Air-dried amniotic membrane powder and application
An amniotic membrane, air-drying technology, applied in powder delivery, medical raw materials derived from mammals, drug combinations, etc., can solve problems such as disease transmission, inconvenience in use, and difficulty in sterilization
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] Embodiment 1: the preparation method of air-dried amnion powder, comprises the following steps:
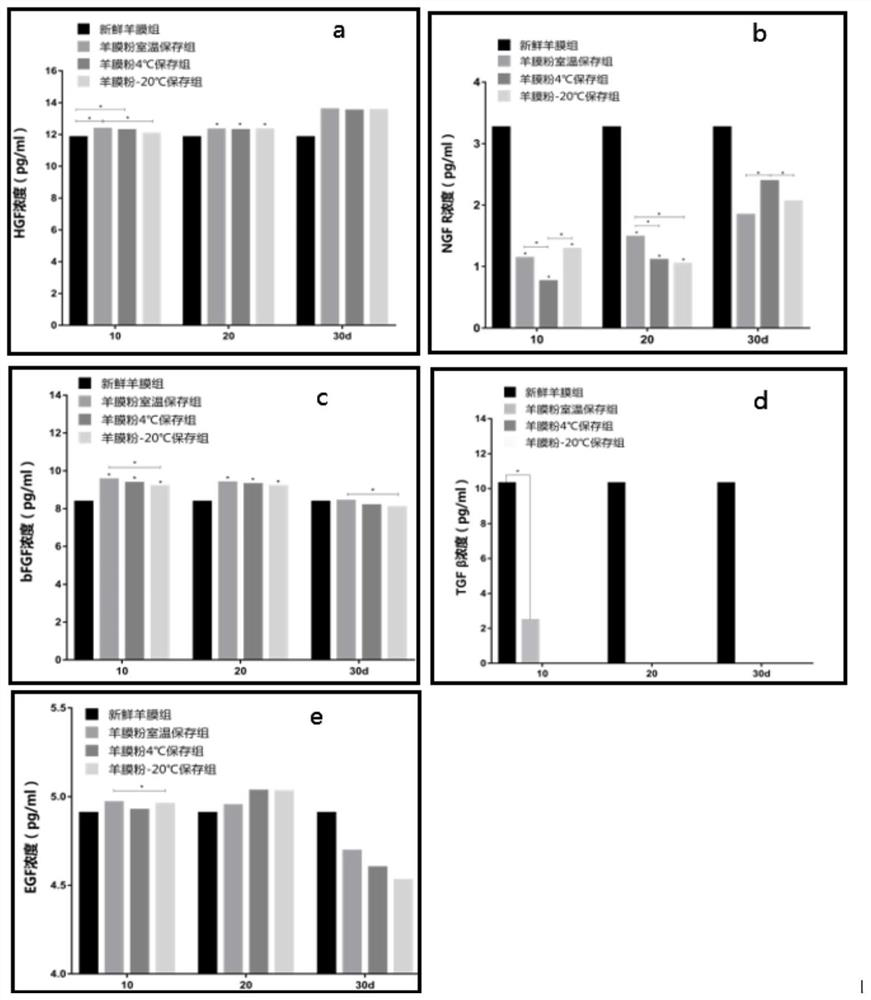
[0030] S1. Obtain amniotic membranes. Select cesarean section pregnant women who are negative for HIV, HBV, HCV, syphilis and other infectious diseases. Obtain discarded placenta within 24 hours after delivery, and bluntly separate the amniotic membranes.
[0031] S2, wash the blood clots and mucus on the amniotic membrane with sterile normal saline, and then place it in normal saline containing 50ug / ml penicillin-streptomycin, 100ug / ml neomycin, and 215ug / ml amphotericin B for 45min repeatedly. , and finally rinsed 3 times with sterile saline.
[0032] S3, air-dry the amniotic membrane, flatten the washed amniotic membrane, fix it on a support frame, put it into a 37°C constant temperature circulating air drying oven, and let it stand for 2 hours to fully remove the water to obtain the air-dried amniotic membrane.
[0033] S4, Grind into powder, put the amnion tissue drie...
Embodiment 2
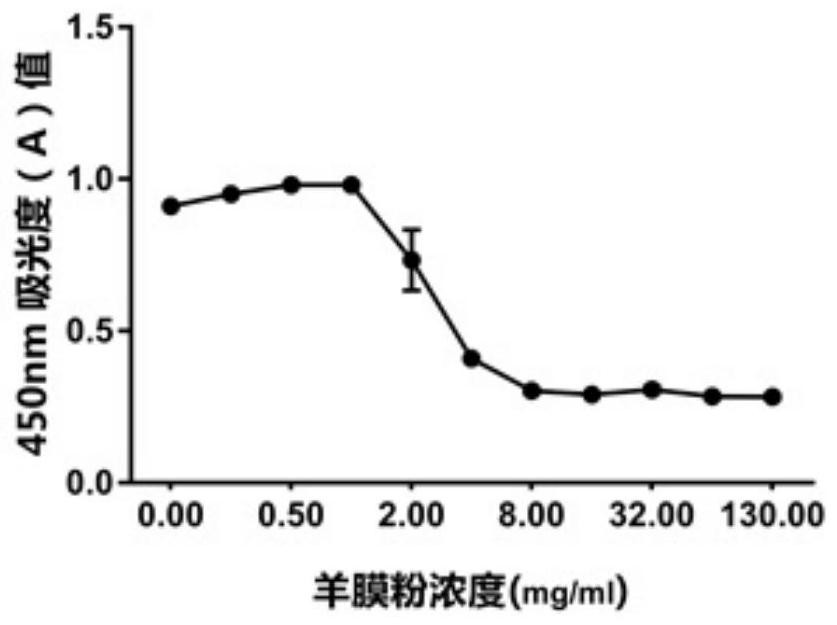
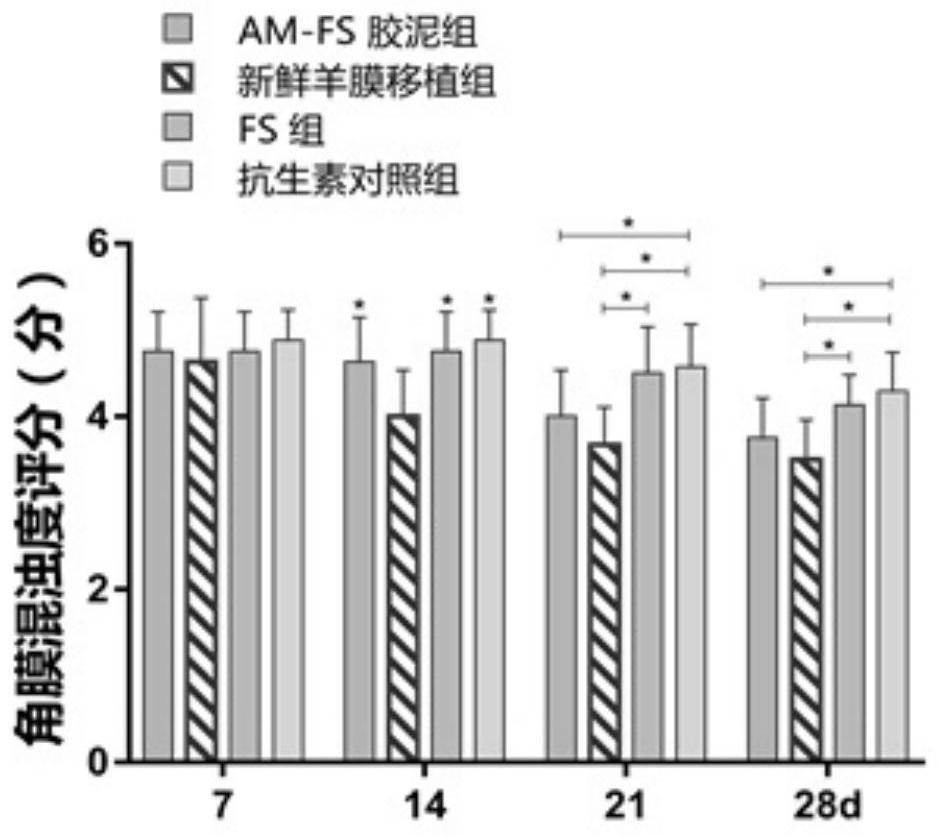
[0042] Example 2: Amniotic membrane-fibrin cement, amniotic membrane-fibrin cement (AM-FS: Amniotic membrane-Fibrin Sealant) containing different concentrations of amniotic membrane powder, the preparation steps are as follows:
[0043] S1, fully mix the amniotic membrane powder prepared in Example 1 with the catalyst solution in pig fibrin glue, and prepare respectively. ml, 4mg / ml, 2mg / ml, 1mg / ml, 0.5mg / ml, 0.25mg / ml, 0mg / ml amnion powder-catalyst mixture.
[0044] S2, sequentially take 50ul of amnion powder-catalyst with the above concentration, respectively add them to 24-well cell culture plates, spread them evenly on the bottom of the culture plates, add an equal volume of main glue solution to each of the above culture wells in turn, oscillate gently, mix evenly, and form According to the mass ratio, the concentration of amniotic membrane powder is: 65mg / ml, 32mg / ml, 16mg / ml, 8mg / ml, 4mg / ml, 2mg / ml, 1mg / ml, 0.5mg / ml, 0.25mg / ml, 0.125mg / ml ml, 0mg / ml AM-FS cement.
[004...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com