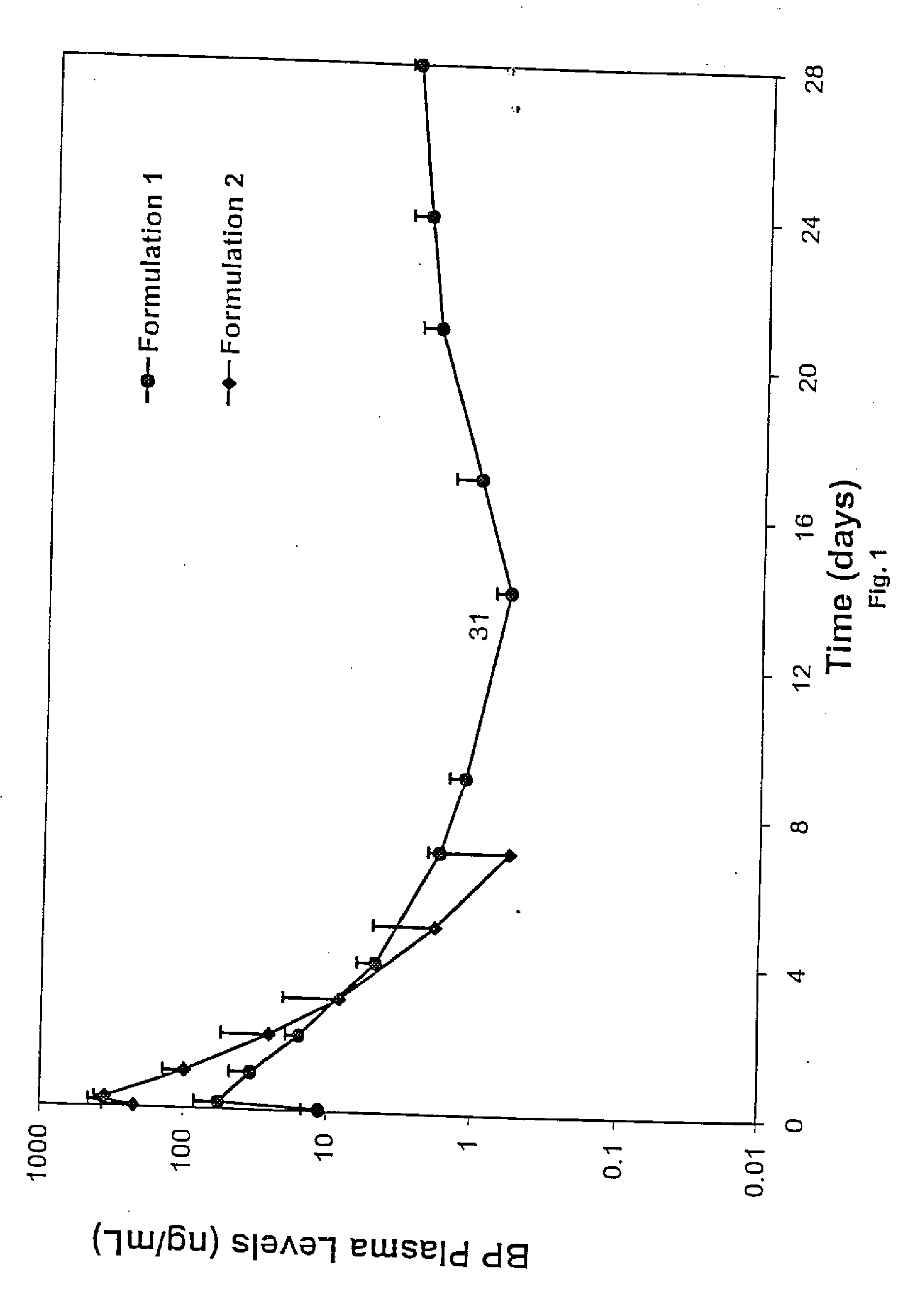
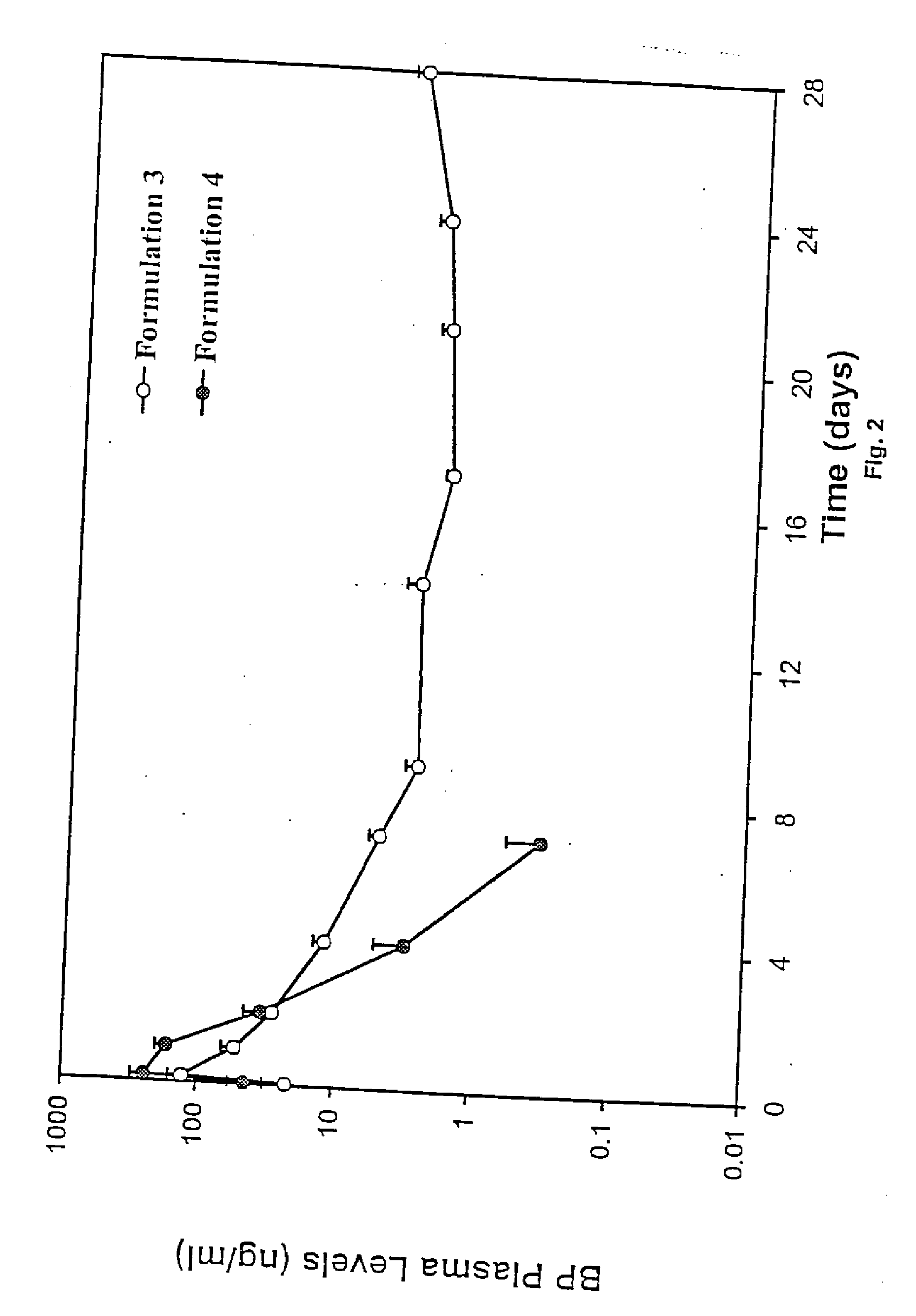
Short duration depot formulations
a depot and formulation technology, applied in the field of depot gel compositions, can solve the problems of reluctance of patients to accept such an implant or drug delivery system, important limitations of their use in the body of various animals, and materials that do not always meet the demand for biodegradable implants
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Depot Gel Preparation
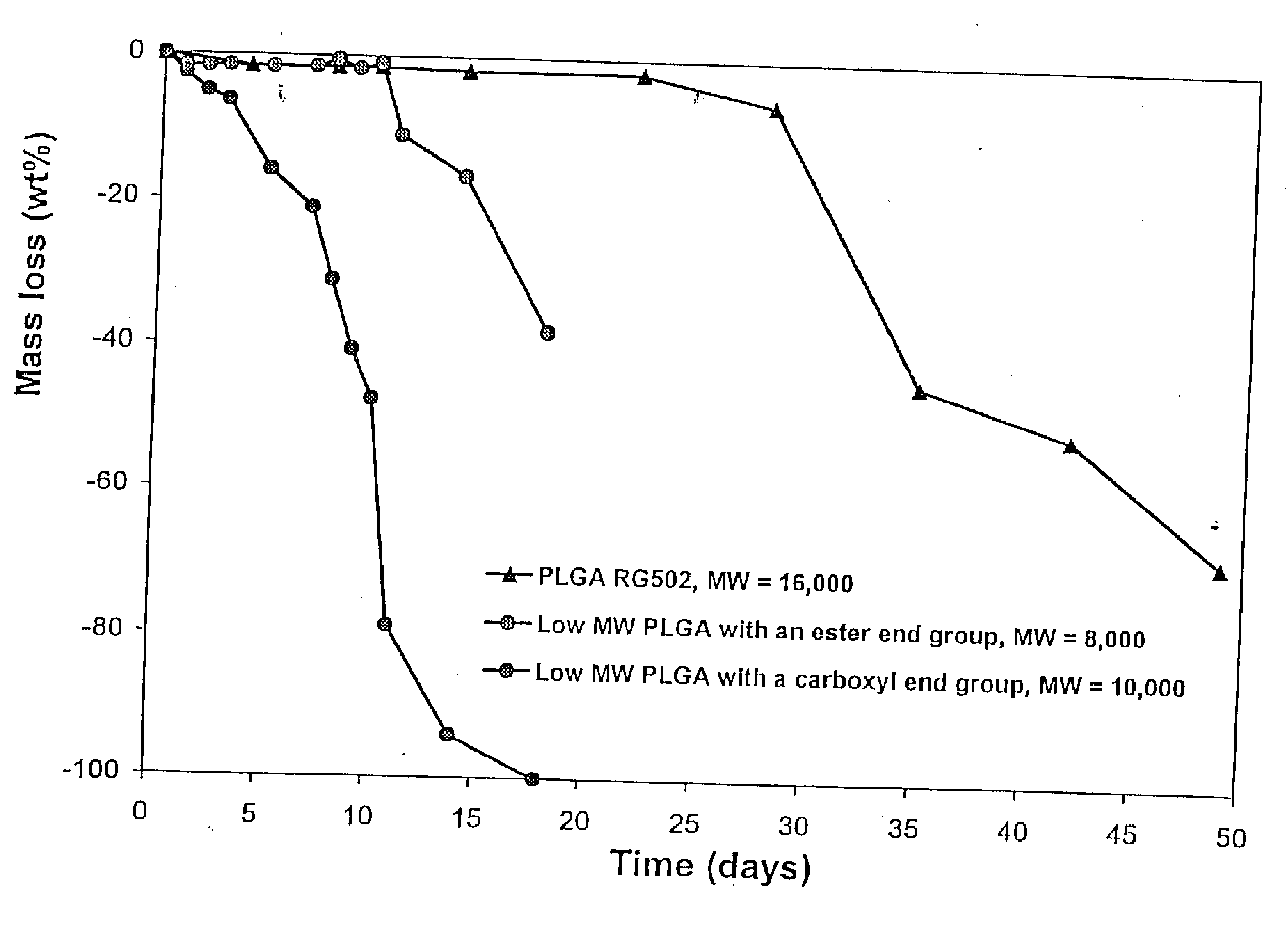
[0151] A gel vehicle for use in an injectable depot of the composition was prepared as follows. A glass vessel was tared on a Mettler PJ3000 top loader balance. Poly (D,L-lactide-co-glycolide) (PLGA), available as 50:50 DL-PLG with an inherent viscosity of 0.15 (PLGA-BPI, Birmingham Polymers, Inc., Birmingham, Ala.) and 50:50 Resomer® R RG502 (PLGA RG 502), was weighed into the glass vessel. The glass vessel containing the polymer was tared and the corresponding solvent was added. Amounts expressed as percentages for various polymer / solvent combinations are set forth in Table 1, below. The polymer / solvent mixture was stirred at 250±50 rpm (IKA electric stirrer, IKH-Werke GmbH and Co., Stanfen, Germany) for about 5-10 minutes, resulting in a sticky paste-like substance containing polymer particles. The vessel containing the polymer / solvent mixture was sealed and placed in a temperature controlled incubator equilibrated to 37° C. for 1 to 4 days, with intermitten...
example 2
hGH Particle Preparation
[0153] Human growth hormone (hGH) particles (optionally containing zinc acetate) were prepared as follows: hGH solution (5 mg / ml) solution in water (BresaGen Corporation, Adelaide, Australia) was concentrated to 10 mg / mL using a Concentration / Dialysis Selector diafiltering apparatus. The diafiltered hGH solution was washed with 5 times volume of tris or phosphate buffer solution (pH 7.6). Particles of hGH were then formed by spray drying or lyophilization using conventional techniques. Phosphate buffer solutions (5 or 50 mM) containing hGH (5 mg / mL) (and optionally various levels of zinc acetate (0 to 30 mM) when Zn complexed particles were prepared) were spray-dried using a Yamato Mini Spray dryer set at the following parameters:
Spray Dryer ParameterSettingAtomizing Air2 psiInlet Temperature120° C.Aspirator Dial7.5Solution Pump2-4Main Air Valve40-45 psi
[0154] hGH particles having a size range between 2-100 microns were obtained. Lyophilized particles were...
example 3
hGH-Stearic Acid Particle Preparation
[0155] Human growth hormone (hGH) particles were prepared as follows: Lyophilized hGH (3.22 grams, Pharmacia-Upjohn, Stockholm, Sweden) and stearic acid (3.22 grams, 95% pure, Sigma-Aldrich Corporation, St. Louis, Mo.) were blended and ground. The ground material was compressed in a 13 mm round die, with a force of 10,000 pounds for 5 minutes. Compressed tablets were ground and sieved through a 70 mesh screen followed by a 400 mesh screen to obtain particles having a size range between 38-212 microns.
PUM
| Property | Measurement | Unit |
|---|---|---|
| miscibility | aaaaa | aaaaa |
| wt. % | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More