 μm water-based filter membrane to obtain a madecassoside liposome solution. In the madecassoside liposome solution, the mass of madecassoside accounts for 22.22%, and the mass of egg yolk lecithin accounts for 55.56%.
[0045] (2) Preparation of blank gel: add 0.1 g of azone and 1.5 g of propylene glycol in 8 mL of water, and then add 0.2 g of sodium carboxymethyl cellulose, and stir thoroughly to obtain a blank gel.
[0046] (3) Add 2 mL of madecassoside liposome solution in step (1) to the blank gel in step (2), stir we...
Embodiment 2
[0049] (1) Preparation of madecassoside liposome solution: Weigh 0.50 g of egg yolk lecithin, 0.10 g of cholesterol, and 0.1 g of VE acetate and dissolve it in 10 mL of ethanol to prepare an organic phase solution; Add dropwise into 15 mg / mL madecassoside solution, stir while adding dropwise until the solvent is removed, and pass through a 0.22 μm oily filter membrane to obtain madecassoside liposome solution. In the madecassoside liposome solution, the mass of madecassoside accounts for 17.65%, and the mass of egg yolk lecithin accounts for 58.82%.
[0050] (2) Preparation of blank gel: add 0.1 g of azone and 2.0 g of propylene glycol in 7.5 mL of water, and then add 0.4 g of sodium carboxymethylcellulose, and stir thoroughly to obtain a blank gel.
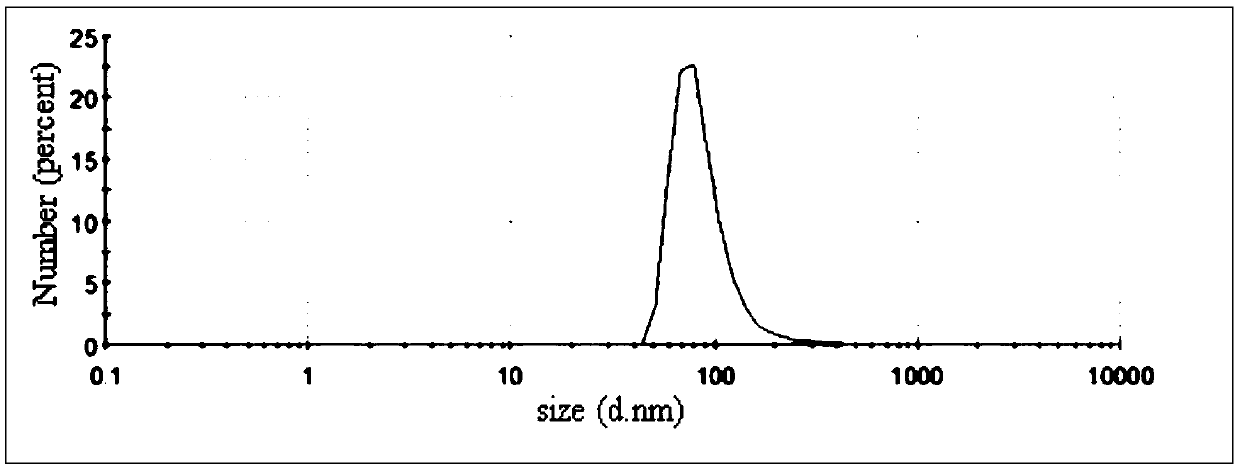
[0051] (3) Add 2.5mL madecassoside liposome solution in step (1) to the blank gel in step (2), adjust the pH value to 7 with 7% NaOH solution to obtain madecassoside plastid gel. The madecassoside liposome average particle diam...
Embodiment 3
[0054] (1) Preparation of madecassoside liposome solution: weigh 0.50 g of egg yolk lecithin, 0.10 g of cholesterol, and 0.1 g of VE acetate and dissolve it in 10 mL of dichloromethane to prepare an organic solution; remove the organic solvent by rotary evaporation To form a film, add 10mg / mL madecassoside solution into the film, rotate and hydrate to obtain a liposome solution, add 0.005g mannitol, mix well, freeze-dry in vacuum, shake the liposome freeze-dried powder with PBS Shake to disperse to obtain madecassoside liposome solution. In the madecassoside liposome solution, the mass of madecassoside accounts for 12.42%, and the mass of egg yolk lecithin accounts for 62.11%.
[0055] (2) Preparation of blank gel: add 0.1 g of azone and 1.8 g of propylene glycol in 7 mL of water, and then add 0.6 g of sodium carboxymethylcellulose, and stir thoroughly to obtain a blank gel.
[0056] (3) Add 2 mL of madecassoside liposome solution in step (1) to the blank gel in step (2), adj...
PUM
| Property | Measurement | Unit |
|---|---|---|
| The average particle size | aaaaa | aaaaa |
| The average particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More