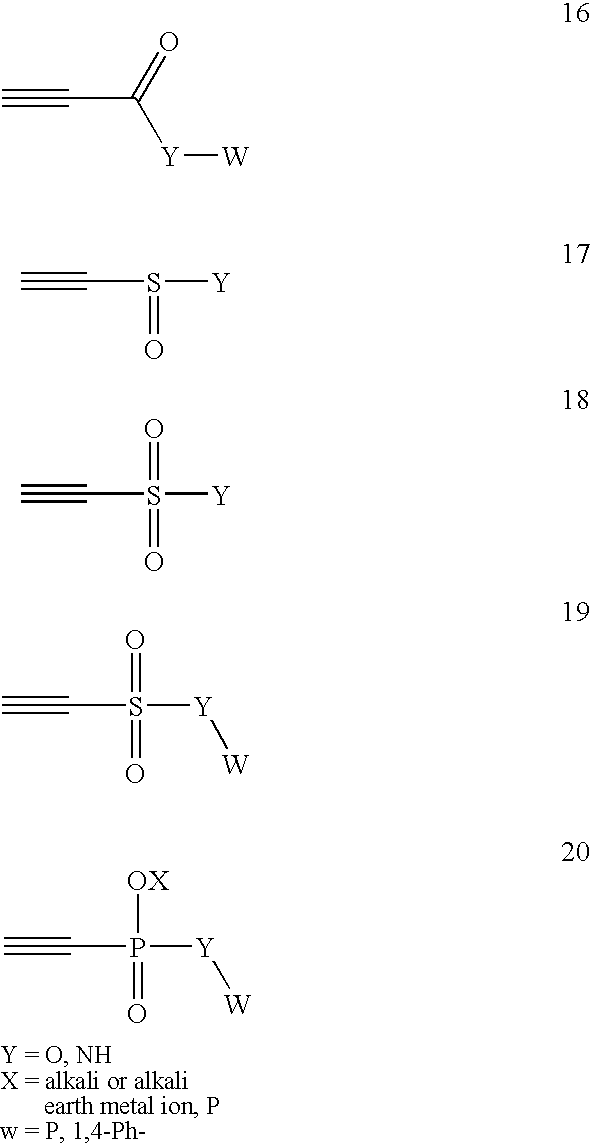Patents
Literature
1038 results about "Controlled delivery" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
CONTROLLED DELIVERIES If a package is identified as containing a controlled substance, the authorities can conduct what the government calls a controlled delivery of the package.
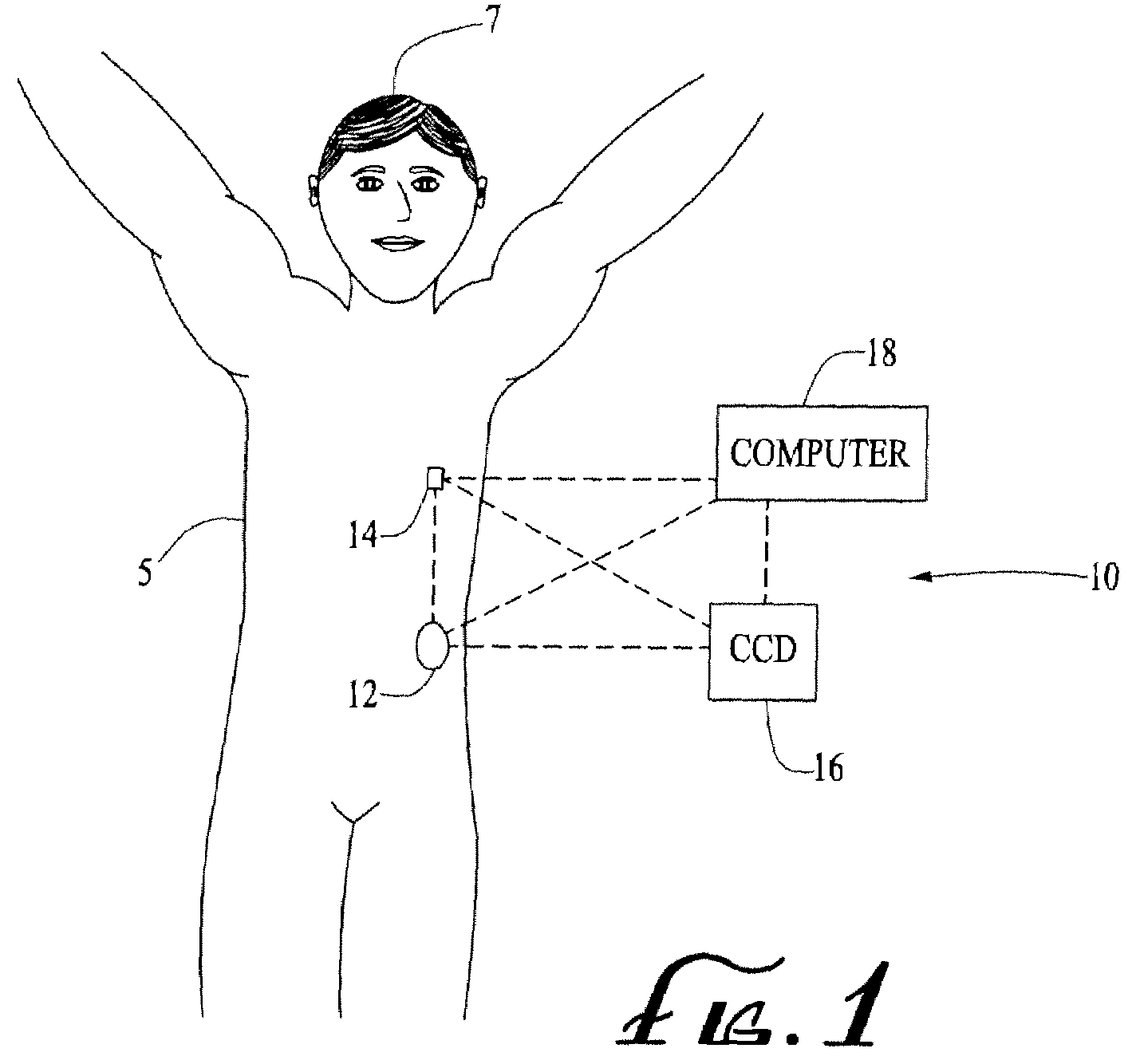
Energetically-controlled delivery of biologically active material from an implanted medical device
InactiveUS7101394B2Facilitated releaseReduce deliveryOrganic active ingredientsElectrotherapyMedical deviceBiomedical engineering
A medical device and system capable of providing on-demand delivery of biologically active material to a body lumen patient, and a method of making such medical device. A first coating layer comprising a biologically active material and optionally a polymeric material is disposed on the surface of the medical device. A second coating layer comprising magnetic particles and a polymeric material is disposed on the first coating layer. The second coating layer, which is substantially free of a biologically active material, protects the biologically active material prior to delivery. The system includes the medical device and a source of energy, such as an electromagnetic or mechanical vibrational energy. When the patient is exposed to the energy source, the magnetic particles move out of the second coating layer and create channels therein through which the biologically active material can be released.
Owner:BOSTON SCI SCIMED INC
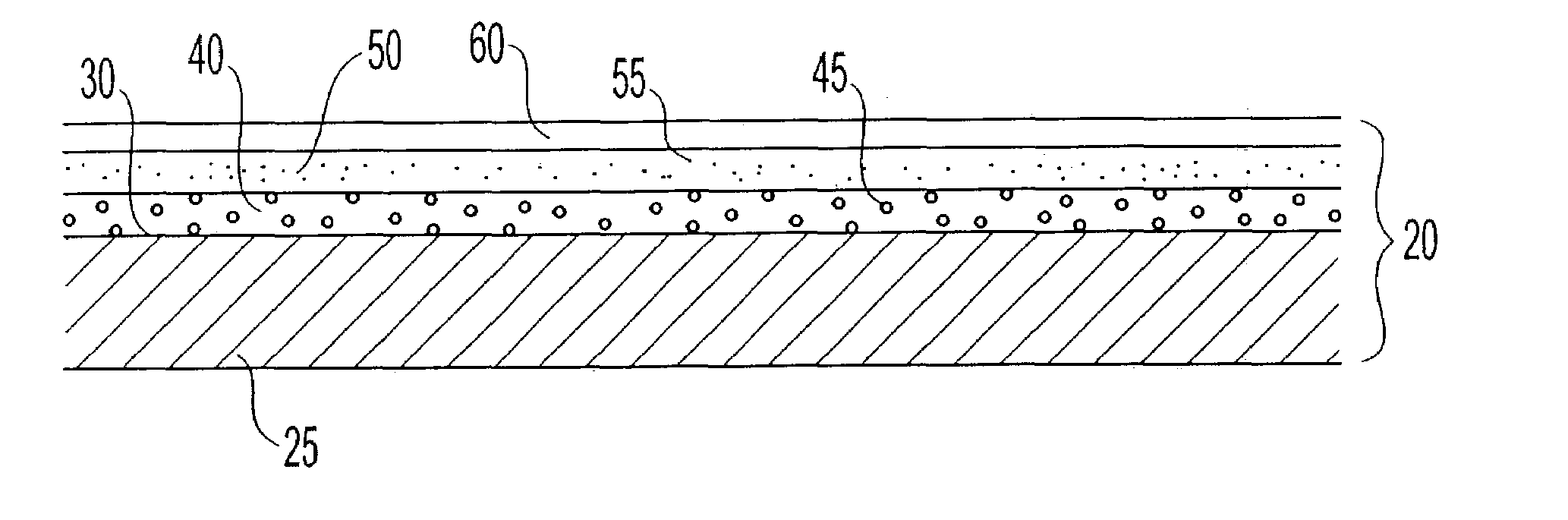
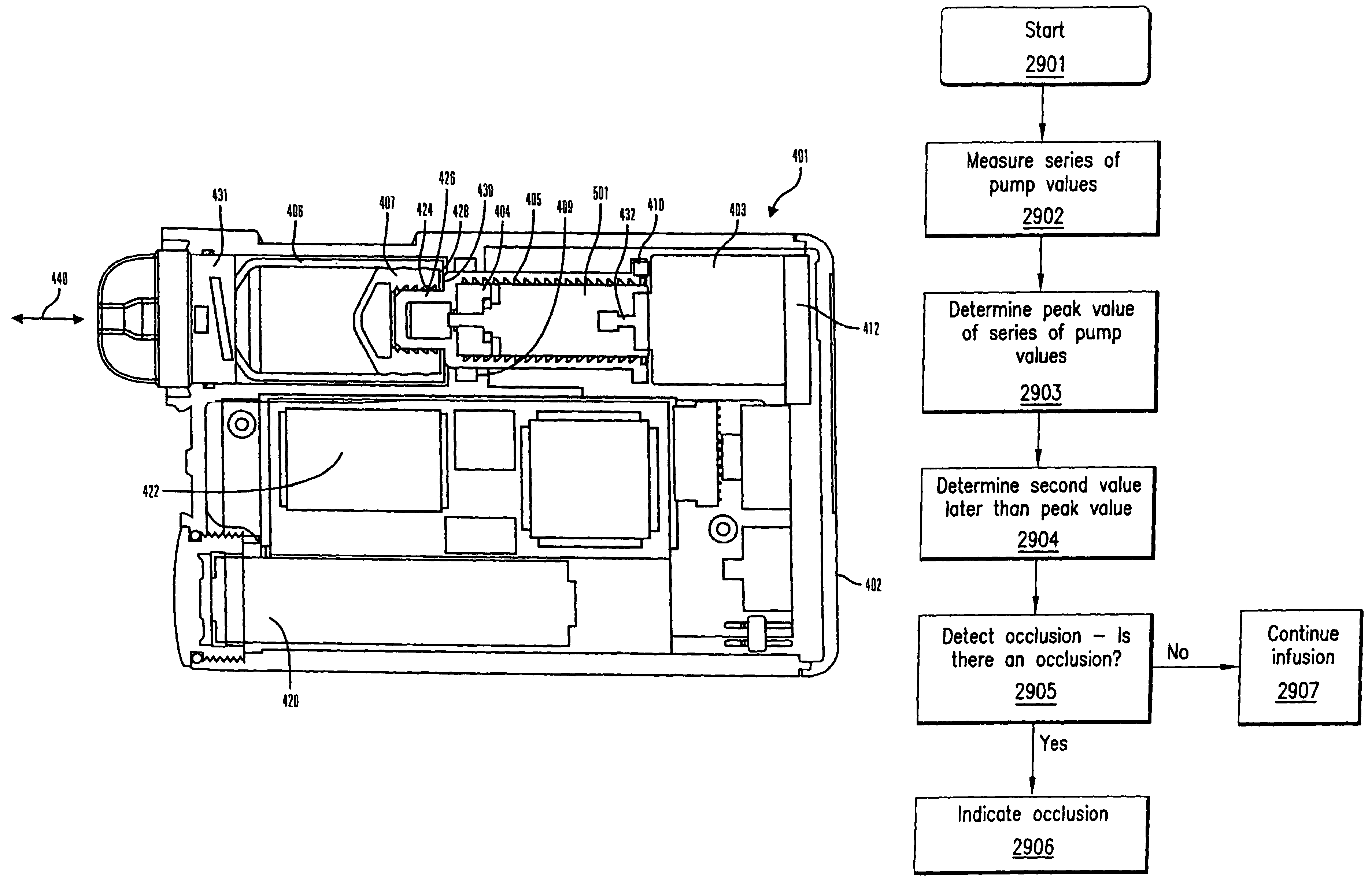
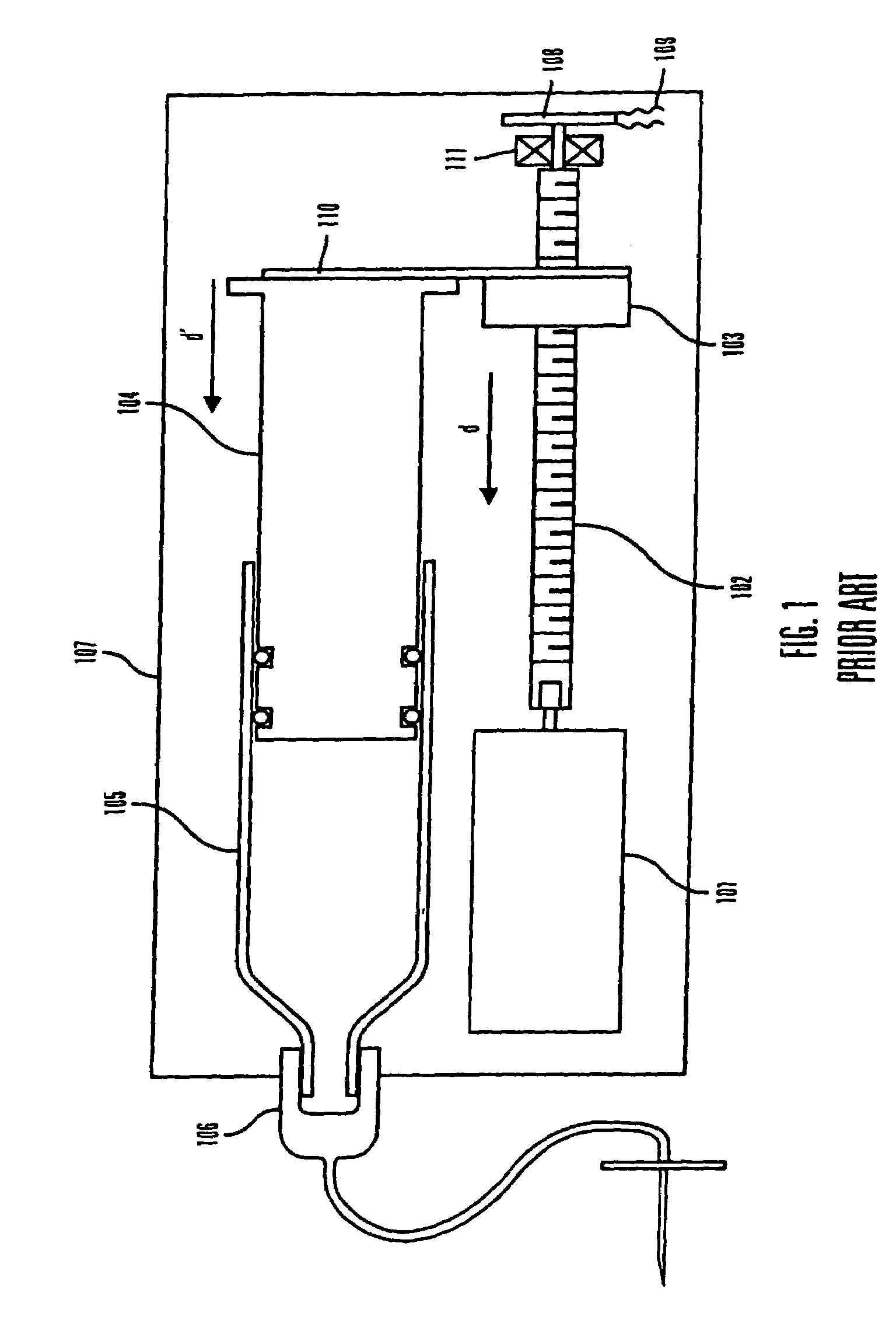
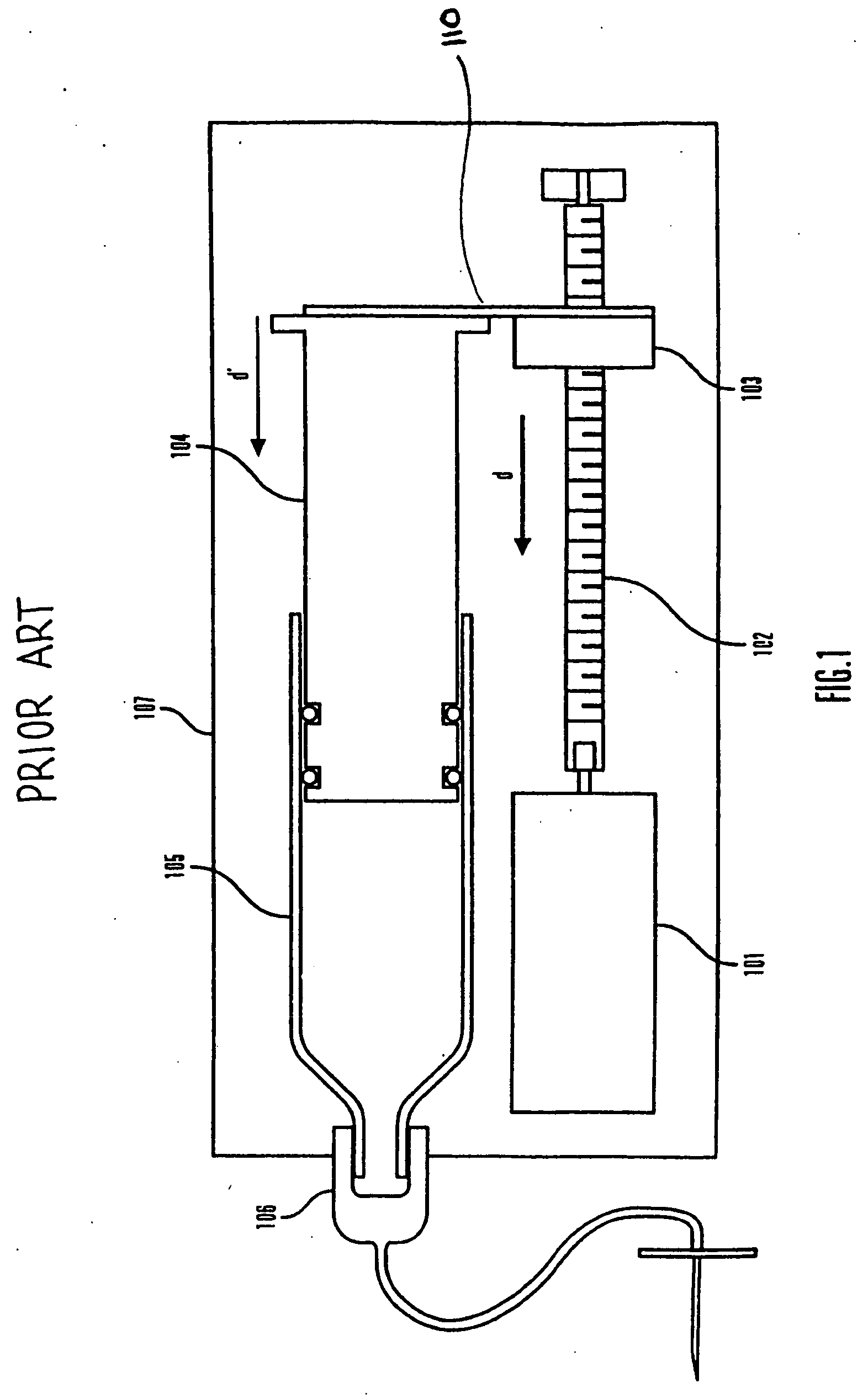
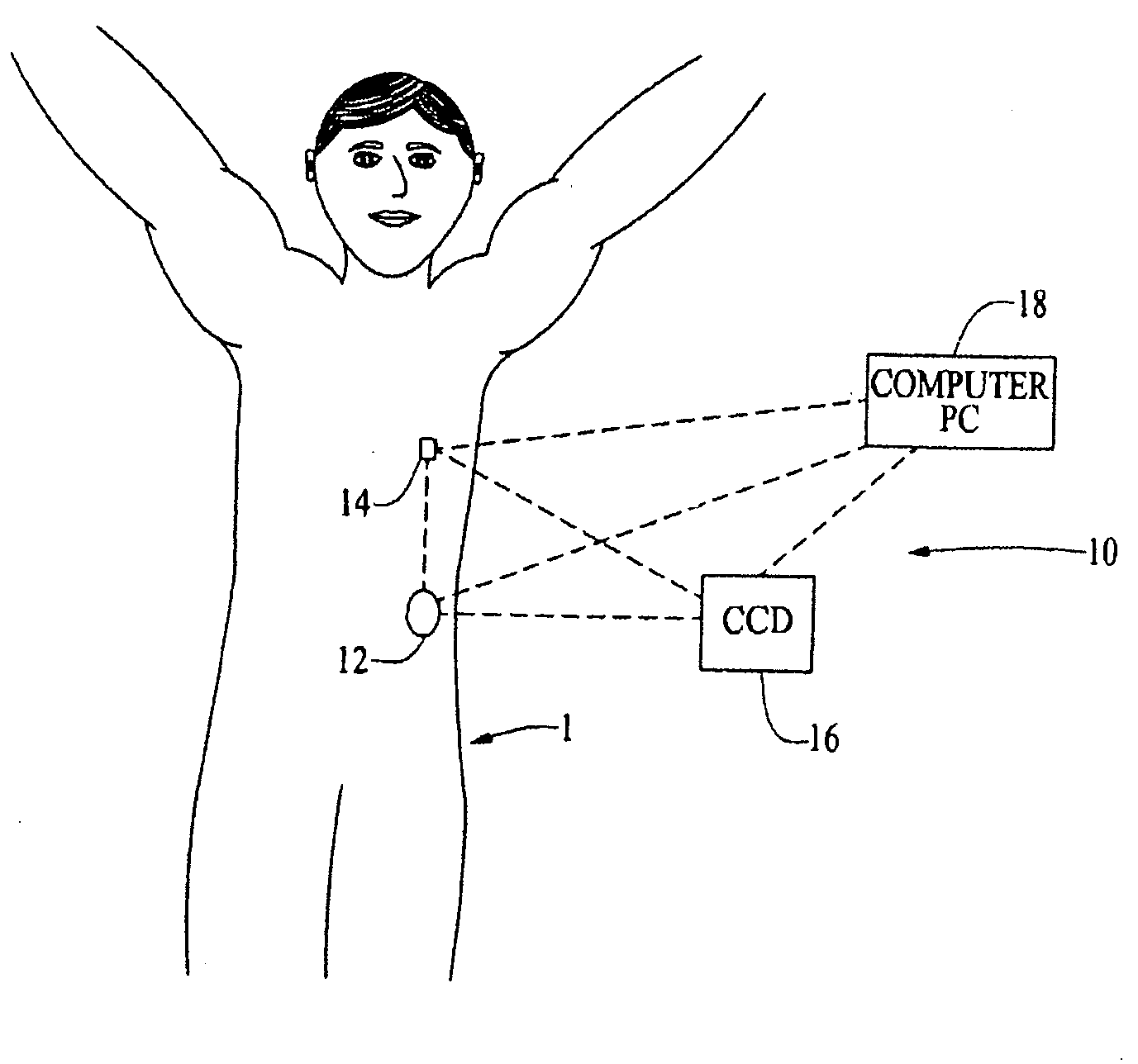
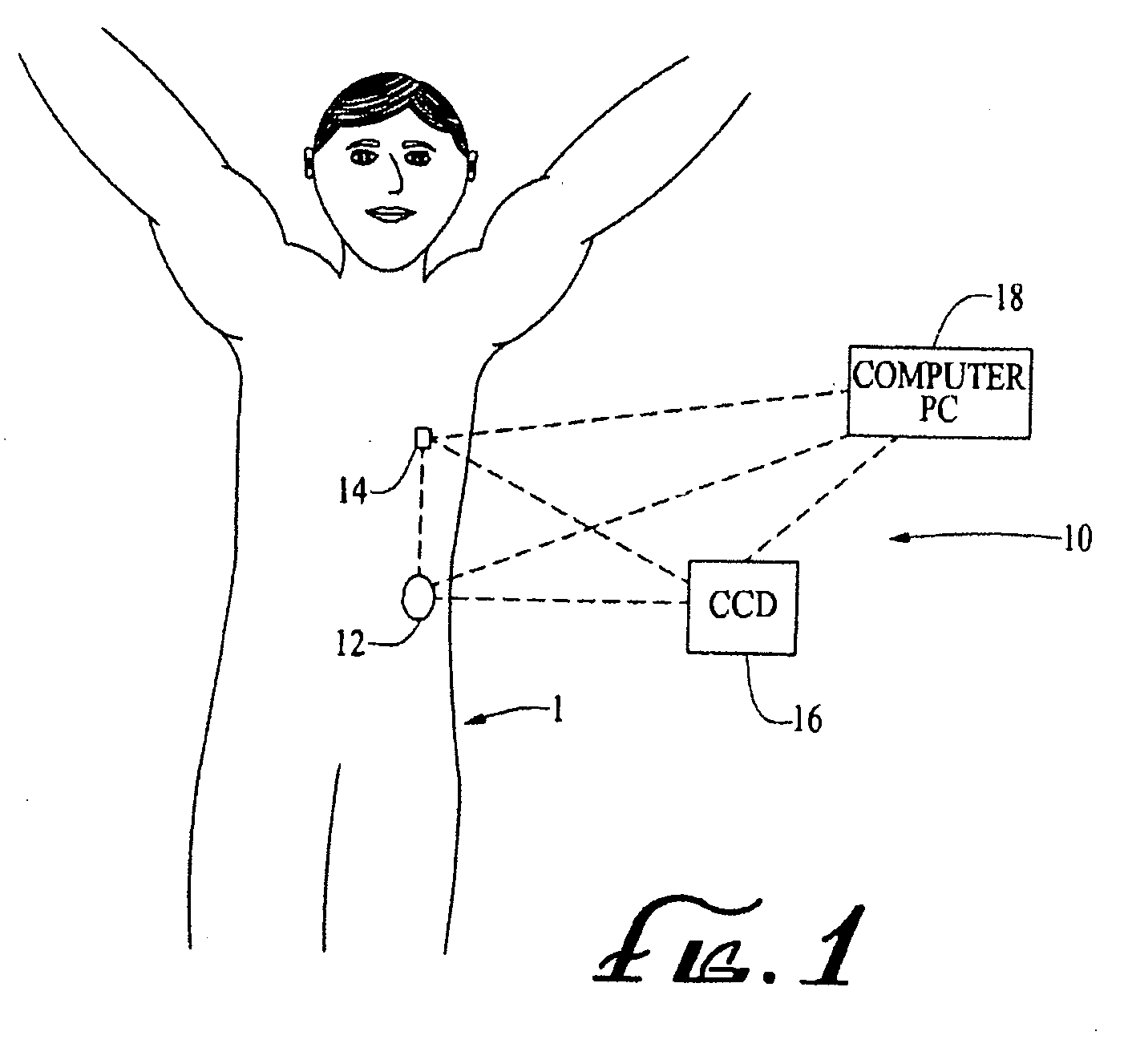
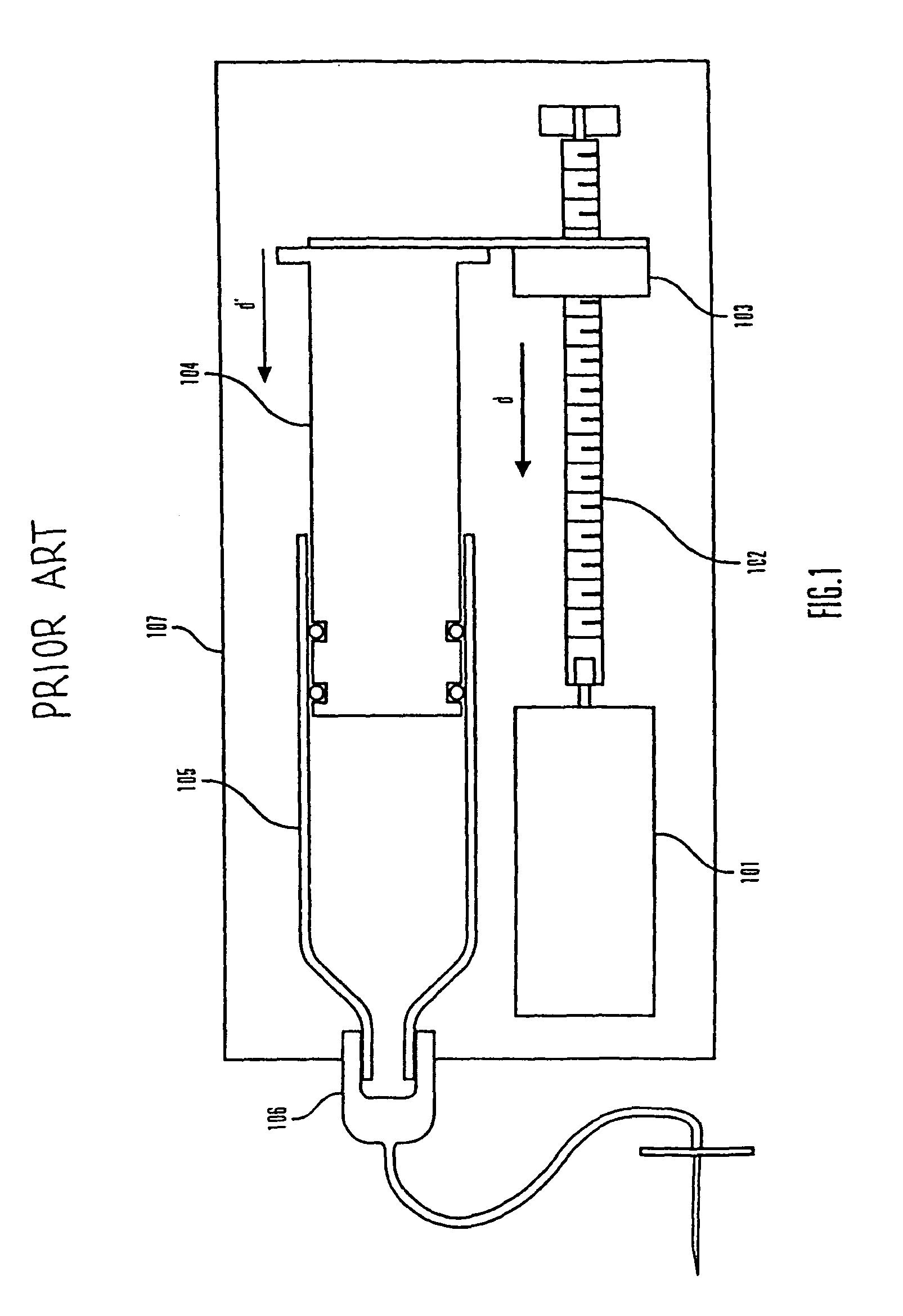
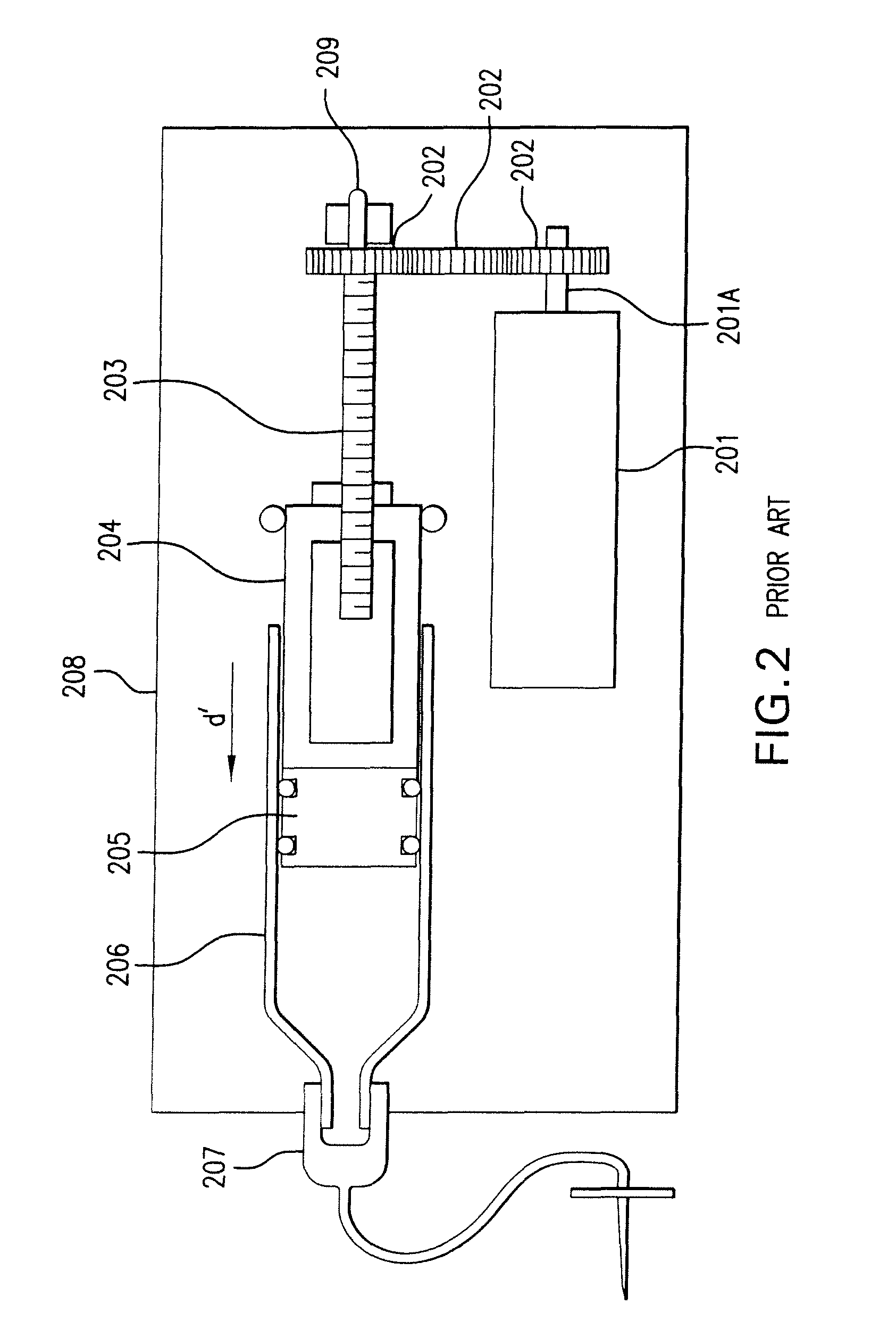
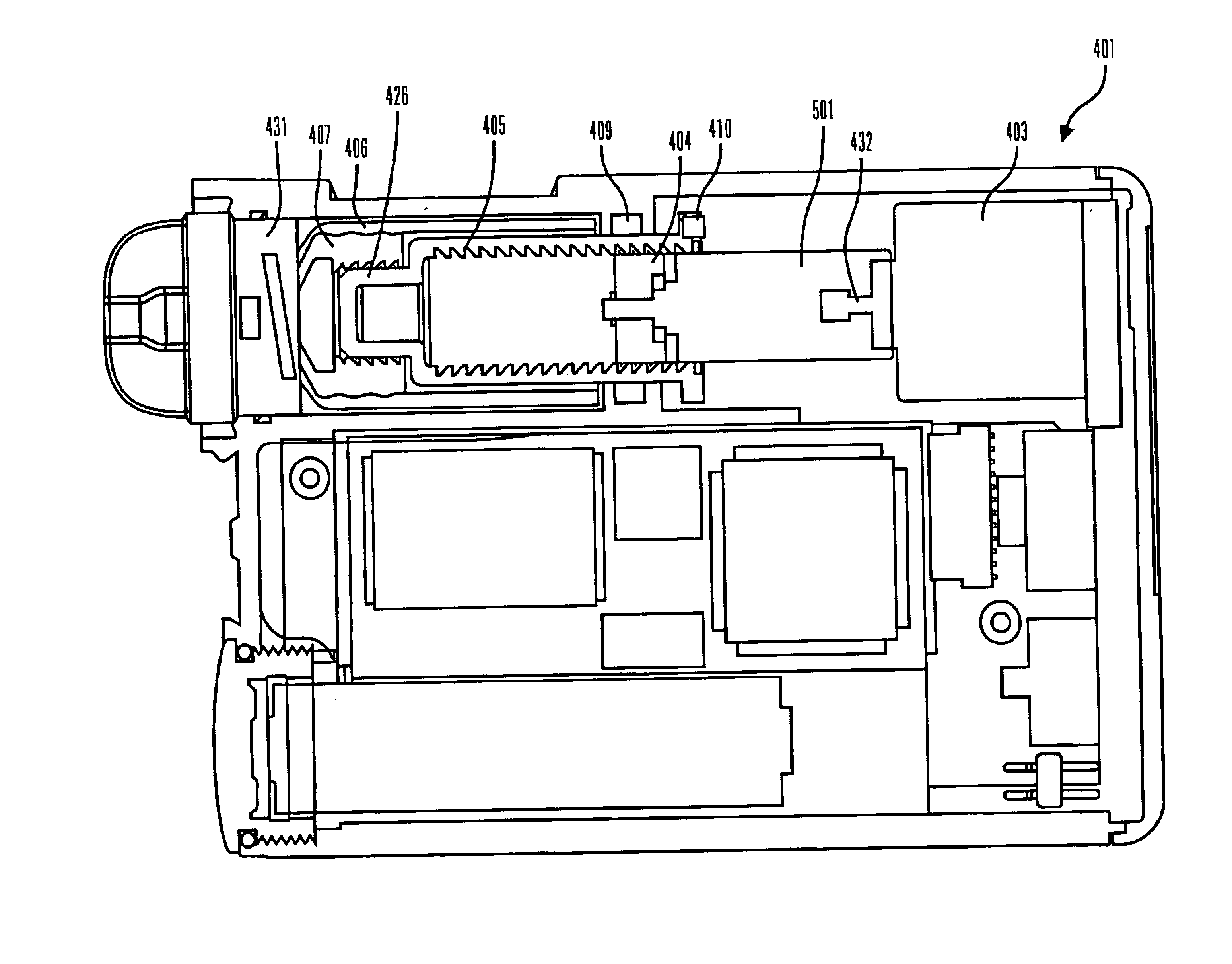
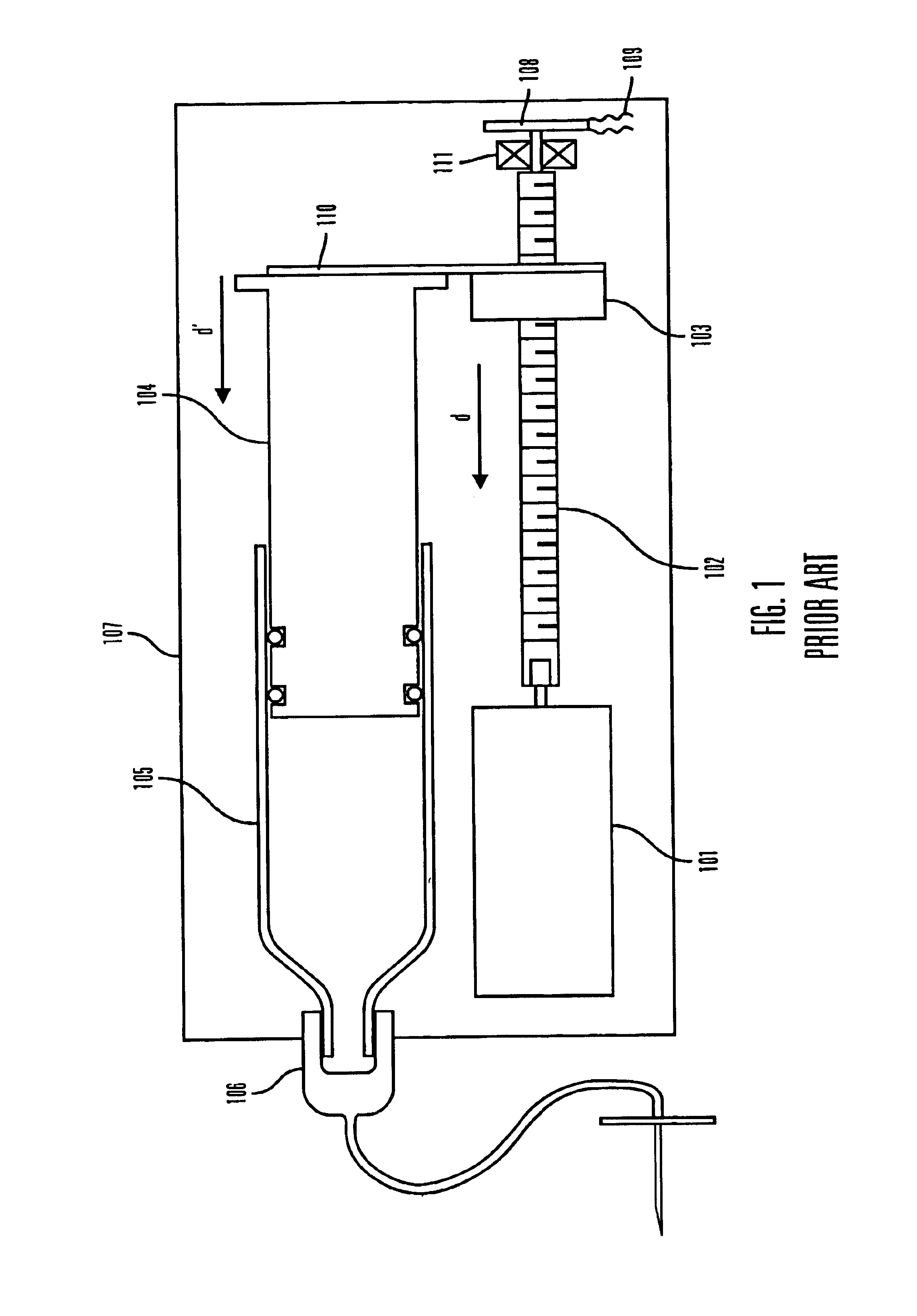
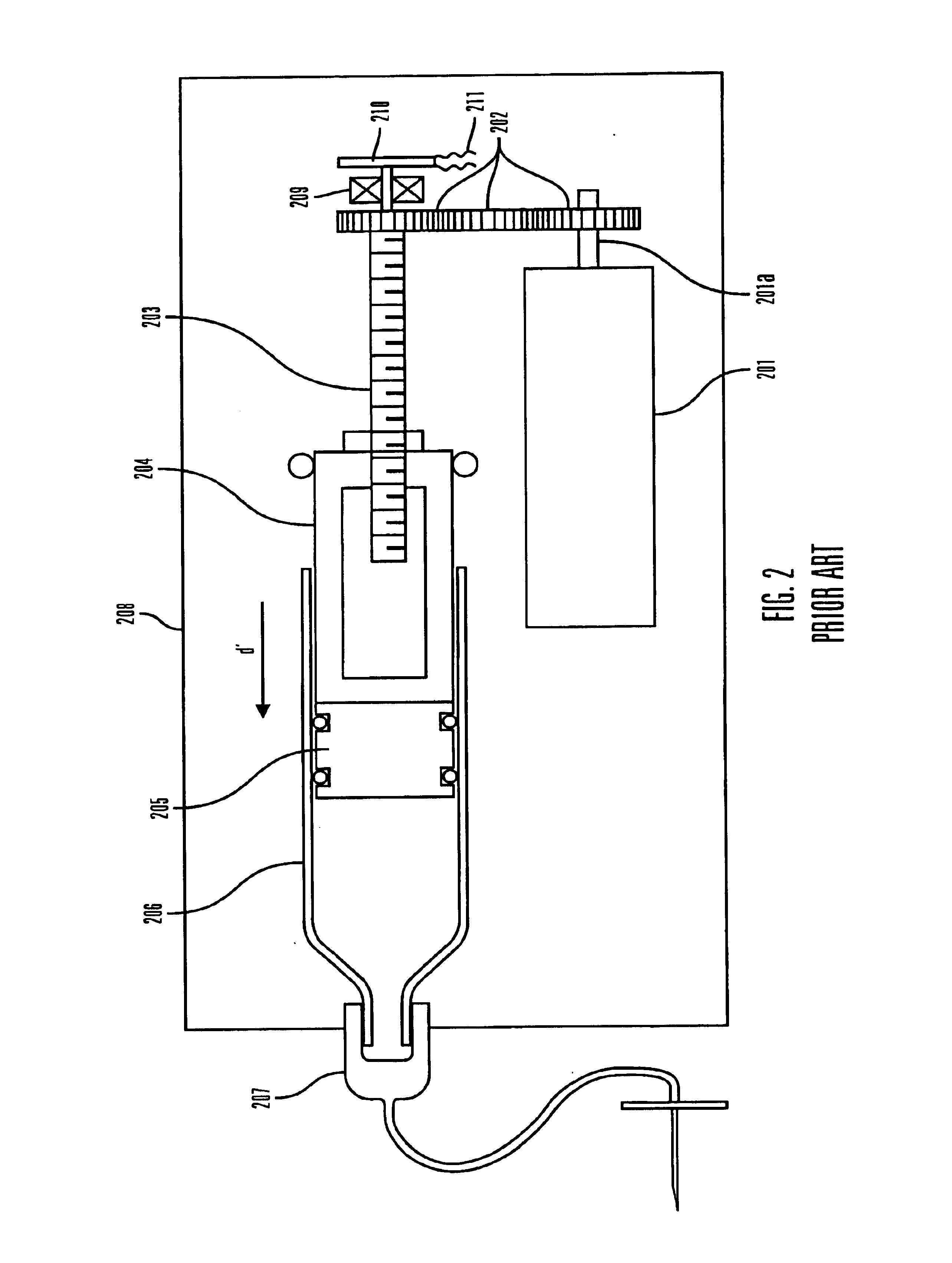
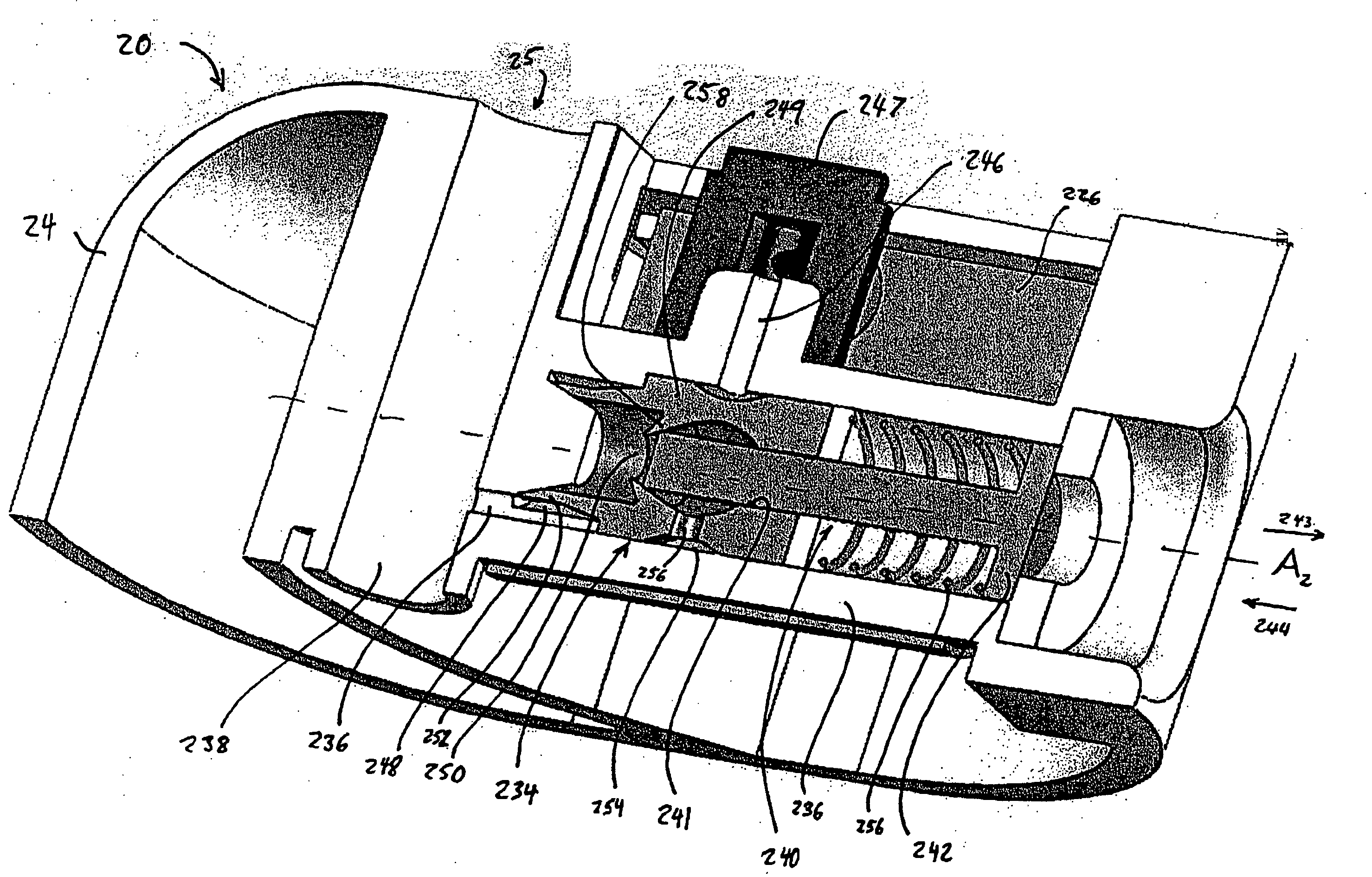
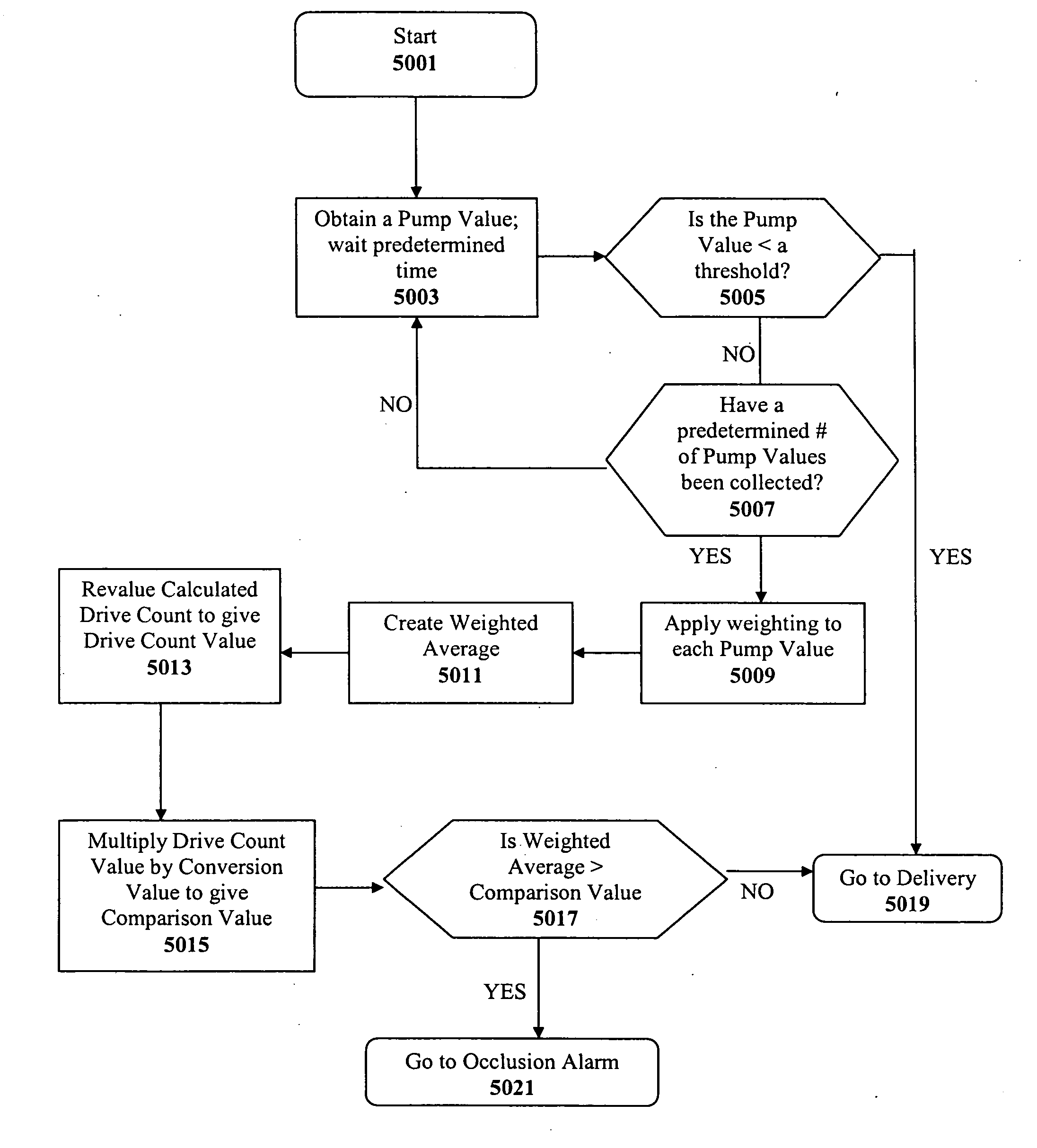
Methods and apparatuses for detecting occlusions in an ambulatory infusion pump
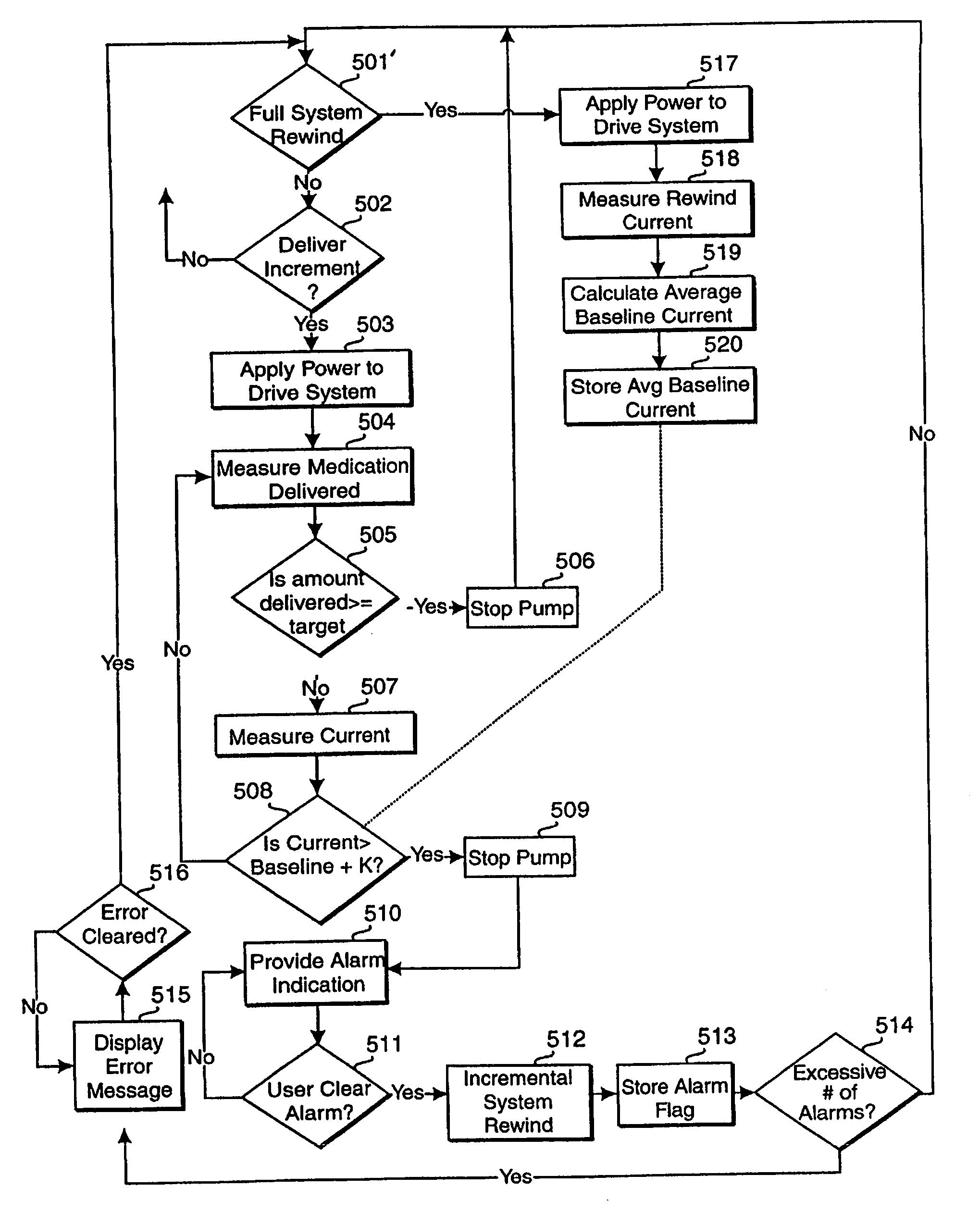
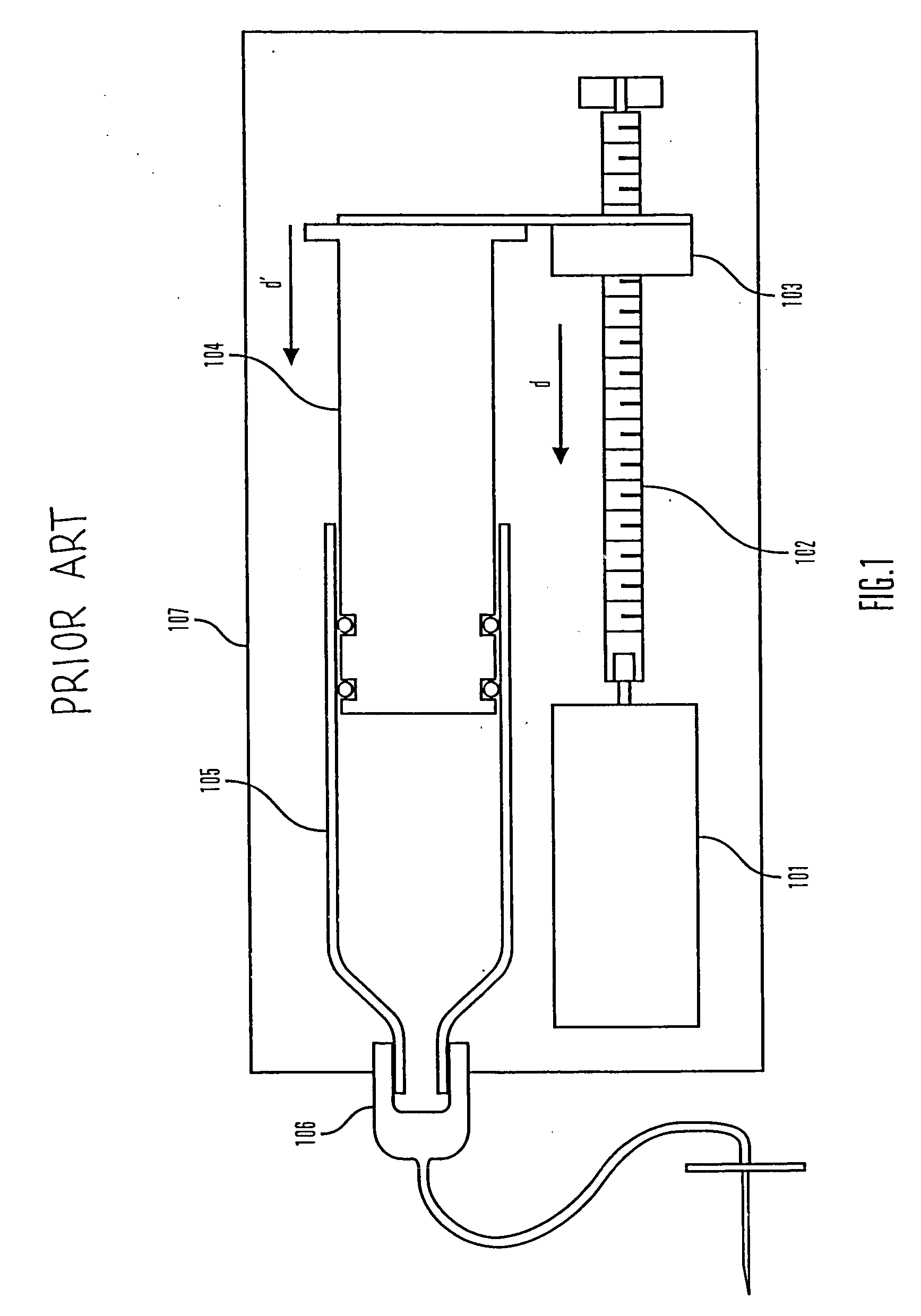
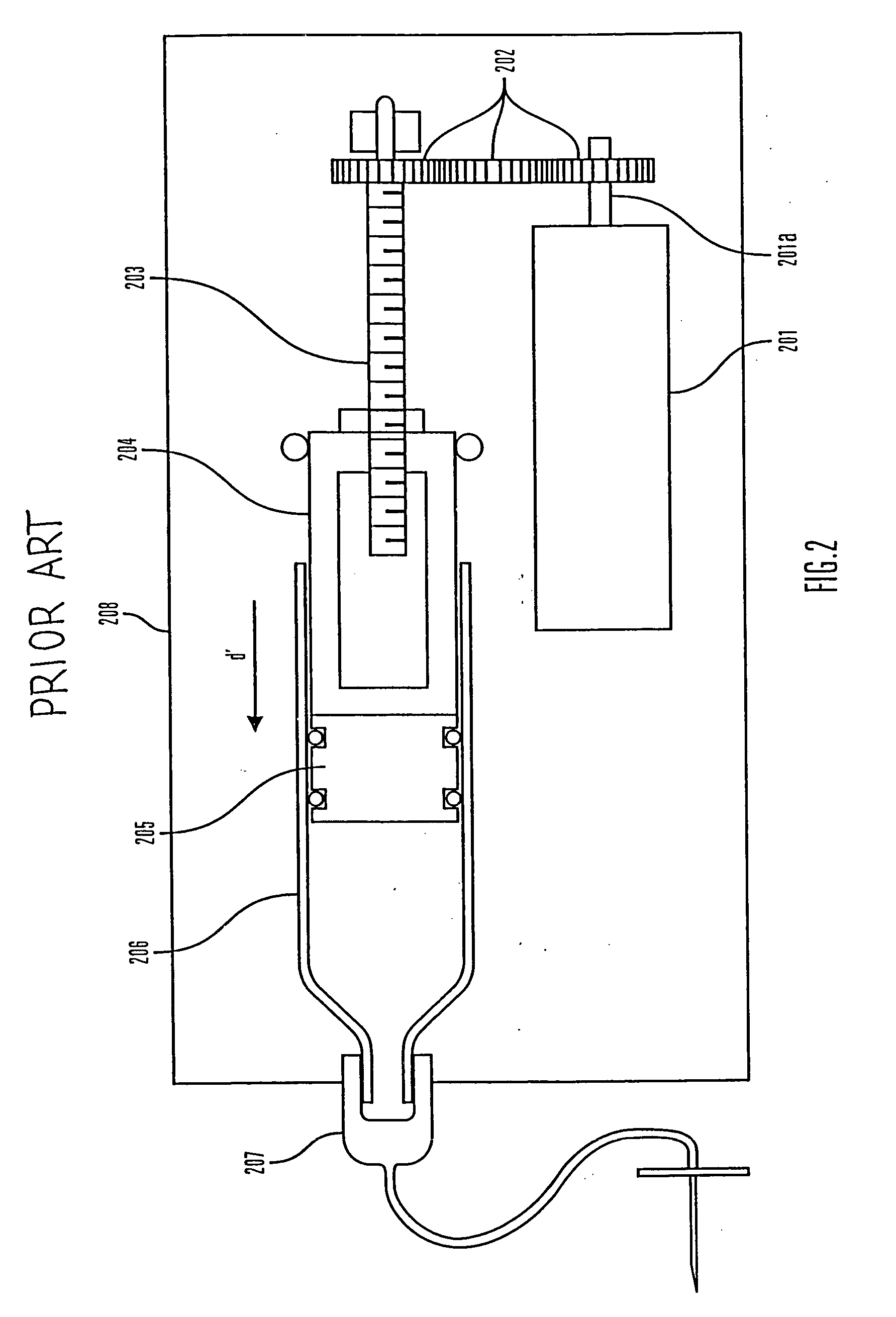
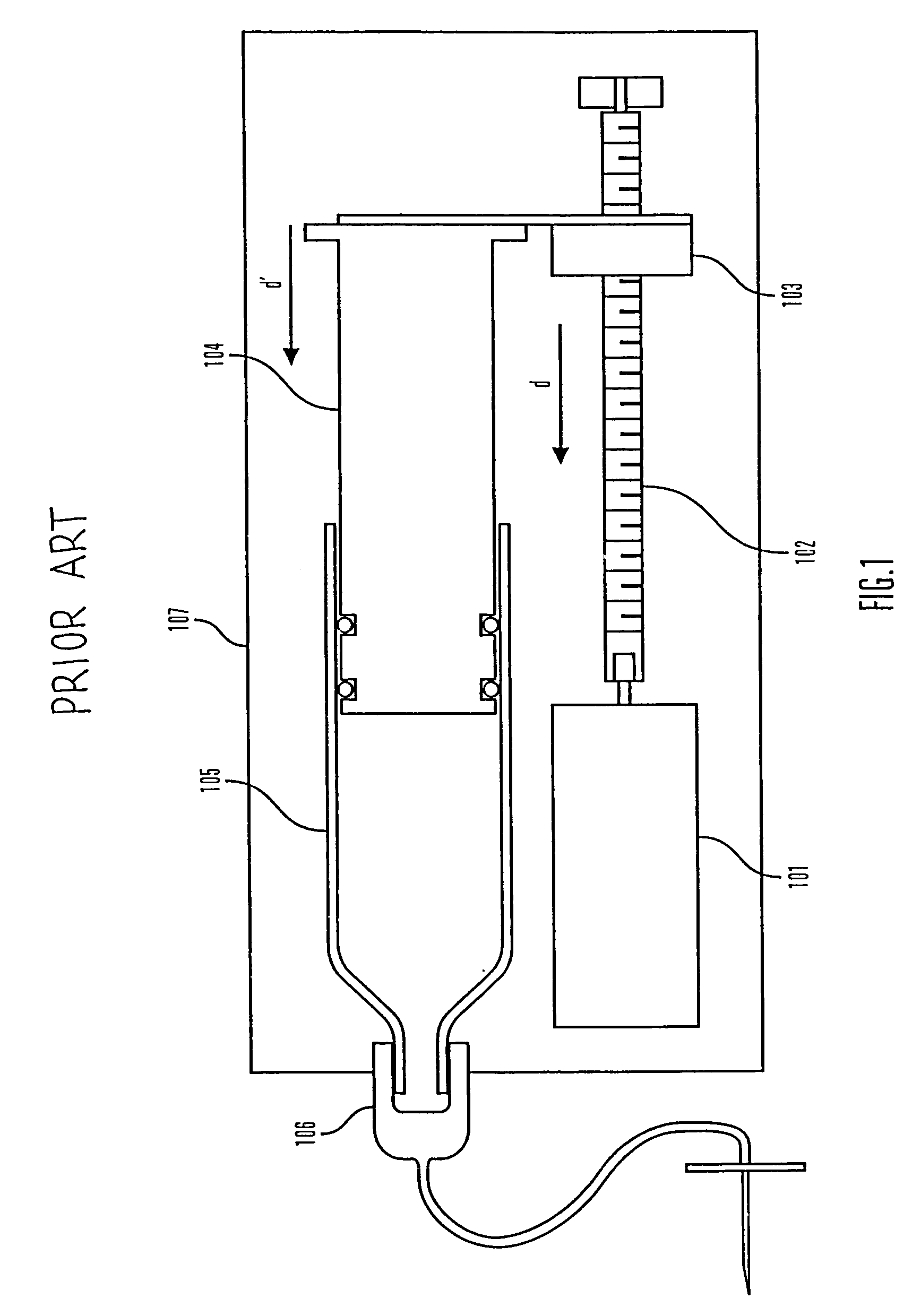
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. A method, system, and an article of manufacture for automatically detecting an occlusion in a medication infusion pump is provided. The electrical current to an infusion pump is measured. Based on measurements of one or more variables, the infusion pump detects whether there is an occlusion in the system. The methods of detecting occlusions may be dynamic.
Owner:MEDTRONIC MIMIMED INC
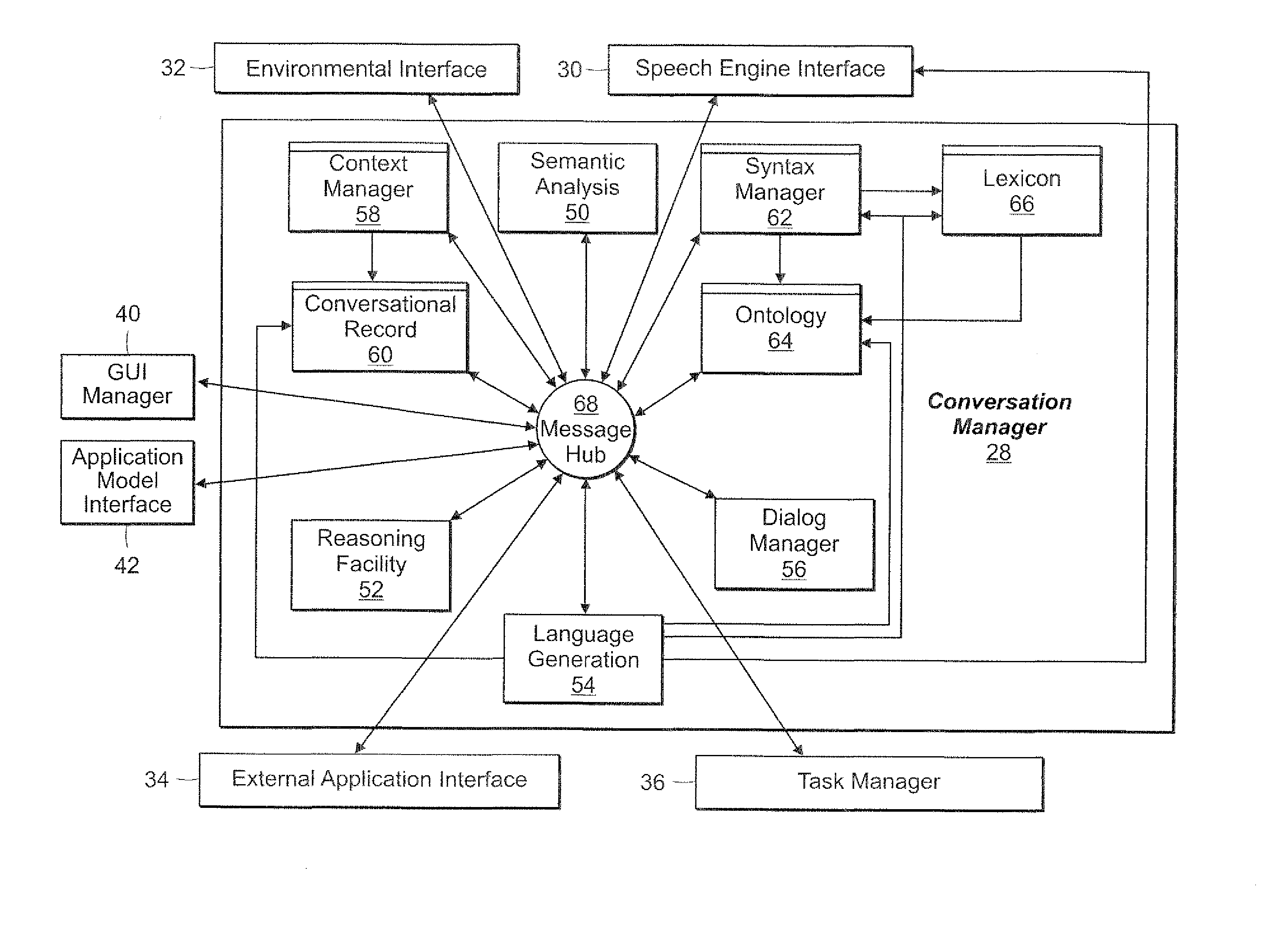
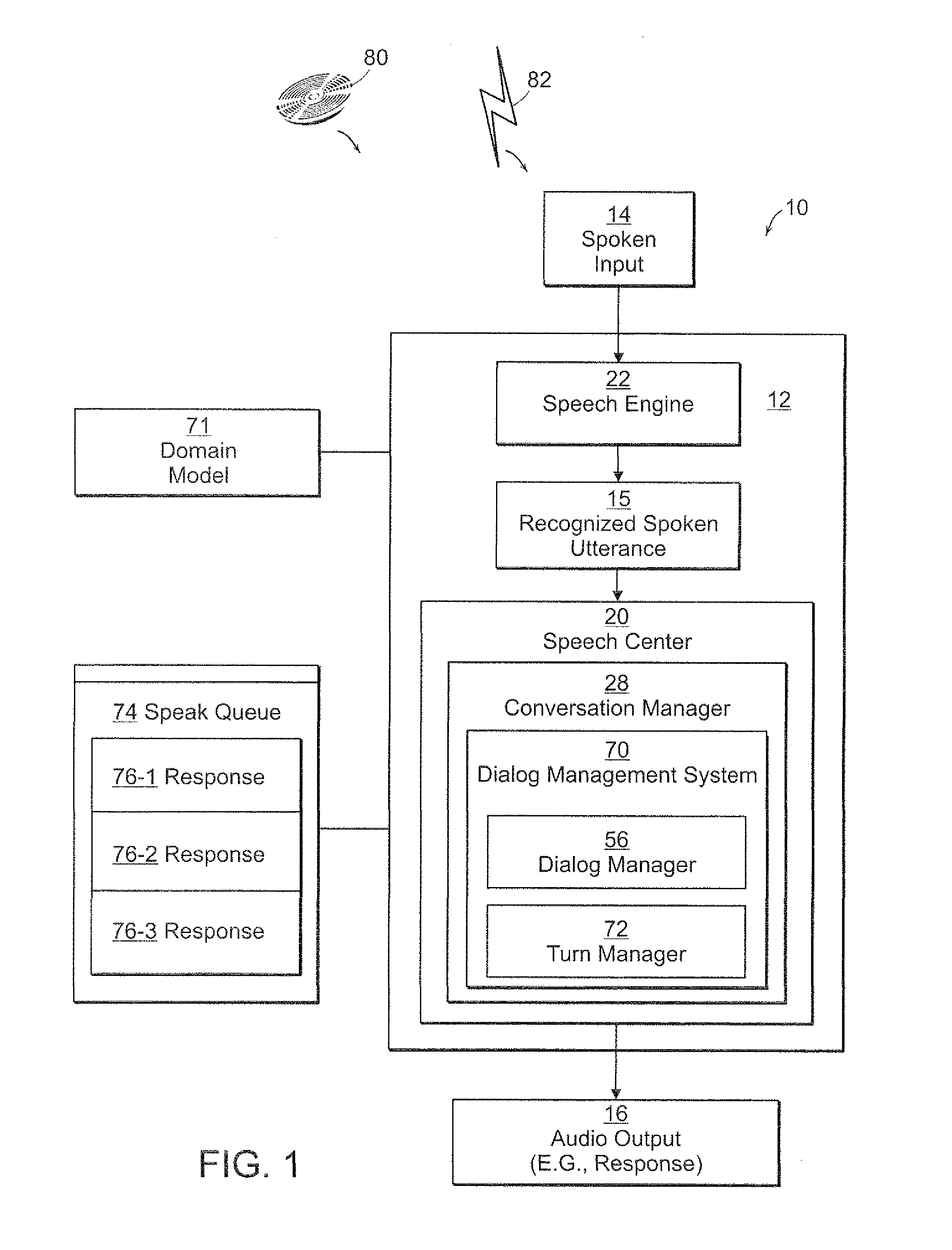
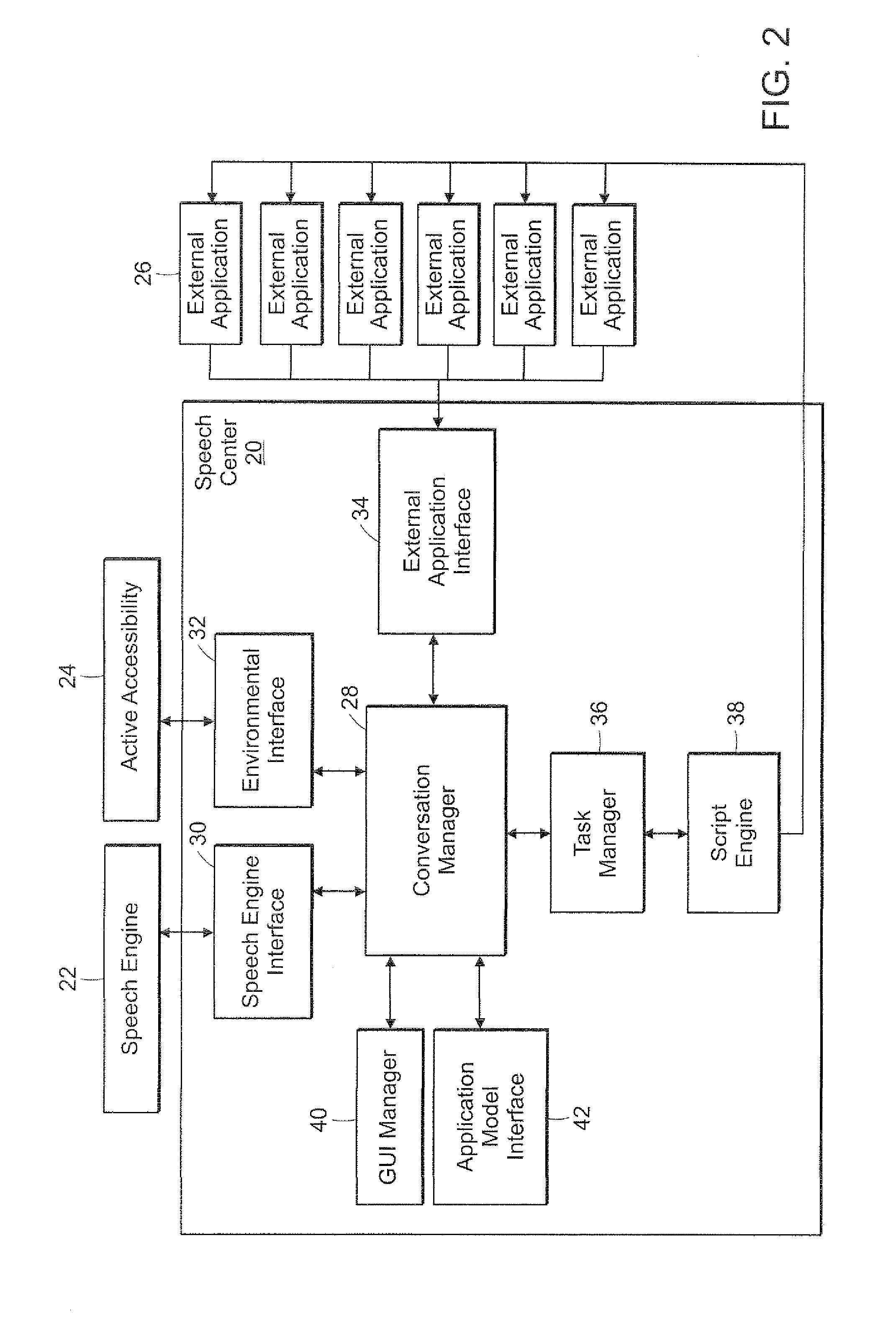
Method and Apparatus for Managing Dialog Management in a Computer Conversation
A dialog management system functions to manage the dialog between a user of a computer system and one or more speech enabled software applications. The user provides spoken input to a microphone connected to the computer system, and hears responses from one or more applications through a speaker connected to the computer system. The dialog management system includes a dialog manager, a turn manager, a speak queue, dialog contexts, and dialog state. The dialog manager provides top-level control of the dialog and stores responses based on the user's spoken input in a speak queue for later output to the user. The turn manager controls delivery of the responses to the user based on the dialog context and the dialog state, to provide a polite asynchronous dialog with the user that enables the user to be in control of the dialog. The dialog context provides information about each dialog. The dialog state provides information about whose turn it is (computer or user) to speak.
Owner:NUANCE COMM INC
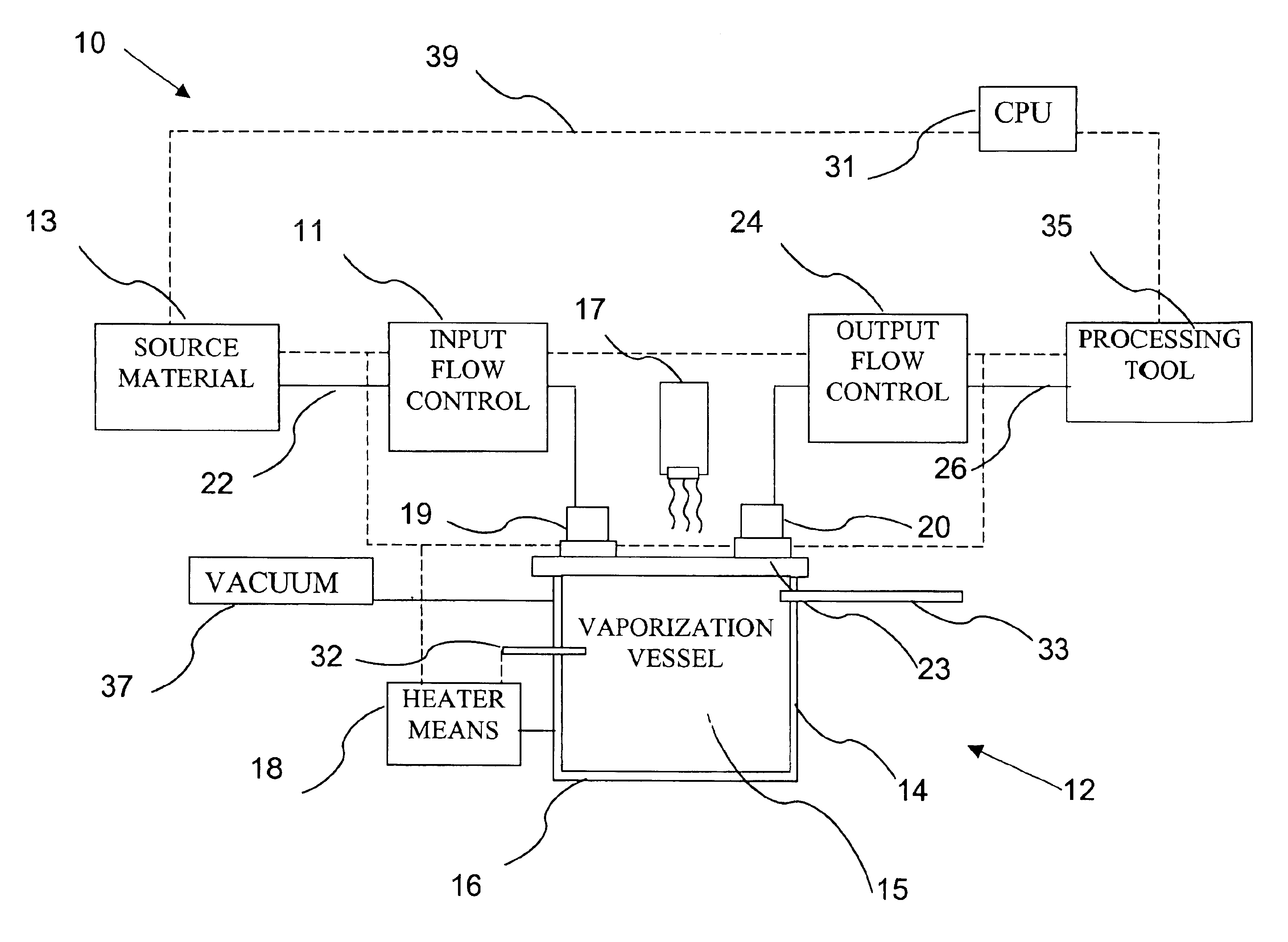
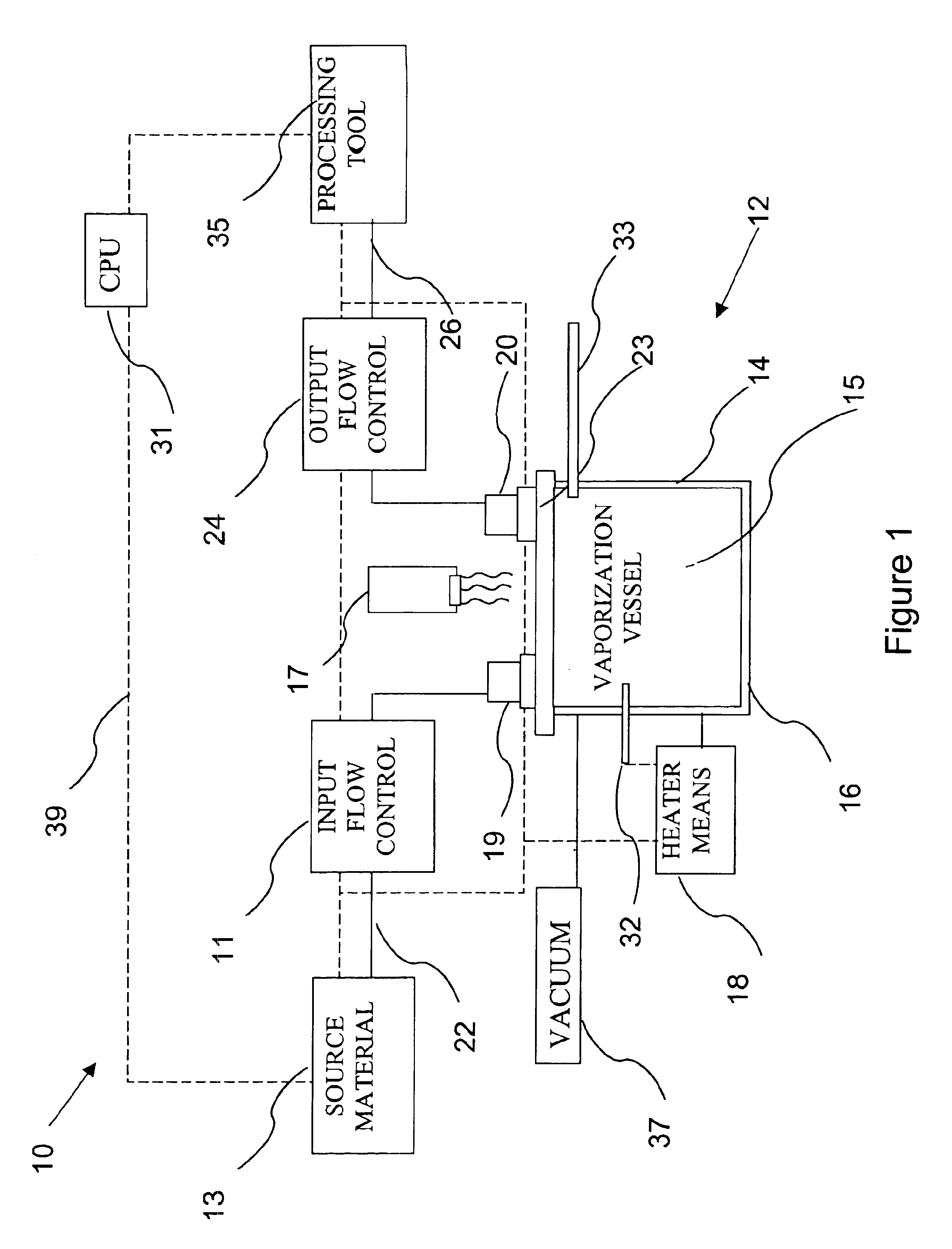
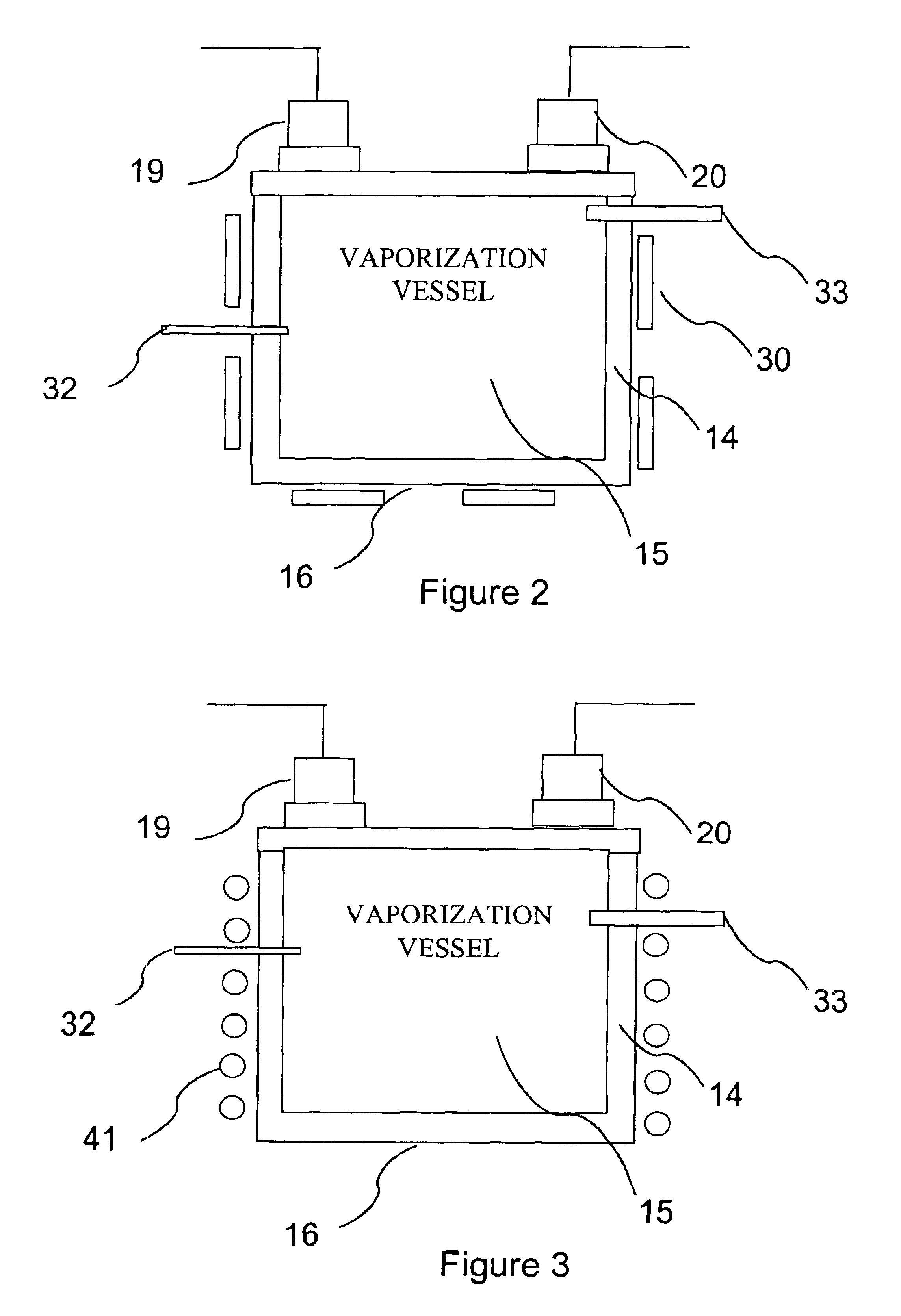
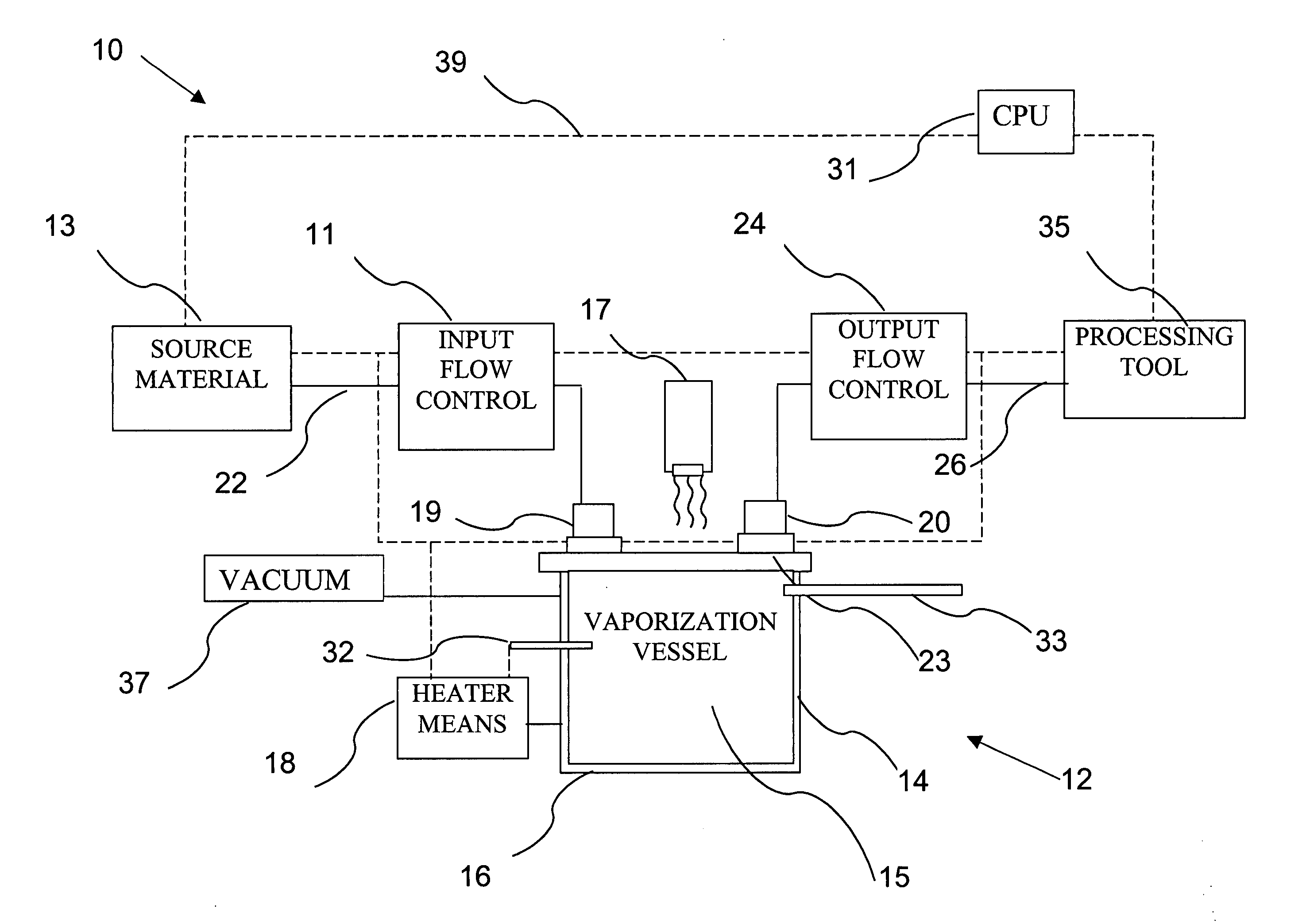
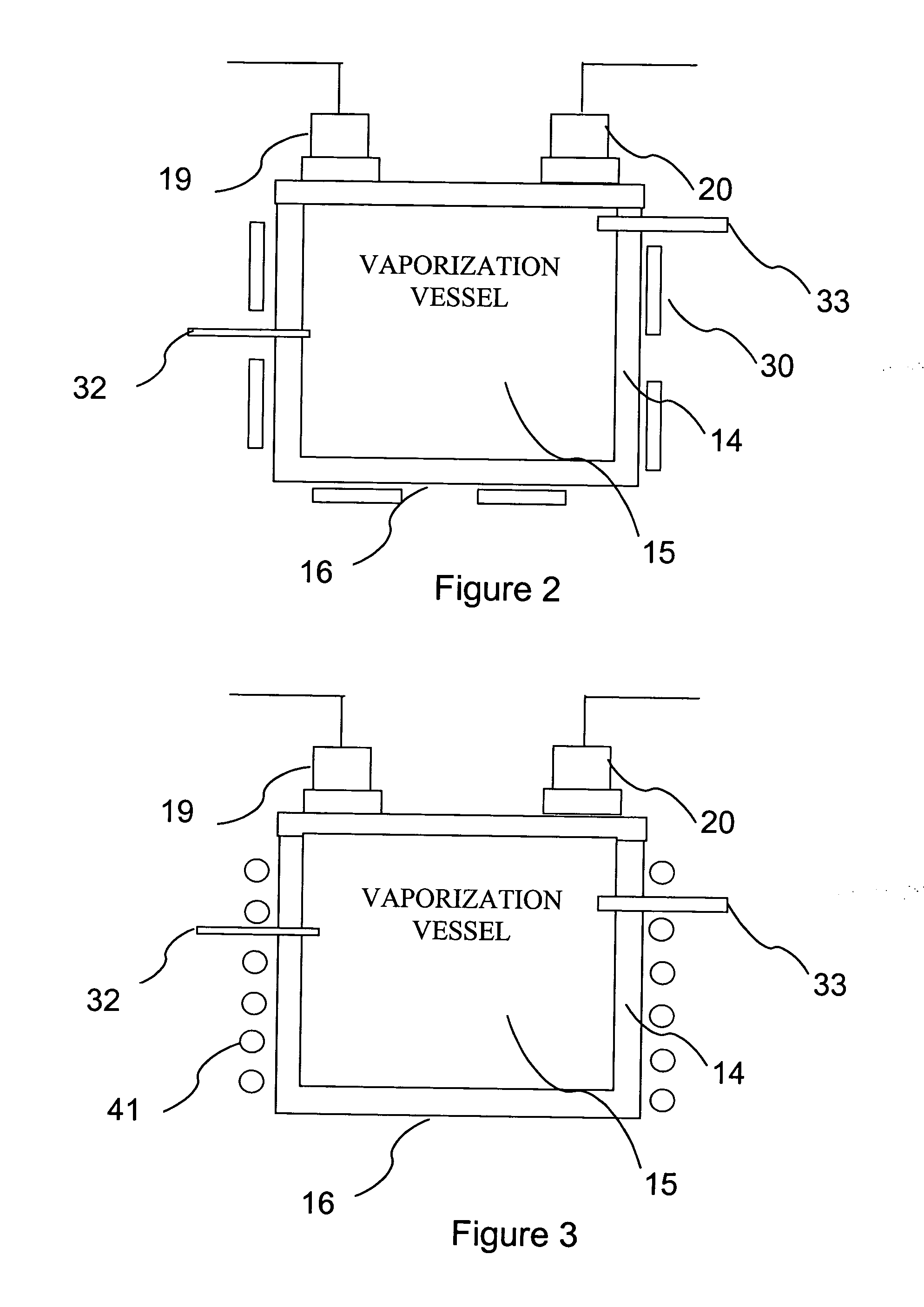
Delivery systems for efficient vaporization of precursor source material
InactiveUS6909839B2Precise deliveryVacuum evaporation coatingSputtering coatingControl flowSource material
The present invention relates to a delivery system for vaporizing and delivering vaporized solid and liquid precursor materials at a controlled rate having particular utility for semiconductor manufacturing applications. The system includes a vaporization vessel, a processing tool and a connecting vapor line therebetween, where the system further includes an input flow controller and / or an output flow controller to provide a controlled delivery of a vaporizable source material to the vaporization vessel and a controlled flow rate of vaporized source material to the processing tool.
Owner:ENTEGRIS INC
Biocompatible crosslinked polymers
InactiveUS7009034B2Improve performanceImprove visibilityUltrasonic/sonic/infrasonic diagnosticsPowder deliveryWound dressingPost operative
Biocompatible crosslinked polymers, and methods for their preparation and use, are disclosed in which the biocompatible crosslinked polymers are formed from water soluble precursors having electrophilic and nucleophilic functional groups capable of reacting and crosslinking in situ. Methods for making the resulting biocompatible crosslinked polymers biodegradable or not are provided, as are methods for controlling the rate of degradation. The crosslinking reactions may be carried out in situ on organs or tissues or outside the body. Applications for such biocompatible crosslinked polymers and their precursors include controlled delivery of drugs, prevention of post-operative adhesions, coating of medical devices such as vascular grafts, wound dressings and surgical sealants. Visualization agents may be included with the crosslinked polymers.
Owner:INCEPT LLC
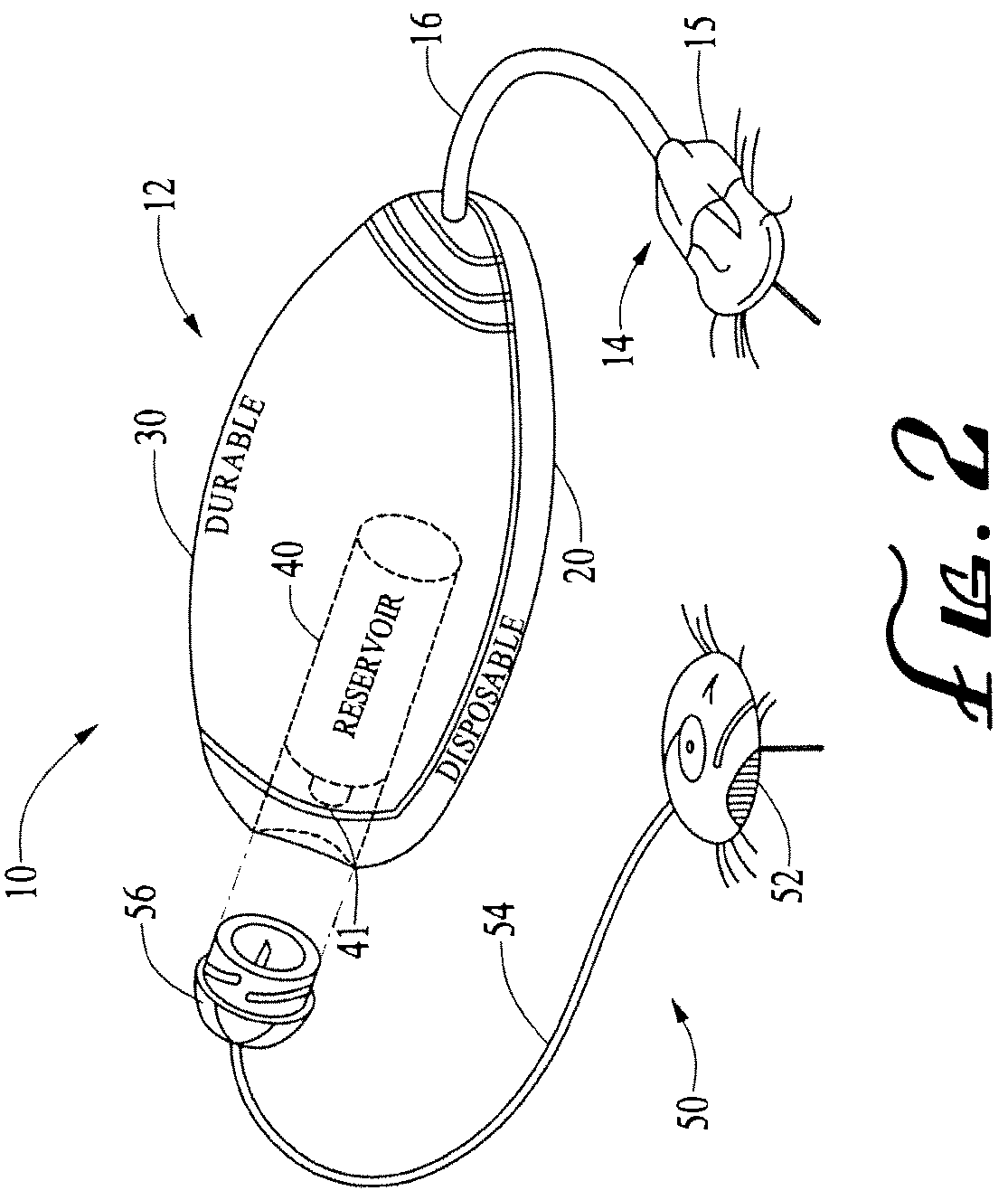
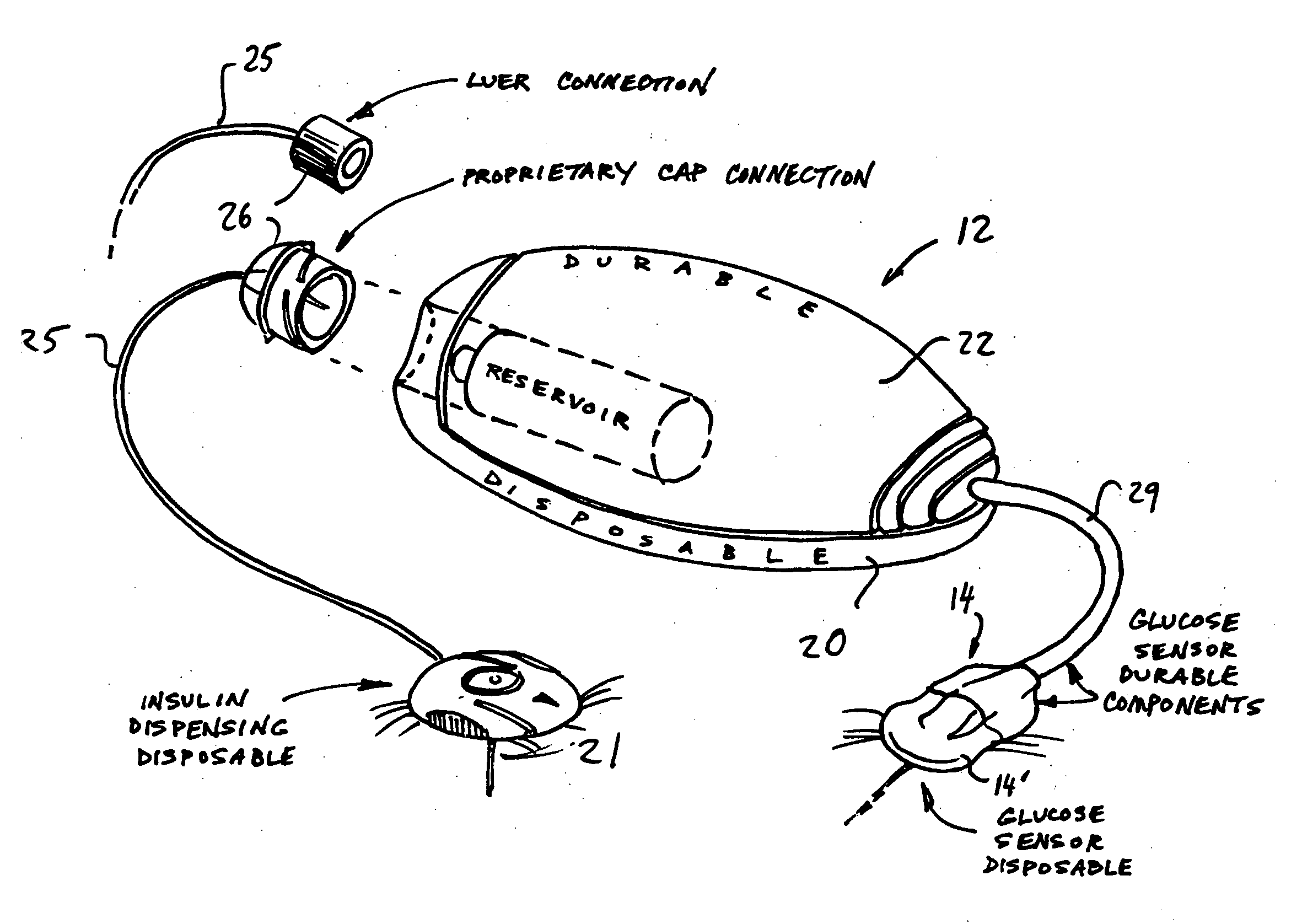
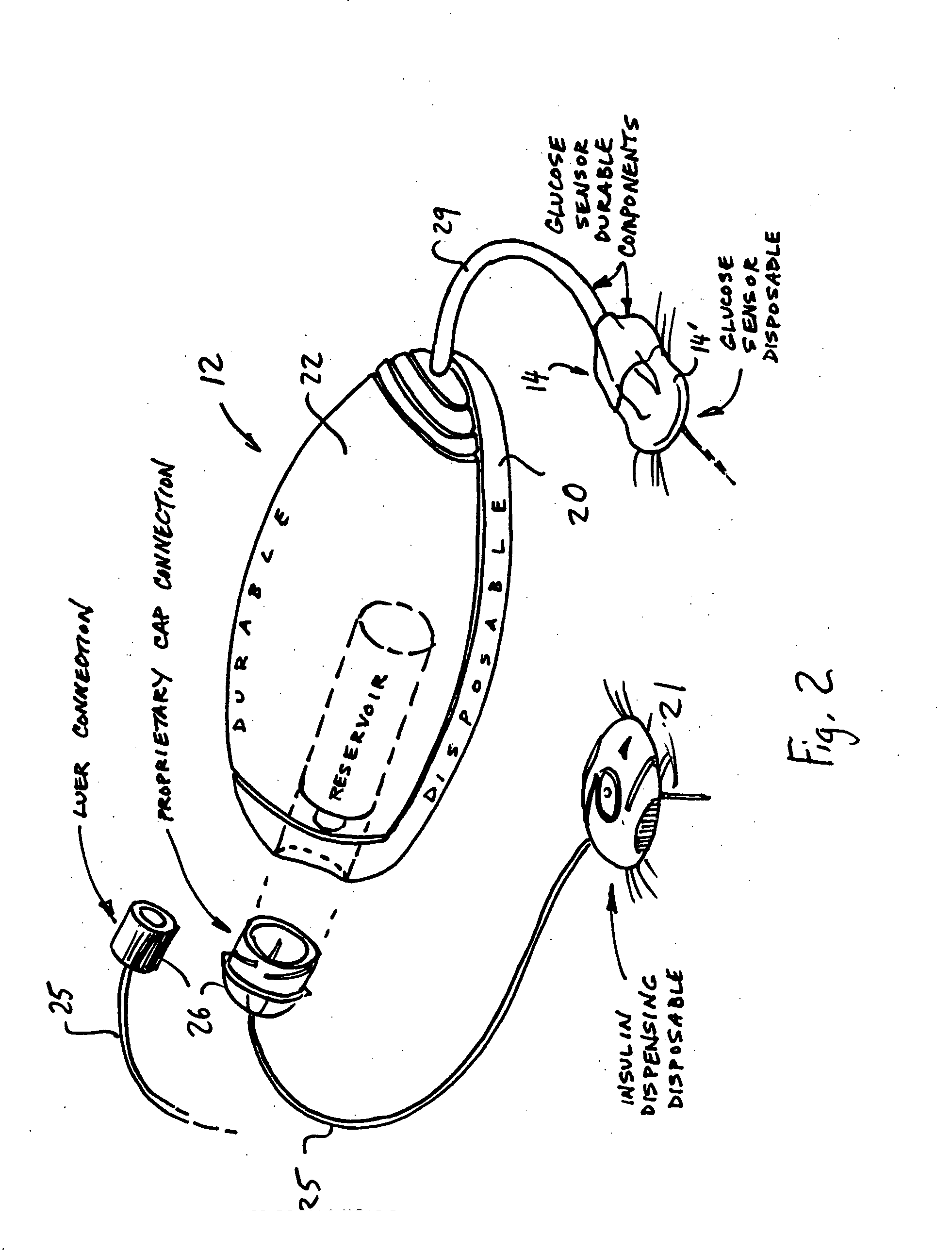
Systems and methods allowing for reservoir filling and infusion medium delivery
ActiveUS7828764B2Reduce internal volumeIncrease the internal volumePharmaceutical containersMedical devicesSurgeryMechanical engineering
A system includes a durable portion with a durable housing and a separable disposable portion with a disposable housing that selectively engage with and disengage from each other. The disposable housing secures to a patient and may be disposed of after it has been in use for a prescribed period. Components that normally come into contact with a patient or with an infusion medium may be part of the disposable portion to allow for disposal after a prescribed use. A reservoir for holding the infusion medium may be part of the disposable portion, and may be supported by the disposable housing. The durable portion may include other components such as electronics for controlling delivery of the infusion medium from the reservoir, and a drive device including a motor and drive linkage.
Owner:MEDTRONIC MIMIMED INC
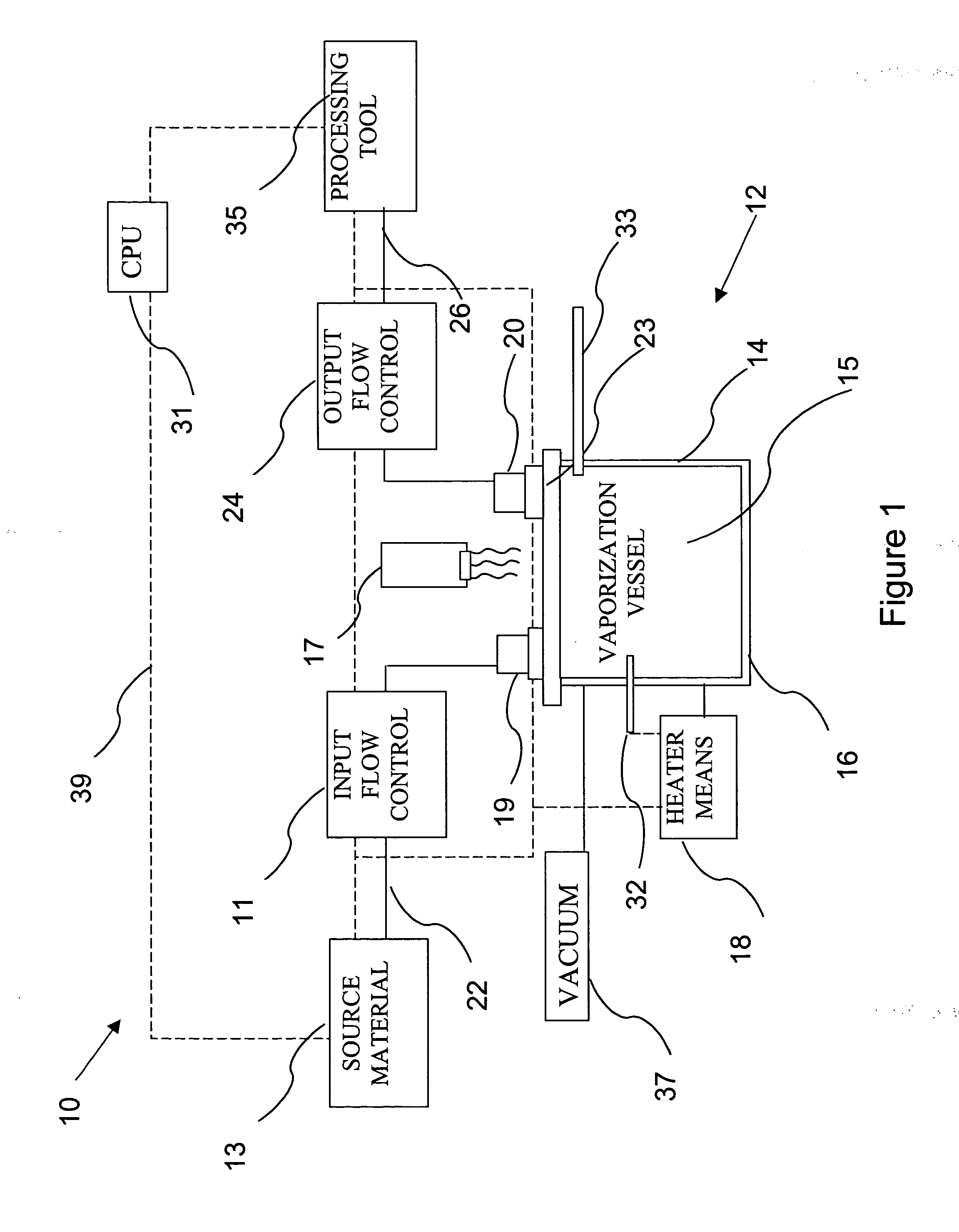
Delivery systems for efficient vaporization of precursor source material
InactiveUS20050019026A1Precise deliveryVacuum evaporation coatingSemiconductor/solid-state device manufacturingControl flowSource material
The present invention relates to a delivery system for vaporizing and delivering vaporized solid and liquid precursor materials at a controlled rate having particular utility for semiconductor manufacturing applications. The system includes a vaporization vessel, a processing tool and a connecting vapor line therebetween, where the system further includes an input flow controller and / or an output flow controller to provide a controlled delivery of a vaporizable source material to the vaporization vessel and a controlled flow rate of vaporized source material to the processing tool.
Owner:ENTEGRIS INC
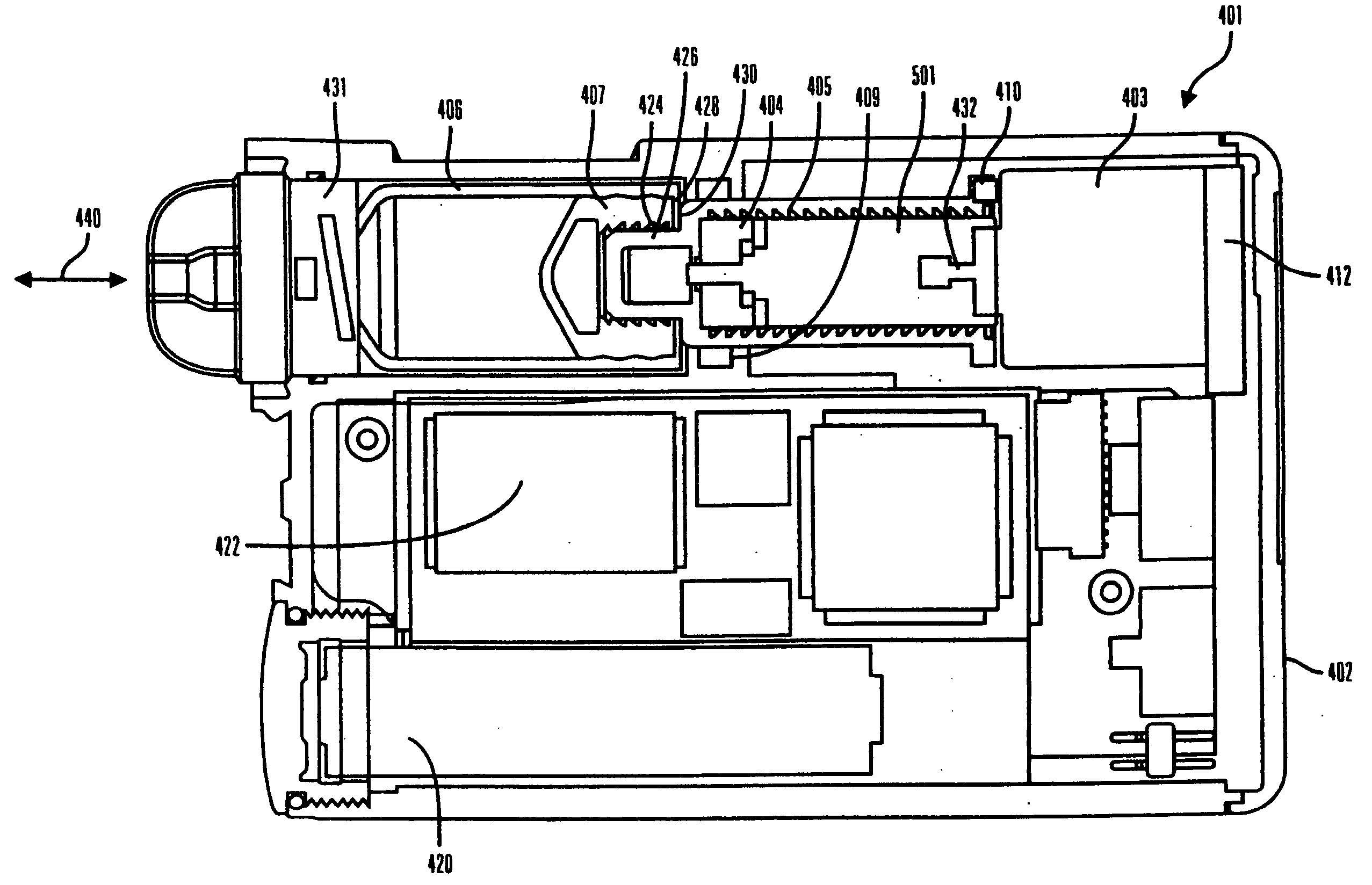
Infusion device and method with disposable portion
ActiveUS20060264894A1Automatic syringesPharmaceutical delivery mechanismEngineeringBiomedical engineering
A delivery device includes a durable housing portion and a separable disposable portion that selectively engage and disengage from each other. The disposable housing portion secures to the patient and may be disposed of after it has been in use for a prescribed period. Components that normally come into contact with a patient or with infusion media are supported by the disposable housing portion for disposal after the prescribed use, while the durable housing portion supports other components such as electronics for controlling delivery of infusion media from the reservoir and a drive device and drive linkage.
Owner:MEDTRONIC MIMIMED INC
Laser ablation process and apparatus
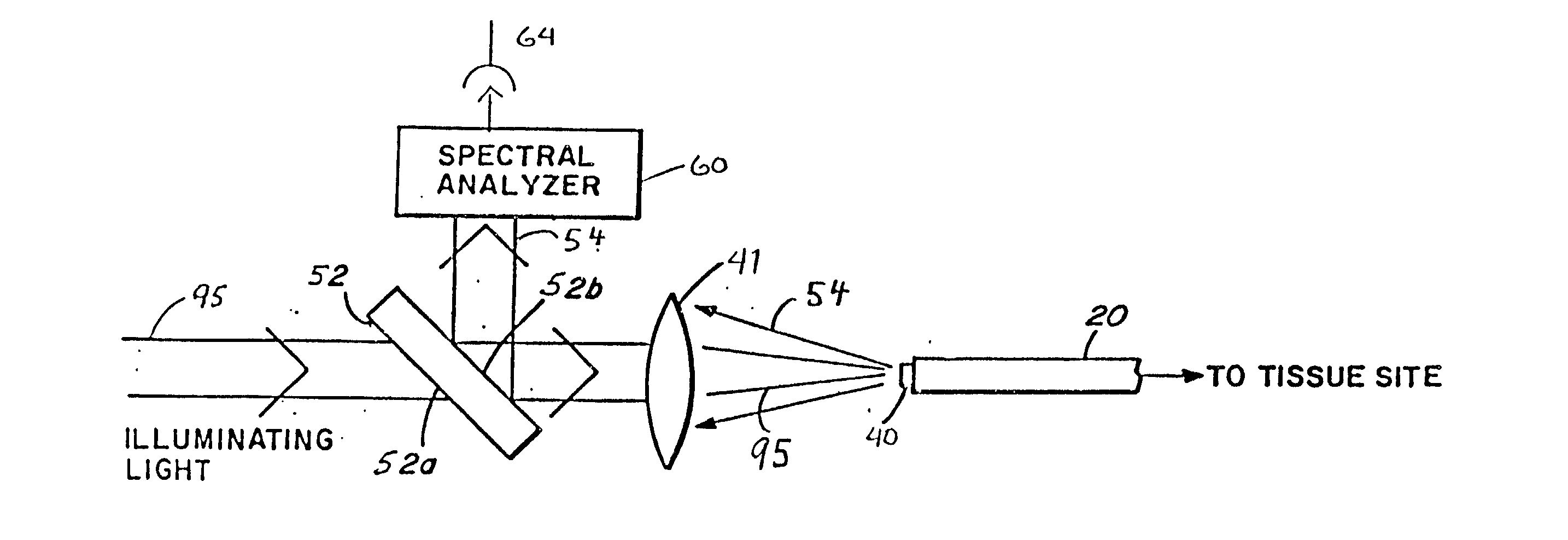
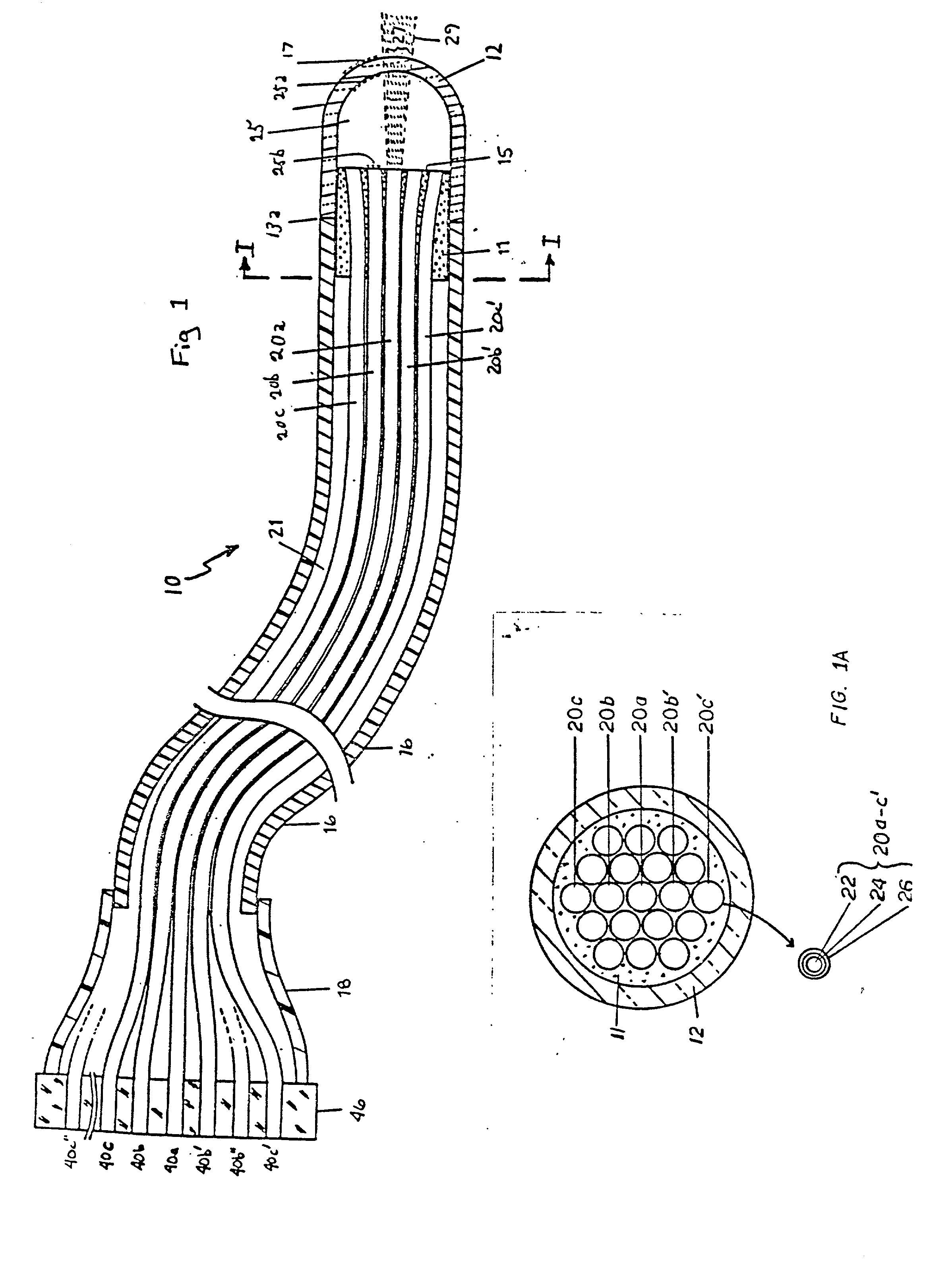
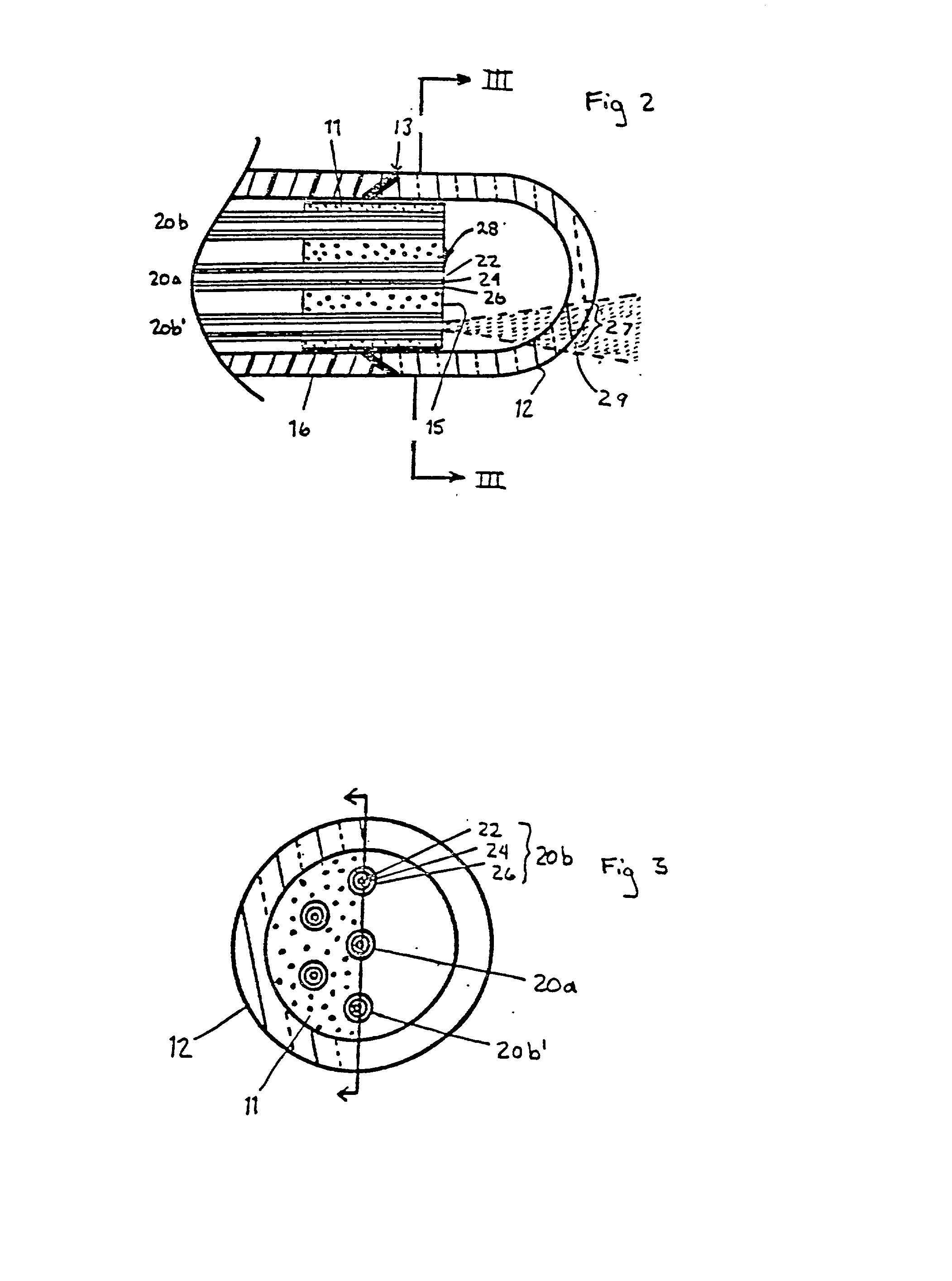
InactiveUS20020045811A1Reduce Fresnel reflectionMaximize transmitted lightControlling energy of instrumentDiagnostics using spectroscopyFiberLaser light
A laser catheter is disclosed wherein optical fibers carrying laser light are mounted in a catheter for insertion into an artery to provide controlled delivery of a laser beam for percutaneous intravascular laser treatment of atherosclerotic disease. A transparent protective shield is provided at the distal end of the catheter for mechanically diplacing intravascular blood and protecting the fibers from the intravascular contents, as well as protecting the patient in the event of failure of the fiber optics. Multiple optical fibers allow the selection of tissue that is to be removed. A computer controlled system automatically aligns fibers with the laser and controls exposure time. Spectroscopic diagnostics determine what tissue is to be removed.
Owner:KITTRELL CARTER +2
Ablation of rectal and other internal body structures
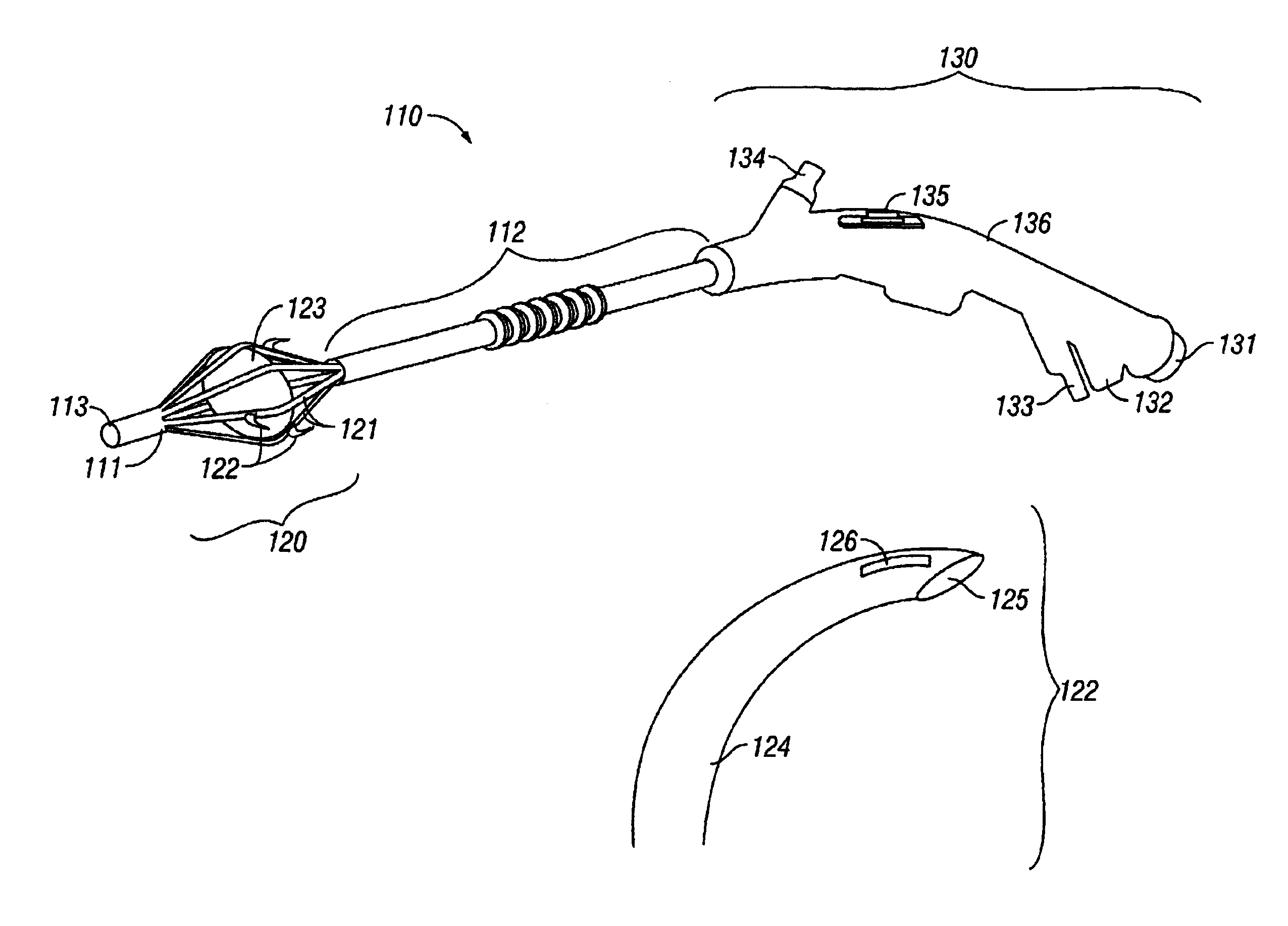
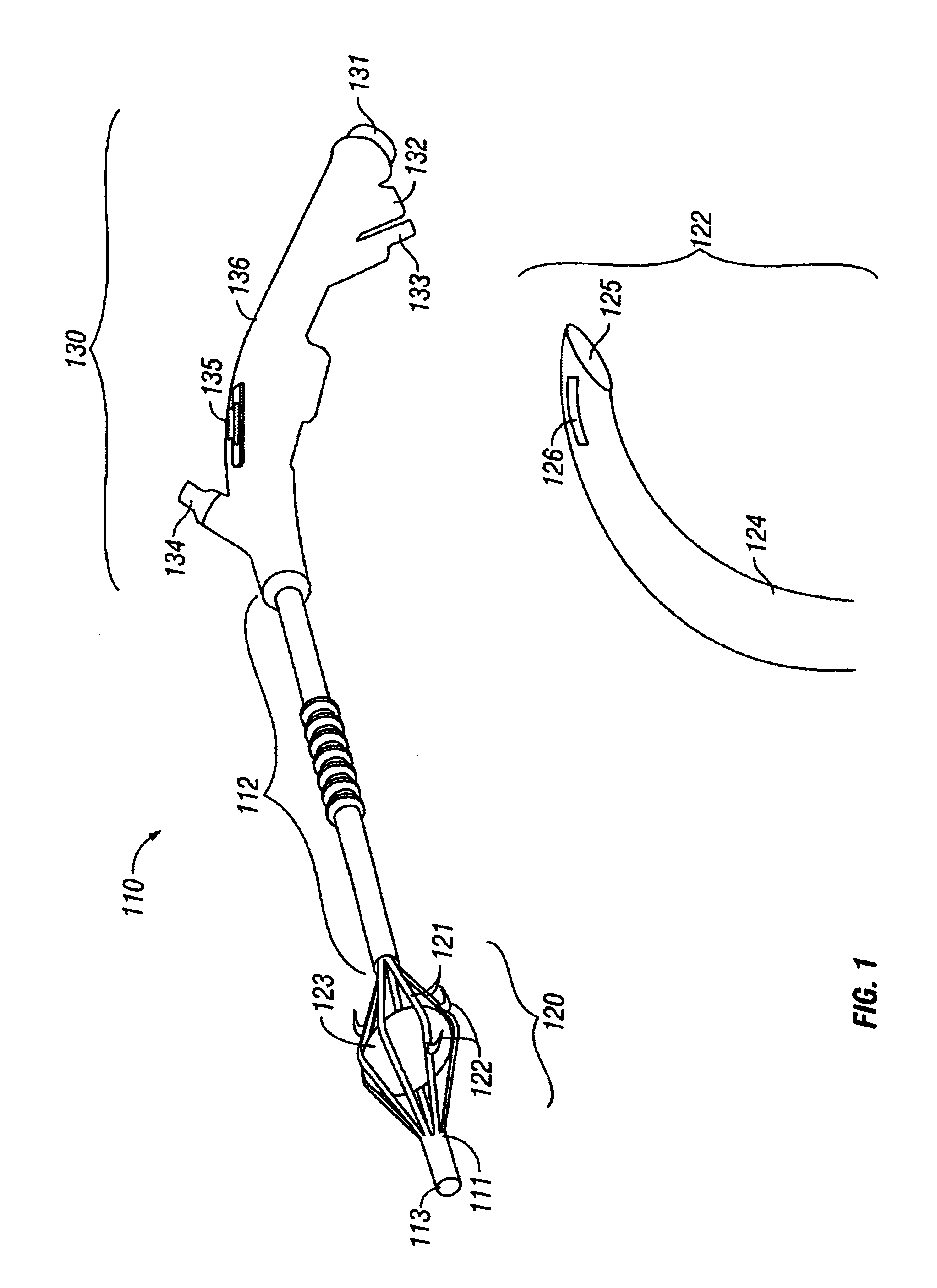
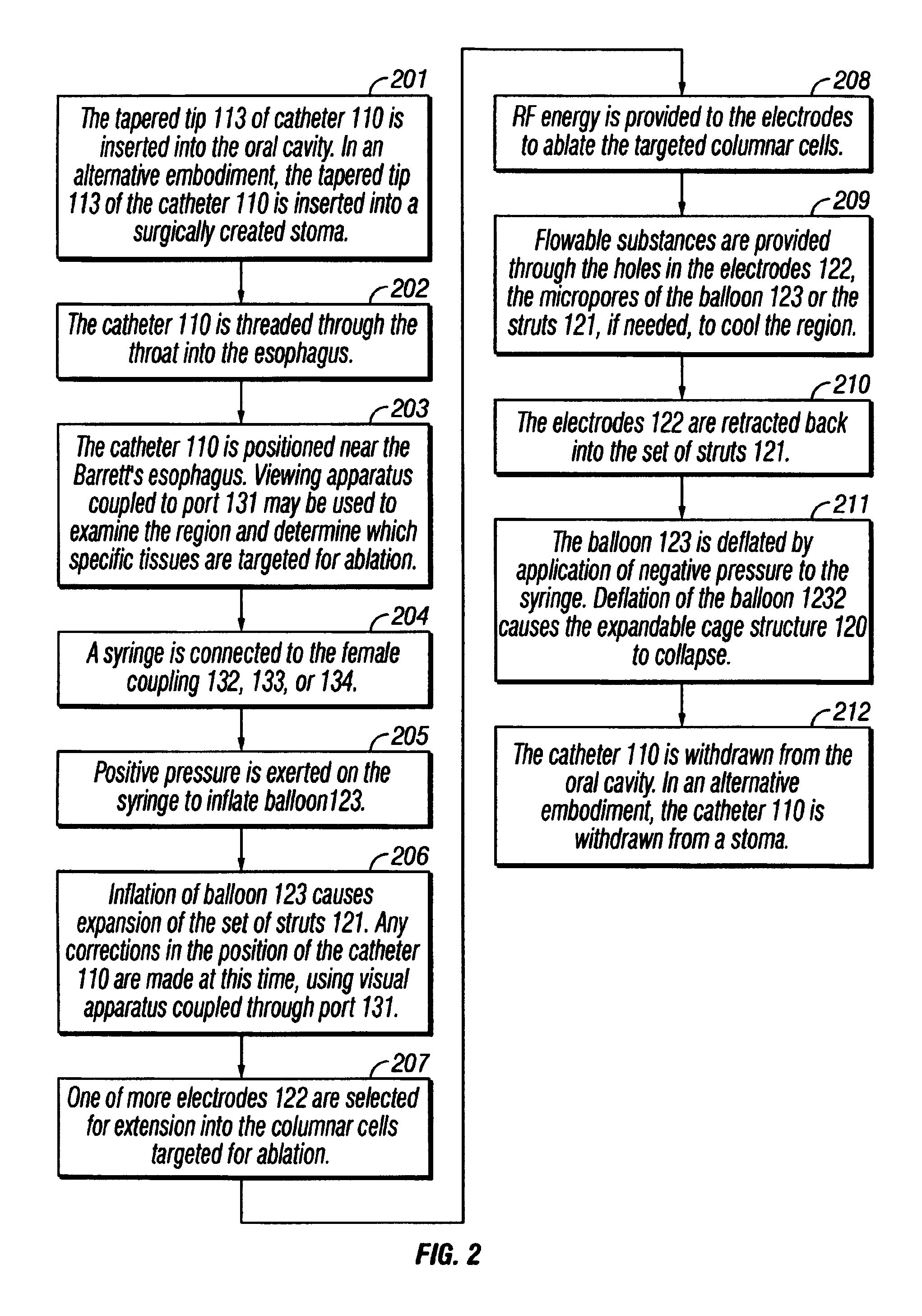
The invention provides an apparatus and system for ablation of body structures or tissue in the region of the rectum. A catheter is inserted into the rectum, and an electrode is disposed thereon for emitting energy. The environment for an ablation region is isolated or otherwise controlled by blocking gas or fluid using a pair of inflatable balloons at upstream and downstream locations. Inflatable balloons also serve to anchor the catheter in place. A plurality of electrodes are disposed on the catheter and at least one such electrode is selected and advanced out of the catheter to penetrate and ablate selected tissue inside the body in the region of the rectum. The electrodes are coupled to sensors to determine control parameters of the body structure or tissue, and which are used by feedback technique to control delivery of energy for ablation or fluids for cooling or hydration. The catheter includes an optical path disposed for coupling to an external view piece, so as to allow medical personnel to view or control positioning of the catheter and operation of the electrodes. The catheter is disposed to deliver flowable substances for aiding in ablation, or for aiding in repair of tissue, such as collagen or another substance for covering lesions or for filling fissures. The flowable substances are delivered using at least one lumen in the catheter, either from at least one hole in the catheter, from an area of the catheter covered by a microporous membrane, or from microporous balloons.
Owner:VIDACARE
Infusion medium delivery device and method with compressible or curved reservoir or conduit
ActiveUS8137314B2Increase fluid pressureReduce internal volumeInfusion syringesFlexible member pumpsCatheterEngineering
A delivery device includes a durable housing portion and a separable disposable portion that selectively engage and disengage from each other. The disposable housing portion secures to the patient-user and may be disposed of after it has been in use for a prescribed period. Components that normally come into contact with a patient-user or with infusion media are supported by the disposable housing portion for disposal after the prescribed use, while the durable housing portion supports other components such as electronics for controlling delivery of infusion media from the reservoir and a drive device and drive linkage.
Owner:MEDTRONIC MIMIMED INC
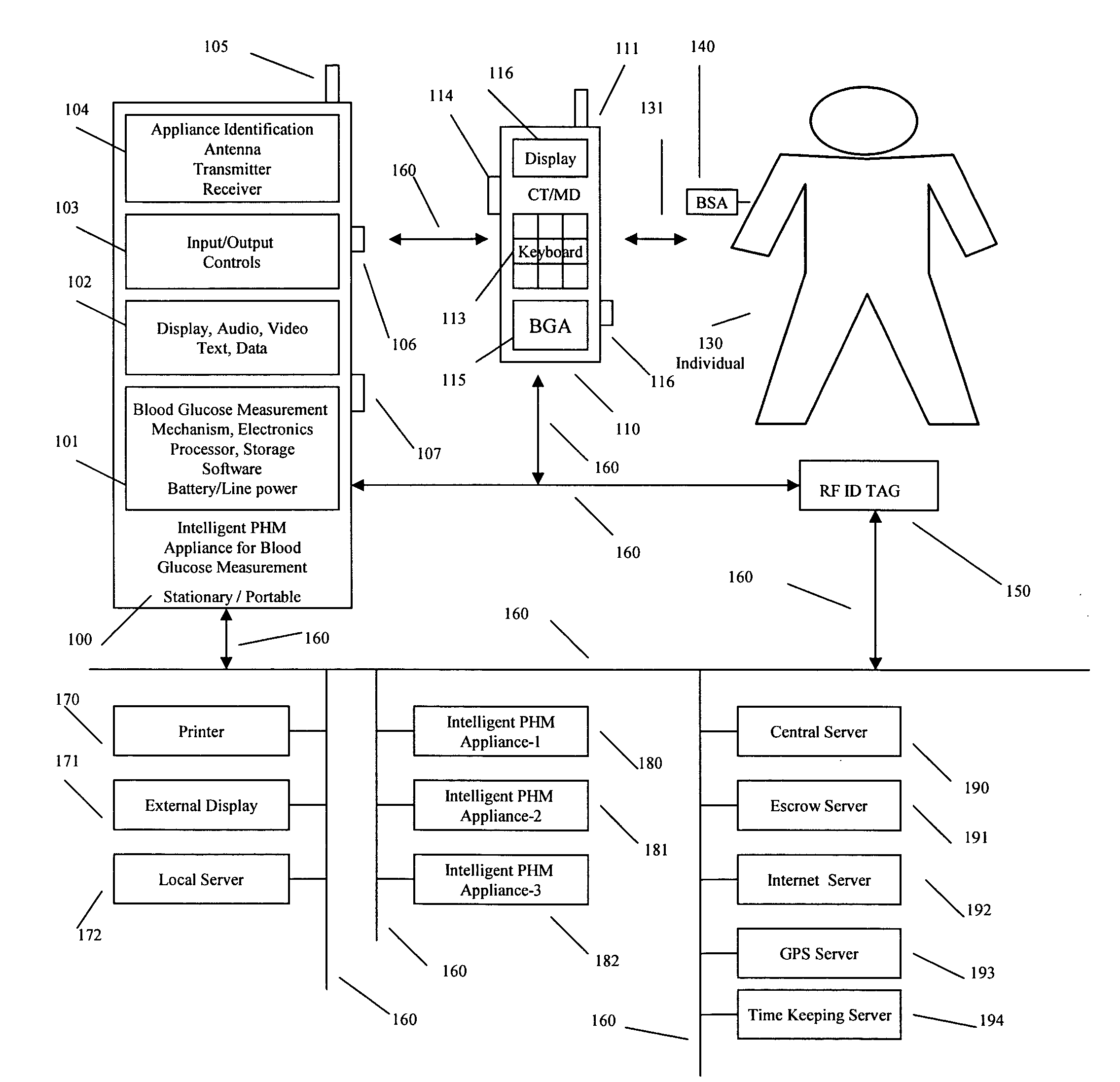
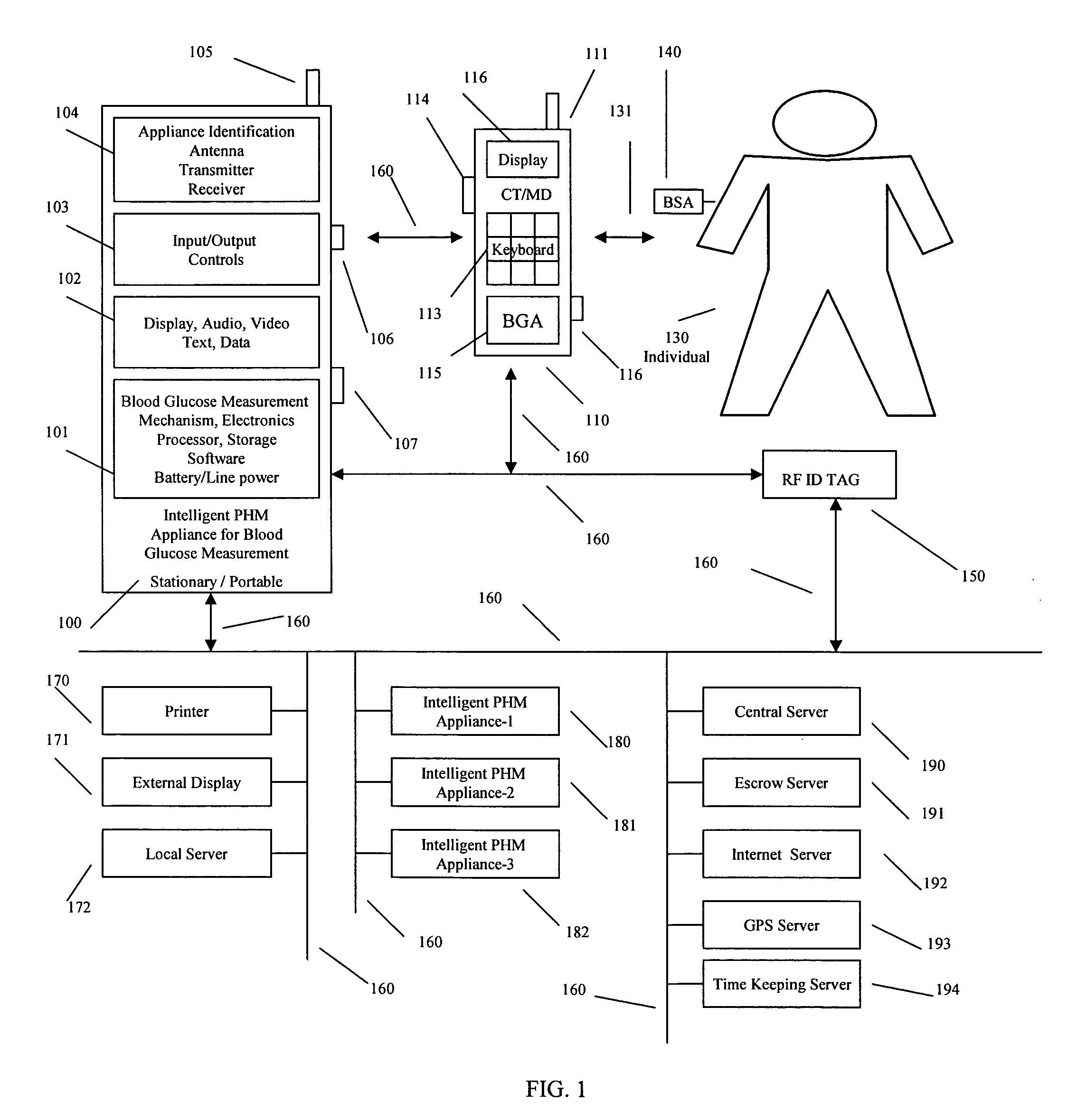
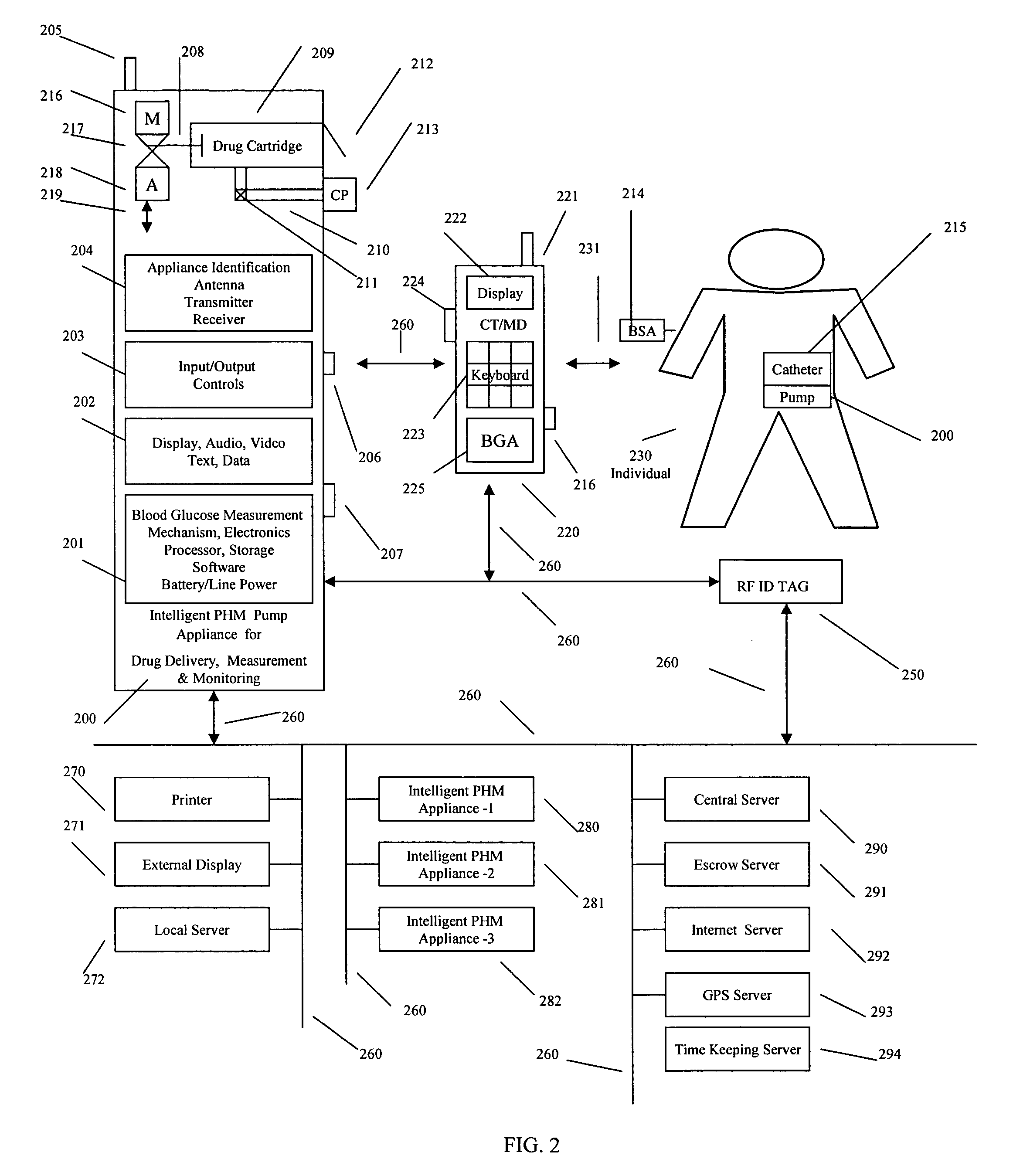
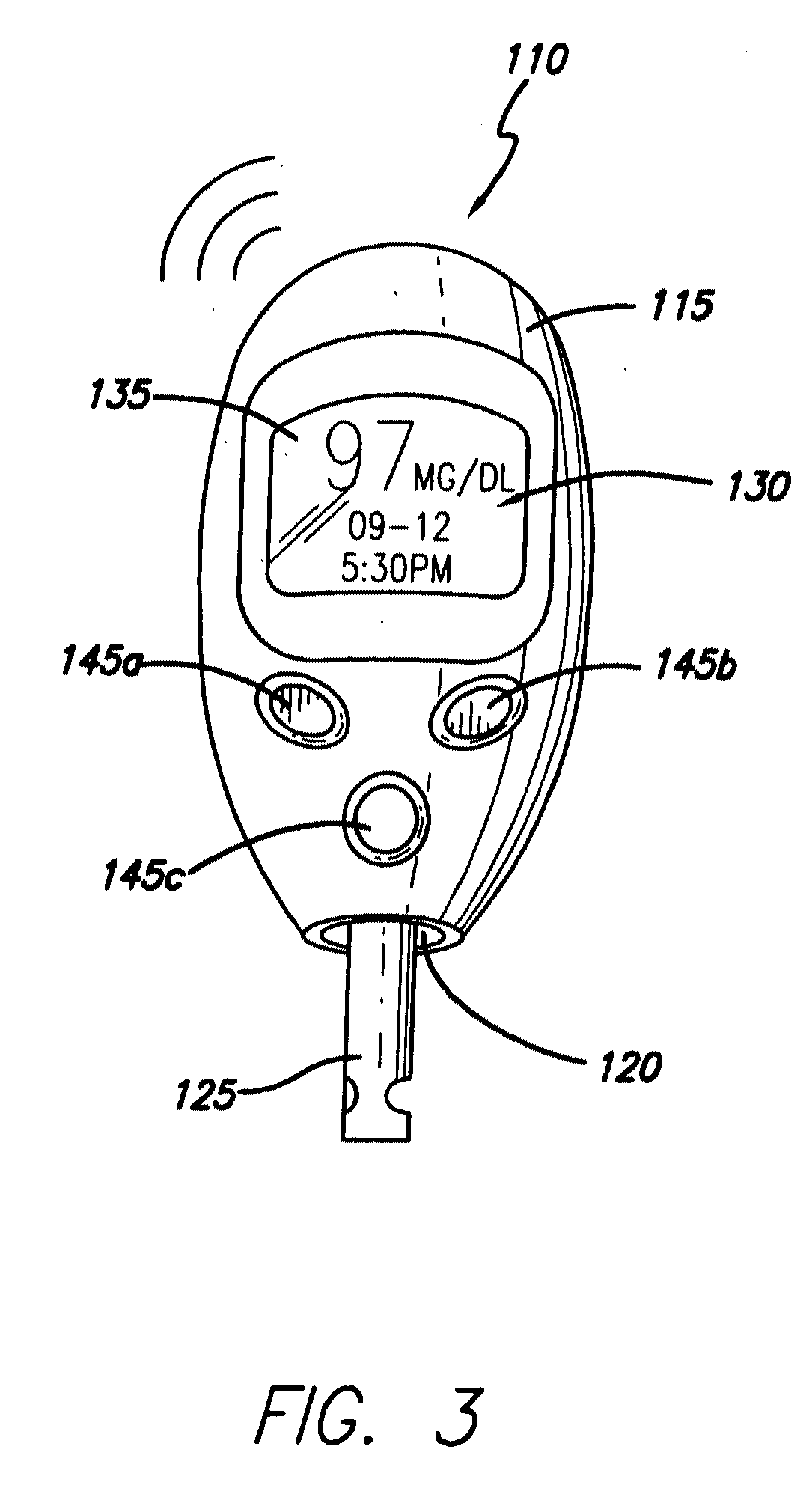
Intelligent personal health management appliances for the measurement and monitoring of health factors and controlled delivery of drugs
InactiveUS20080139907A1Precise positioningPointing accuratelyPhysical therapies and activitiesElectrotherapyDiseaseDiabetes mellitus
A system, method and apparatus for real time measurement and monitoring of various personal health parameters including controlled delivery of drugs / medications by intelligent pump appliances, intelligent inhalation appliances and intelligent skin patch appliances used in a standalone manner or in a wired or wireless networked configuration, in conjunction with various peripheral devices, other intelligent appliances, servers, RF ID Tags and stationary / mobile devices. The intelligent appliances relate to the measurement, monitoring and delivery of insulin / other drugs for the treatment of diabetes and other diseases. The method also additionally includes the application of intelligent appliances for pain management including visualization of organs and body locations exhibiting pain.
Owner:IP HLDG LLC
Treatment of tissue in sphincters, sinuses and orifices
Owner:NOVASYS MEDICAL
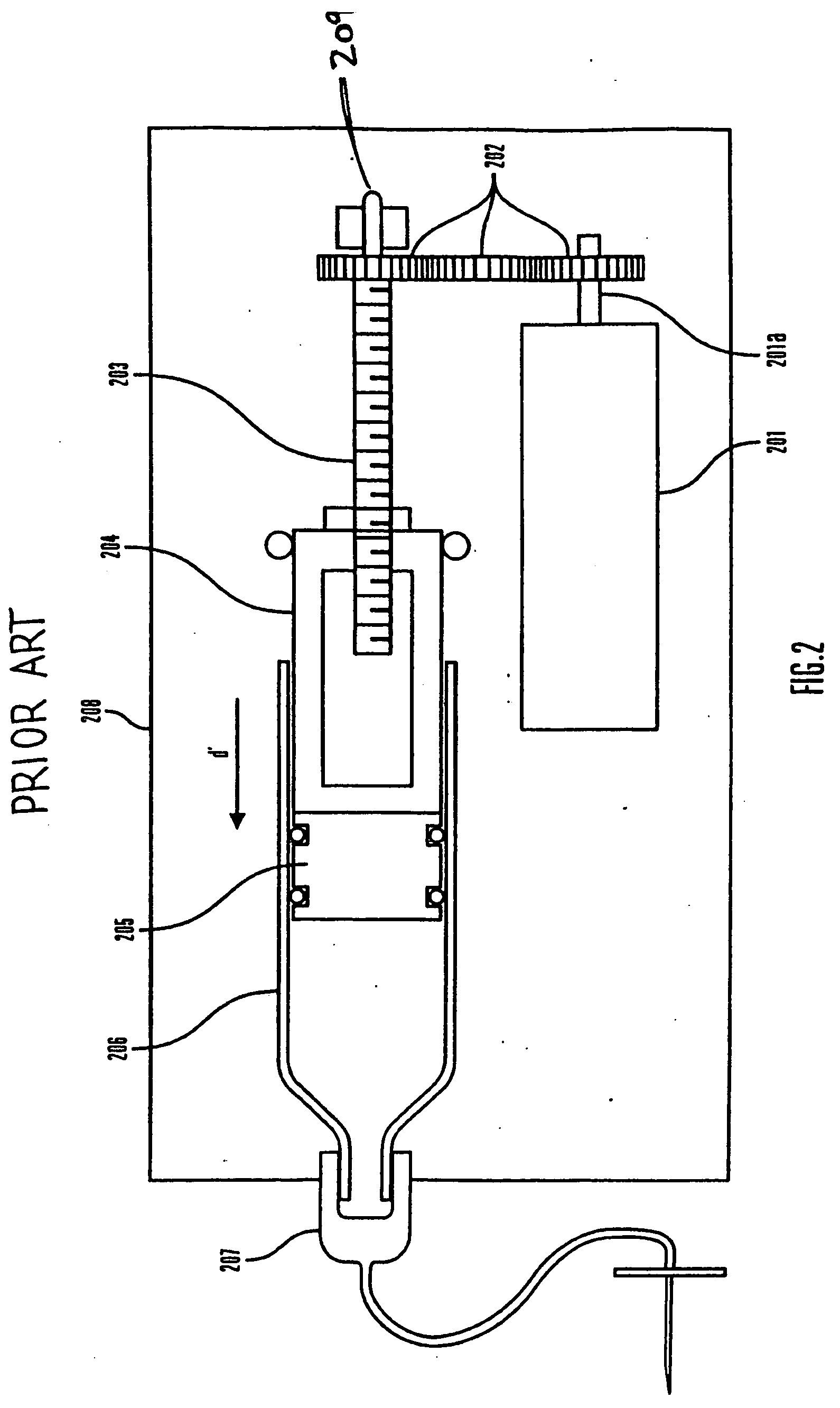
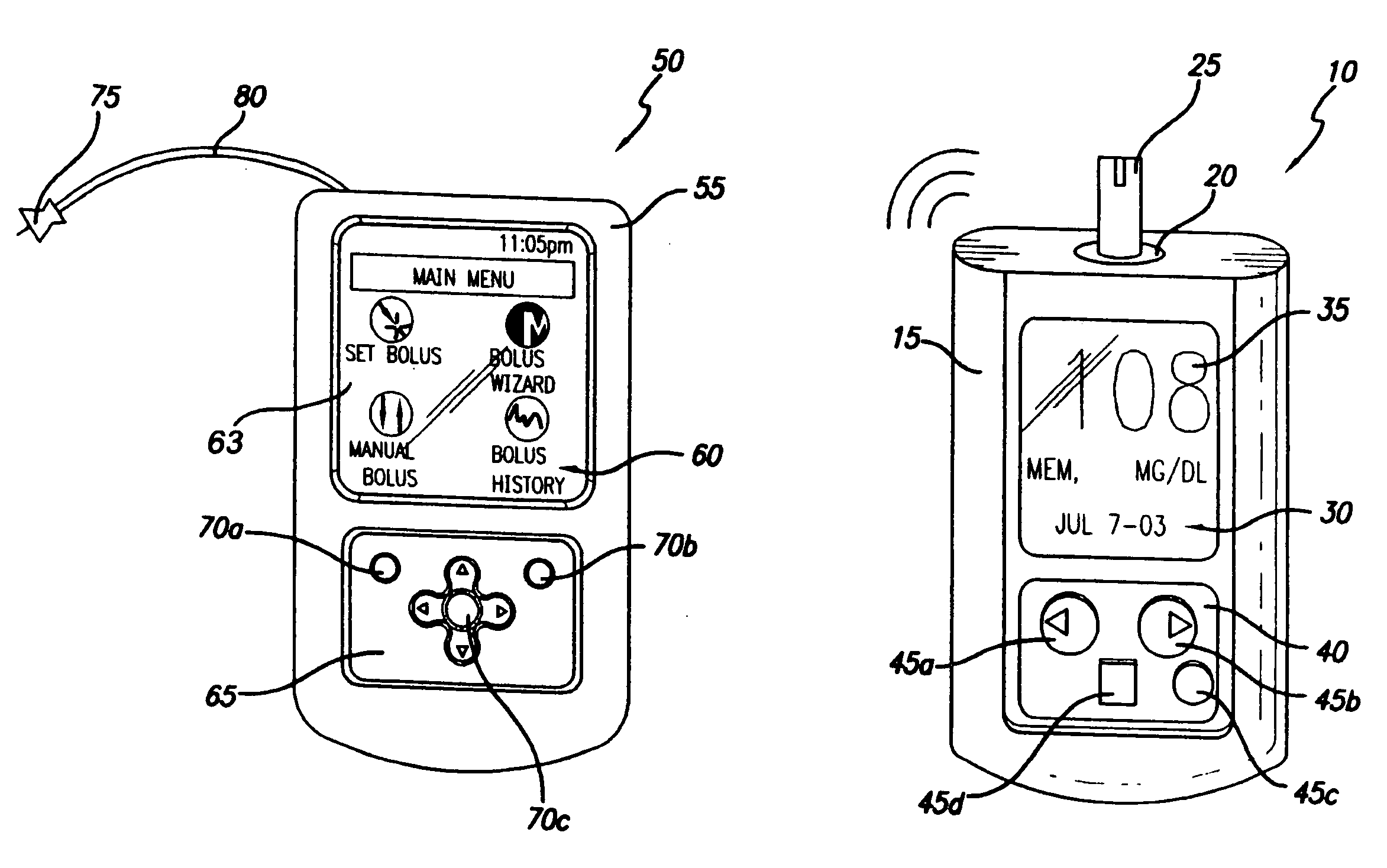
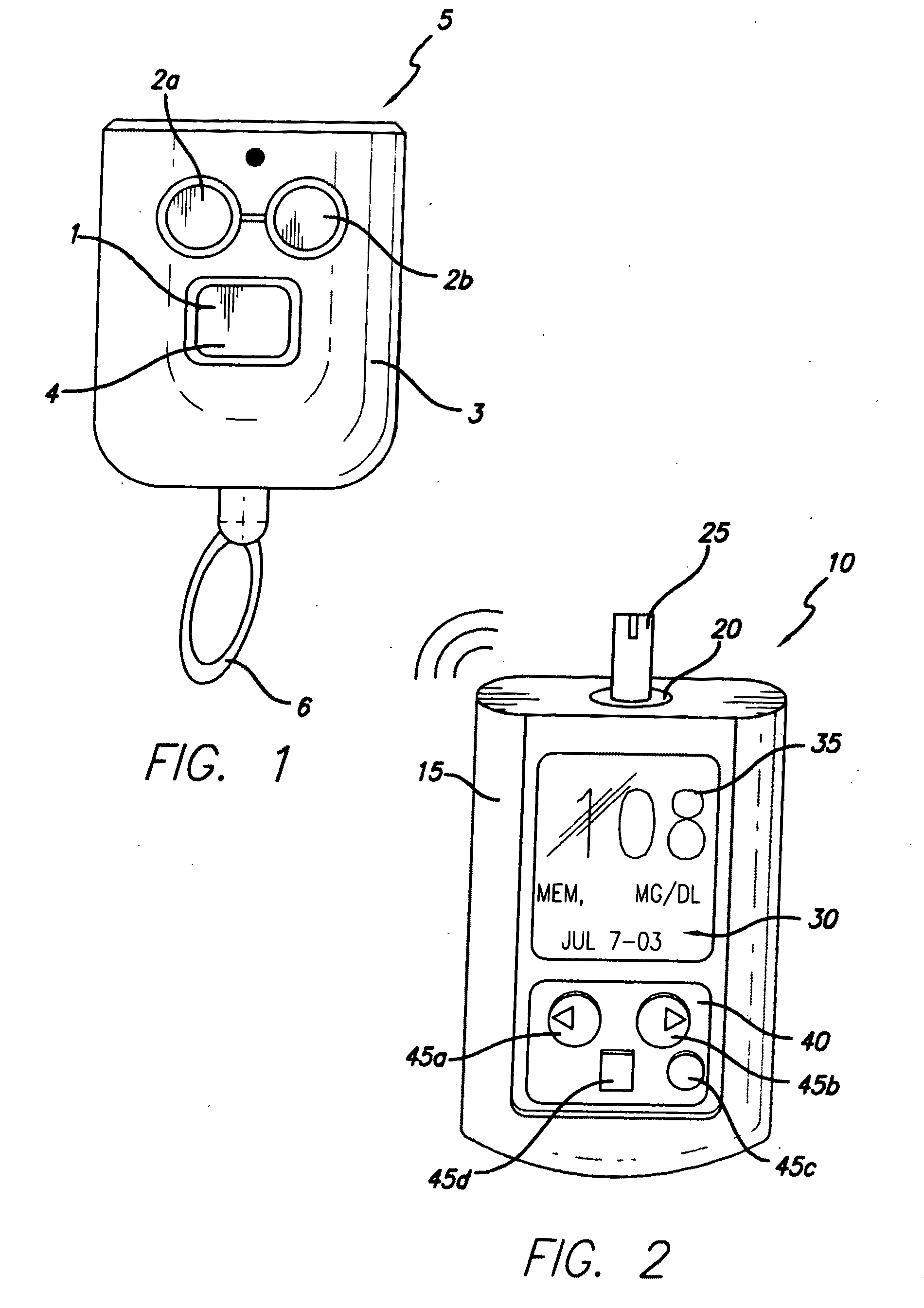
Controller device for an infusion pump
InactiveUS20070060869A1Avoid interferenceMinimizes potential for errorMedical devicesIntravenous devicesCommunications systemEngineering
An infusion system that includes a controller device and a communication system to transmit the communications from the controller device to an infusion device pump that controls delivery of fluids to the user's body. More particularly, these apparatuses and methods are for providing convenient monitoring and control of the infusion pump device in determining the appropriate amount of insulin to deliver.
Owner:MEDTRONIC MIMIMED INC
Controlled Delivery of Content Data Streams to Remote Users
InactiveUS20110197237A1Closed circuit television systemsSelective content distributionData streamControlled delivery
The present system provides a website for distributing a plurality of content data streams converted into a common format, wherein each content data stream is encrypted and transmission of the content data stream is a function of available transmission rates to a given remote device.
Owner:GIGA ENTERTAINMENT MEDIA
Controlled delivery of therapeutic agents by insertable medical devices
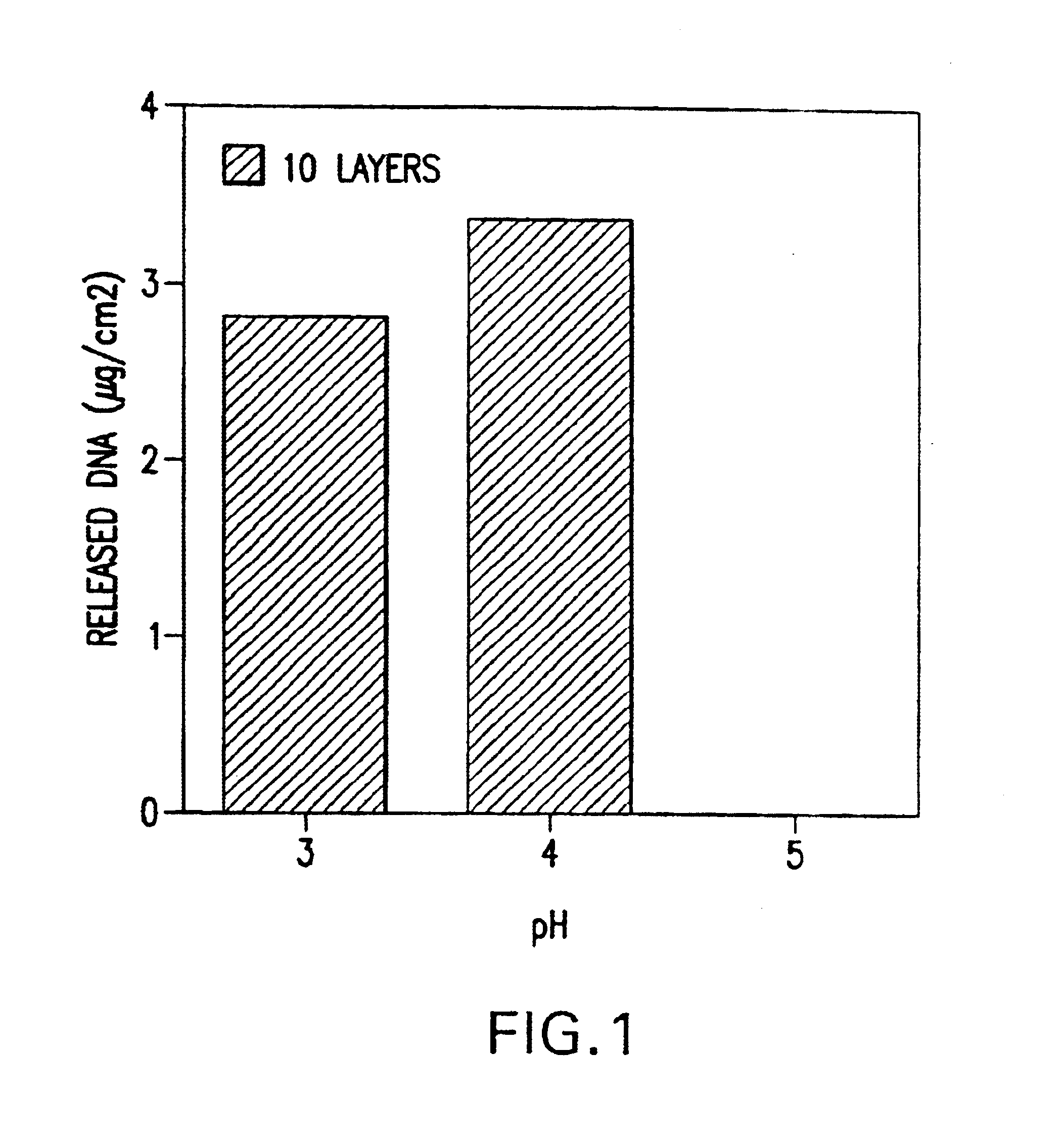
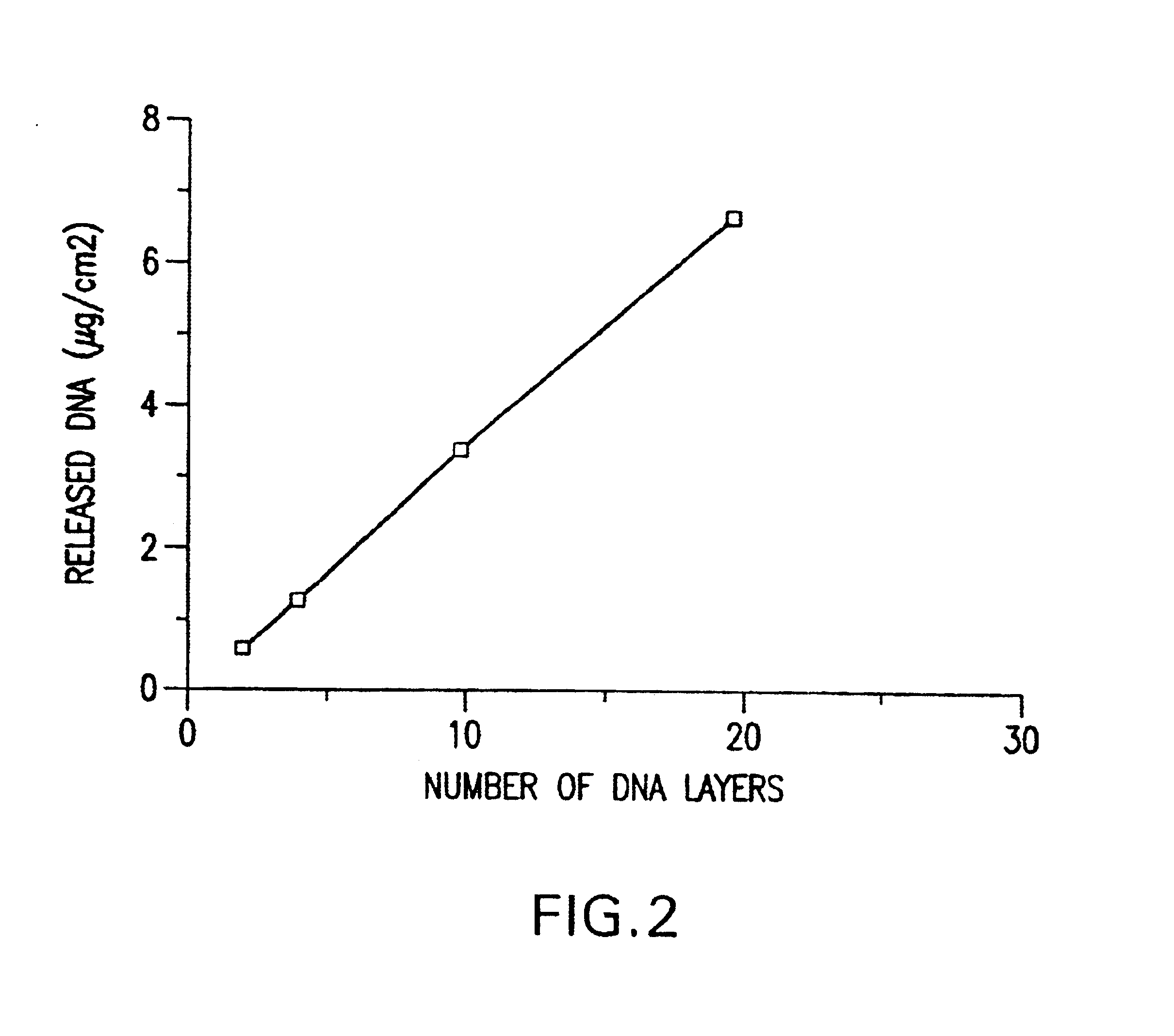
A medical device and method for transportation and release of a therapeutic agent into a mammalian body are disclosed. The medical device is coated with alternating layers of a negatively charged therapeutic agent and a cationic polyelectrolyte, following a controlled adsorption technique. The method is simple, with minimal perturbation to the therapeutic agent and uses clinically acceptable biopolymers such as human serum albumin. The amount of the therapeutic agent that can be delivered by this technique is optimized by the number of the layers of the therapeutic agent adsorbed on the surface of medical device. There is a washing step between alternate layers of the therapeutic agent and cationic polyelectrolyte carrier, so that the amount of the therapeutic agent on the insertable medical device represents the portion that is stably entrapped and adsorbed on to the medical device. The insertable medical device and method according to this invention are capable of reproducibly delivering therapeutic agent to a site in a mammalian body, and allow for a highly reproducible and controllable release kinetics of the therapeutic agent.
Owner:SCI MED LIFE SYST
Infusion pumps and methods and delivery devices and methods with same
ActiveUS20080051716A1Reduce liquid volumeIncrease the amount of fluidContracting/expanding measuring chambersMedical devicesEngineeringInfusion pump
A delivery device includes a durable housing portion and a separable disposable portion that selectively engage and disengage from each other. The disposable housing portion secures to the patient-user and may be disposed of after it has been in use for a prescribed period. Components that normally come into contact with a patient-user or with an infusion medium, including a pump device, are supported by the disposable housing portion for disposal after the prescribed use, while the durable housing portion supports other components such as electronics for controlling delivery of the infusion medium from the reservoir and a drive device and drive linkage.
Owner:MEDTRONIC MIMIMED INC
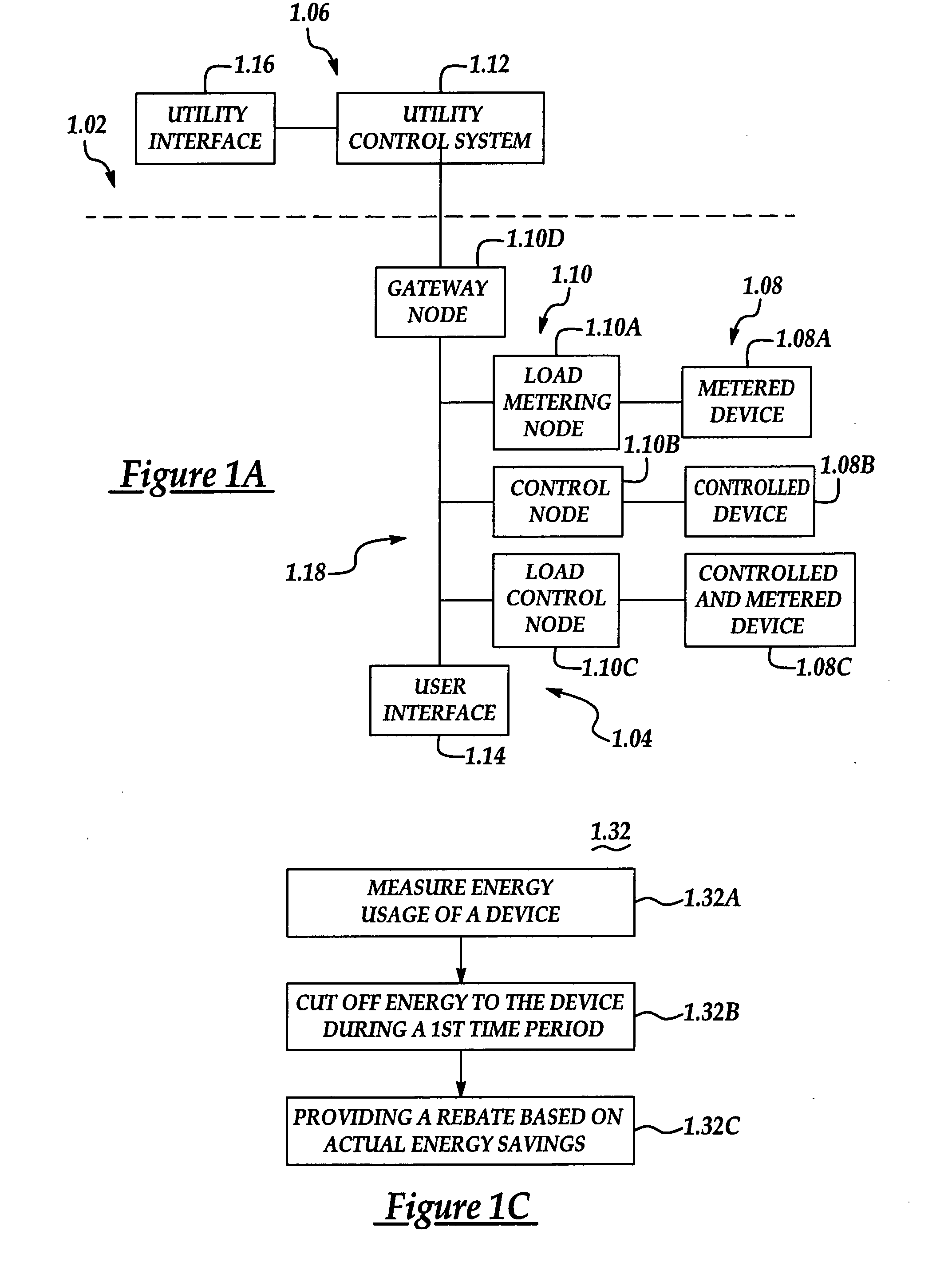
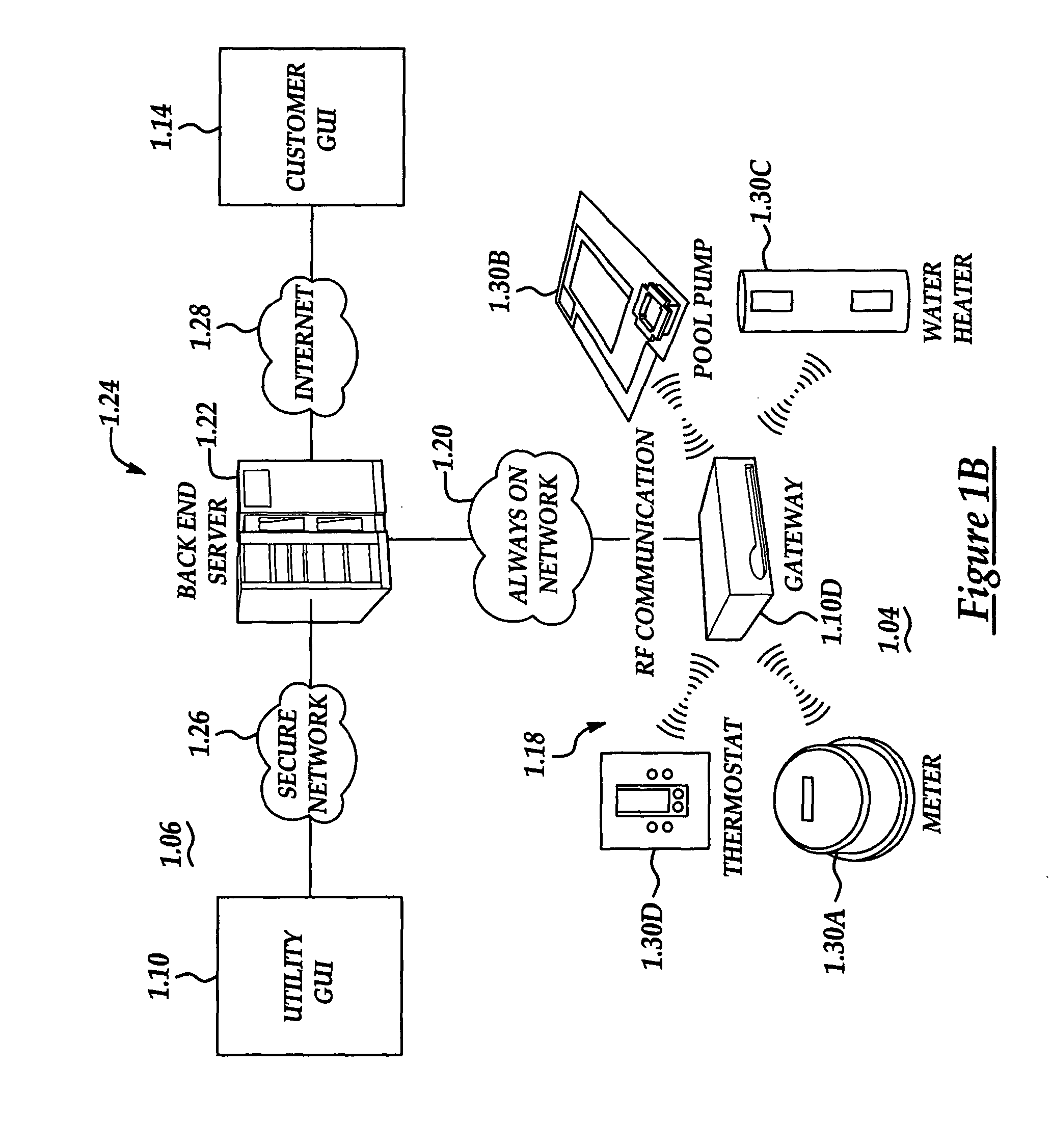
Configurable architecture for controlling delivery and/or usage of a commodity
InactiveUS20050033707A1Dc network circuit arrangementsElectric devicesControl systemComputer science
A system and method manage delivery of energy from a distribution network to one or more sites. Each site has at least one device coupled to the distribution network. The at least one device controllably consumes energy. The system includes a node and a control system. The node is coupled to the at least one device for sensing and controlling energy delivered to the device. A control system is coupled to the node and distribution network for delivering to the node at least one characteristic of the distribution network. The node for controls the supply of energy to the device as a function of the at least one characteristic.
Owner:ROBERTSHAW CONTROLS CO
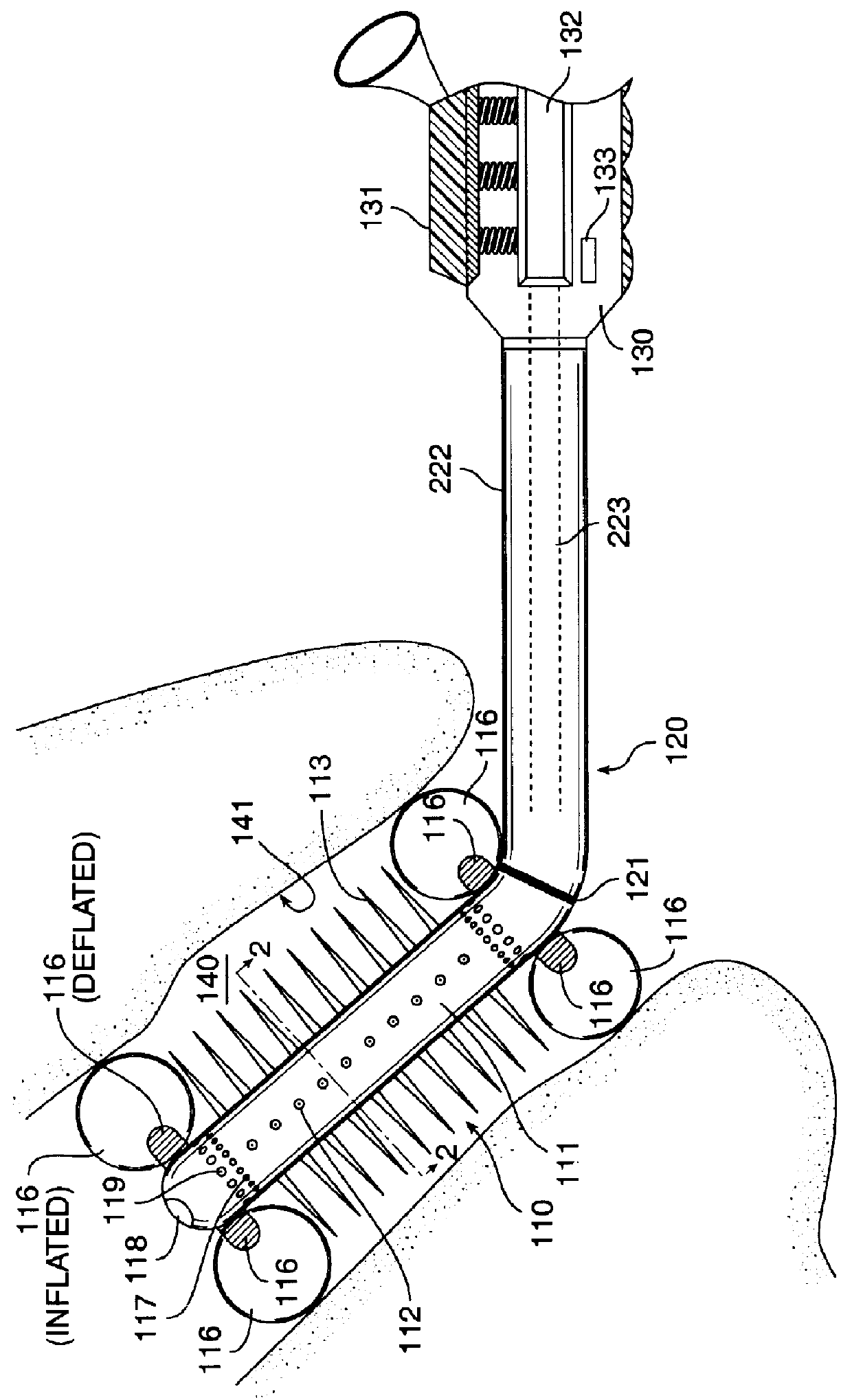
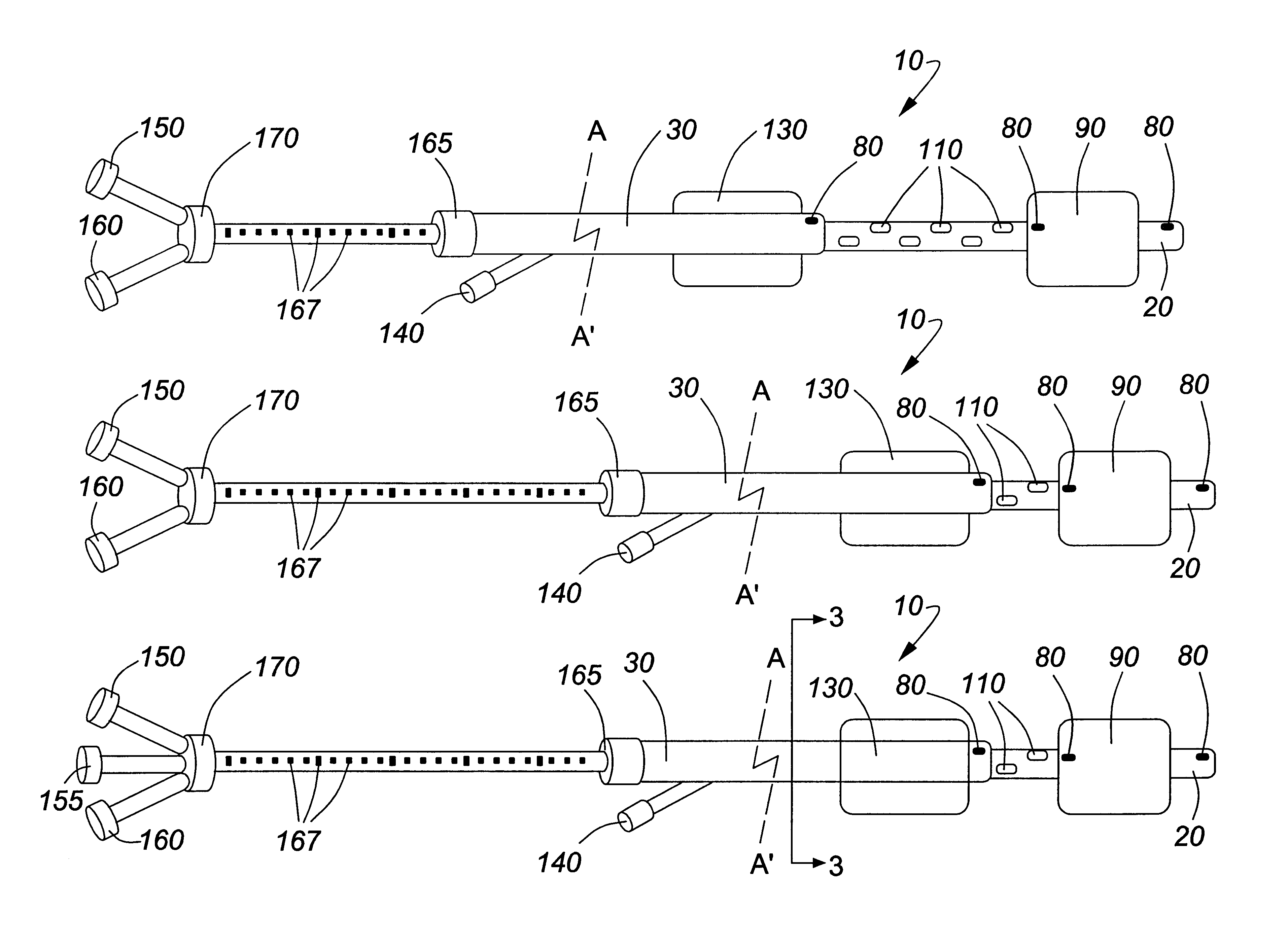
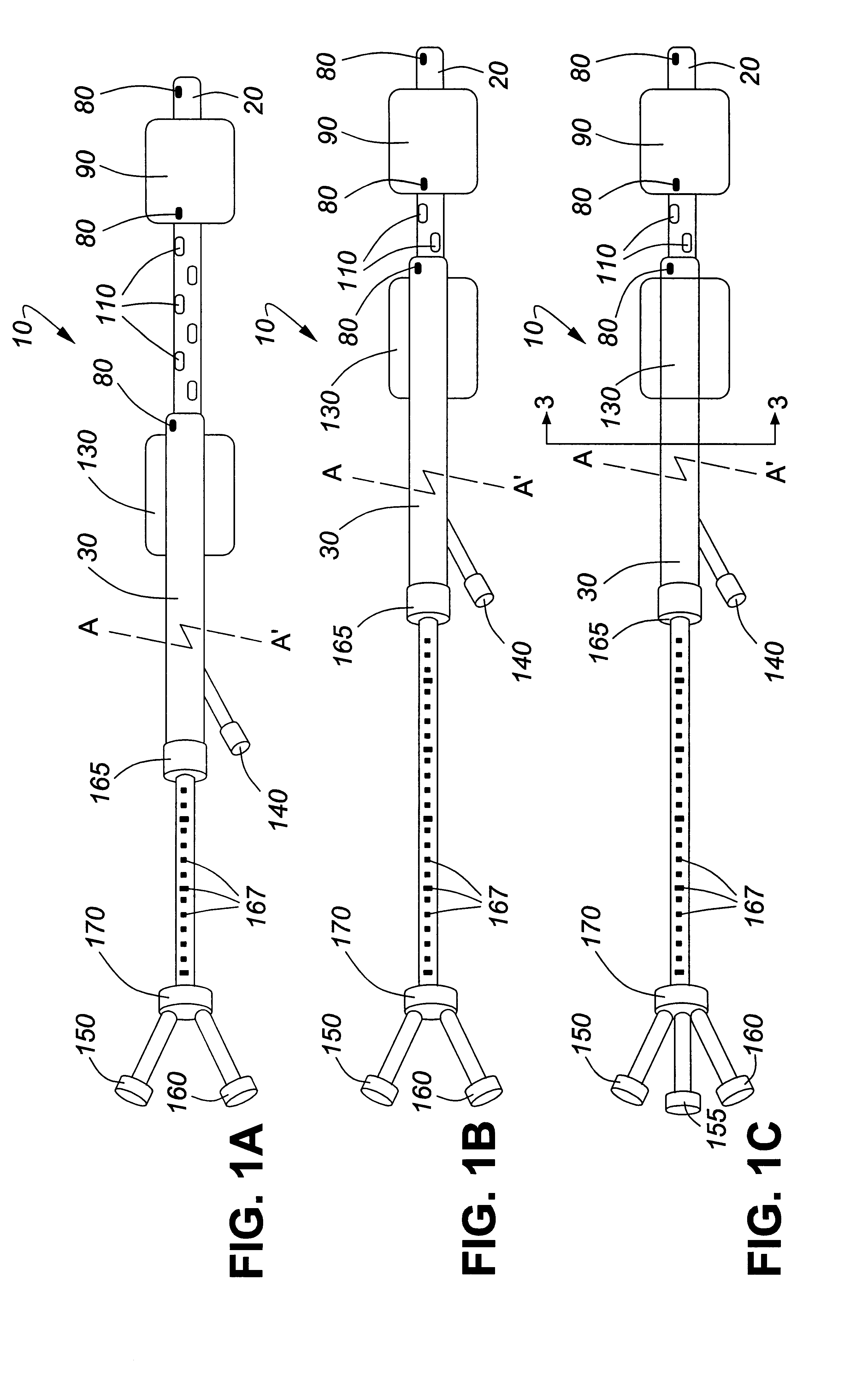
Adjustable multi-balloon local delivery device
The present invention pertains to a local delivery device comprising a distal catheter unit and a proximal catheter unit which may be positioned by sliding over the distal catheter unit. Both the distal and the proximal catheter units have separate inflatable occluding balloons. The slidable positioning of the catheter units in relation to each other provides for variable inter-balloon distances, which in turn provides for a variably sized occlusion region in a hollow tubular organ, for example a vessel. Dispersed on the catheter shaft between the two occluding balloons are multiple infusion ports through which therapeutic agents may be delivered to an occluded region of a hollow tubular organ. The local delivery device may further comprise a quantifying device for determining the distance between the two occluding balloons. Therefore, precise adjustment of the inter-balloon distance permits controlled delivery of therapeutics to a discreet length of the hollow tubular organ wall.
Owner:OTTAWA HEART INST RES
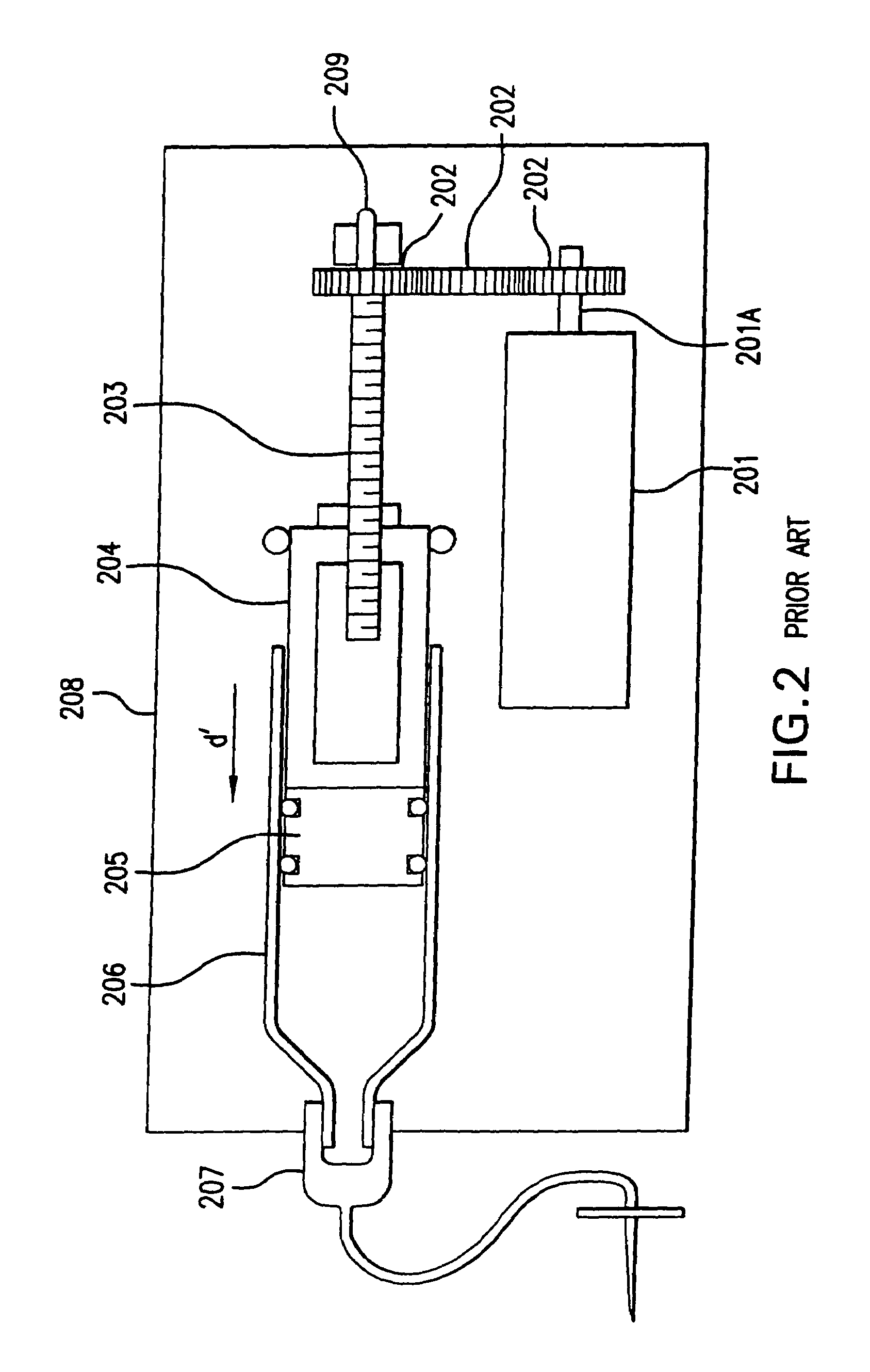
Methods and apparatuses for detecting occlusions in an ambulatory infusion pump
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. A method, system, and an article of manufacture for automatically detecting an occlusion in a medication infusion pump is provided. The electrical current to an infusion pump is measured. Based on measurements of one or more variables, the infusion pump detects whether there is an occlusion in the system. The methods of detecting occlusions may be dynamic.
Owner:MEDTRONIC MIMIMED INC
Infusion medium delivery device and method with compressible or curved reservoir or conduit
ActiveUS20080051709A1Reduce internal volumeIncrease fluid pressureInfusion syringesFlexible member pumpsCatheterGuide tube
A delivery device includes a durable housing portion and a separable disposable portion that selectively engage and disengage from each other. The disposable housing portion secures to the patient-user and may be disposed of after it has been in use for a prescribed period. Components that normally come into contact with a patient-user or with infusion media are supported by the disposable housing portion for disposal after the prescribed use, while the durable housing portion supports other components such as electronics for controlling delivery of infusion media from the reservoir and a drive device and drive linkage.
Owner:MEDTRONIC MIMIMED INC
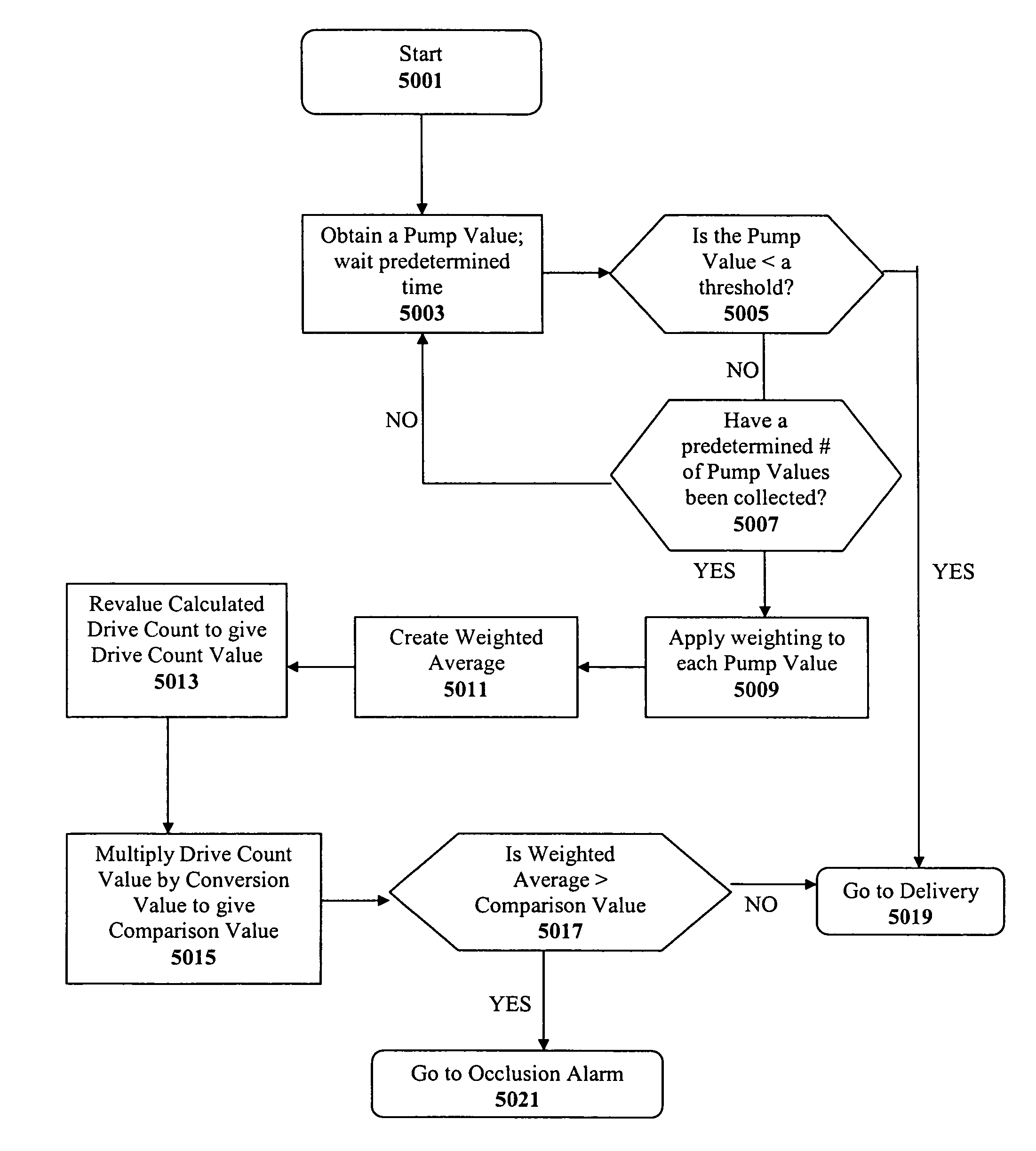
Method and apparatus for detecting occlusions in an ambulatory infusion pump
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. A method, system, and an article of manufacture for automatically detecting an occlusion in a medication infusion pump is provided. The electrical current to an infusion pump is measured. Based on a series of measurements of one or more variables, the infusion pump detects whether there is an occlusion in the system.
Owner:MEDTRONIC MIMIMED INC
Controller device for an infusion pump
InactiveUS20090227855A1Minimizes potential for errorEasy to watchDrug and medicationsMedical devicesEngineeringInfusion pump
An infusion system that includes a controller device and a communication system to provide for two-way communication between the controller device and an infusion device that controls delivery of fluids to a user's body. Either the controller device or the infusion device may be integrated with a characteristic determining device in a single housing. The housing, in turn, may include a test-strip receptacle and an illuminator disposed so as to illuminate an area covering the receptacle and a test-strip inserted therein. The illuminator may be configured to be activated automatically when a test strip is inserted into the receptacle, selectively by the user via a button, key, or similar mechanism, and / or when the ambient light level, measured, e.g., with a light sensor, falls below a predetermined intensity. The illuminator may be a LED emitting white light, and may provide illumination at various levels of intensity.
Owner:MEDTRONIC MIMIMED INC
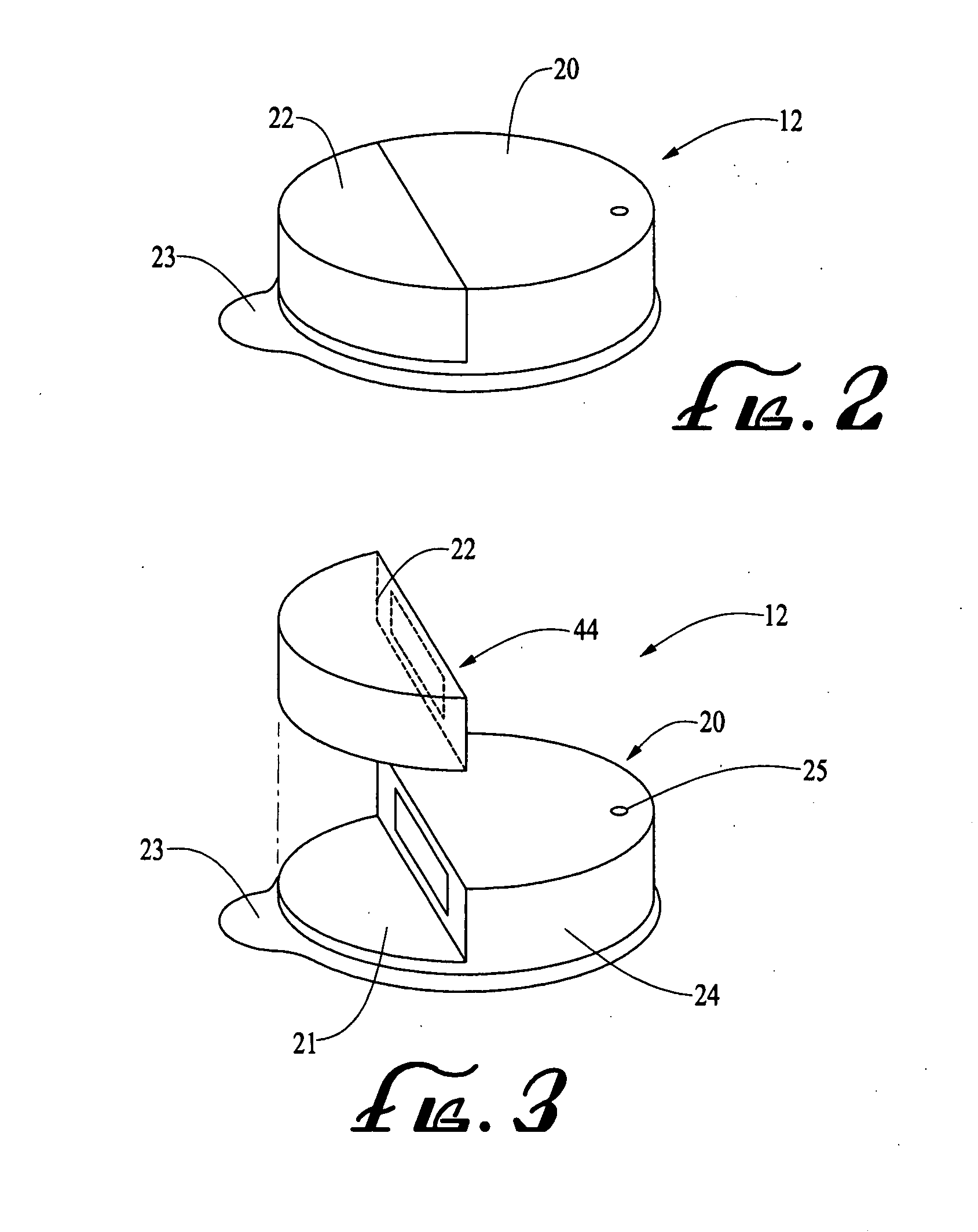
Fluid reservoir piston
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. The piston comprises a first member and a second member. The first member has an external proximate side and an external distal side. The external proximate side is adapted to contact the fluid and is made of a material having a first stiffness. The second member has a first side and a second side and is at least partially disposed within the first member. The first side of the second member is adjacent to the external proximate side of the first member and is made of a material having a stiffness which is greater than the first stiffness.
Owner:MINIMED
Infusion pumps and methods and delivery devices and methods with same
ActiveUS20080097375A1Reduce liquid volumeIncrease the amount of fluidContracting/expanding measuring chambersMedical devicesInfusion pumpBiomedical engineering
A delivery device includes a durable housing portion and a separable disposable portion that selectively engage and disengage from each other. The disposable housing portion secures to the patient-user and may be disposed of after it has been in use for a prescribed period. Components that normally come into contact with a patient-user or with an infusion medium, including a pump device, are supported by the disposable housing portion for disposal after the prescribed use, while the durable housing portion supports other components such as electronics for controlling delivery of the infusion medium from the reservoir and a drive device and drive linkage.
Owner:MEDTRONIC MIMIMED INC
Method and apparatus for detecting errors, fluid pressure, and occlusions in an ambulatory infusion pump
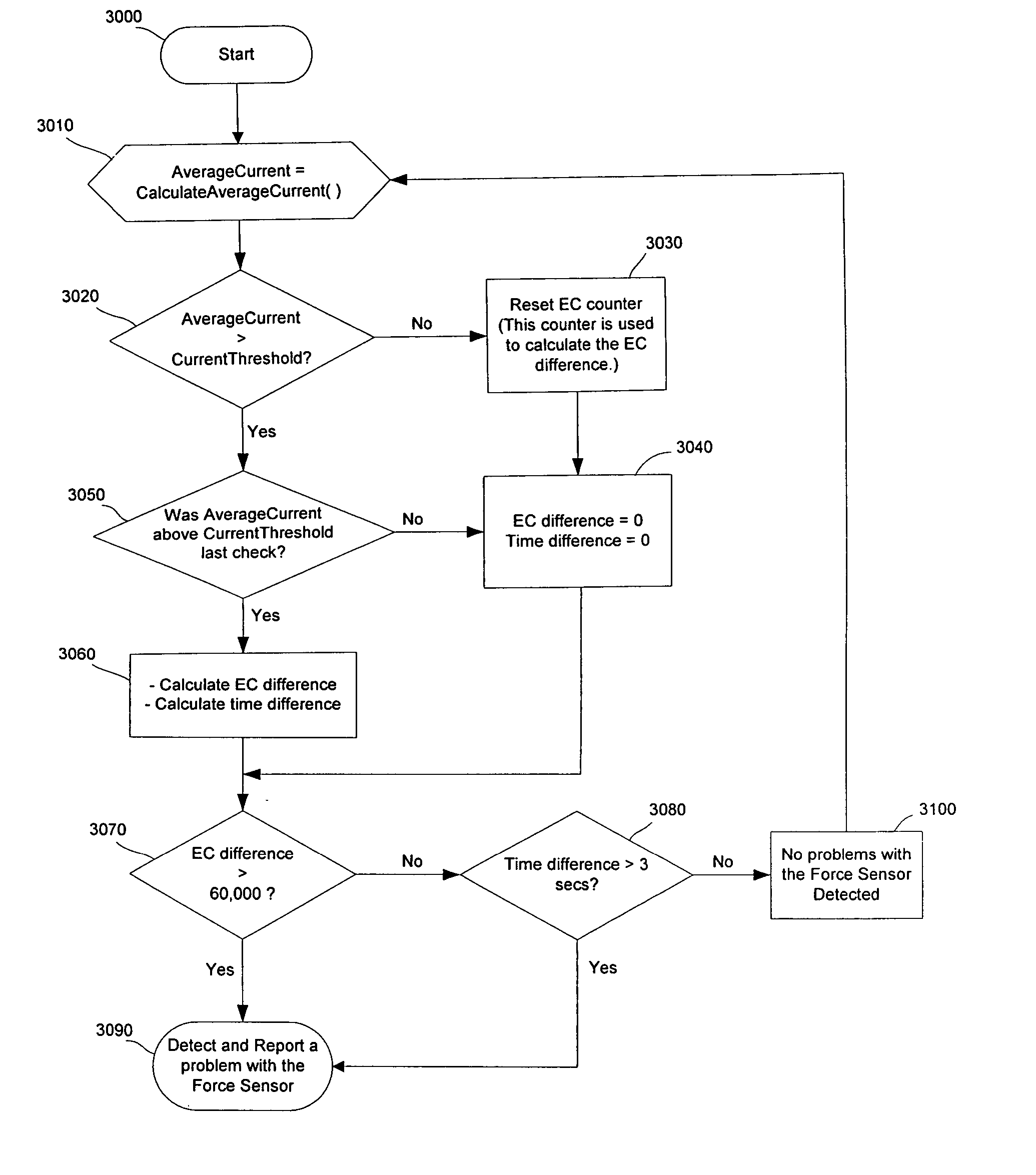
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. A method, system, and an article of manufacture for automatically detecting a force sensor failure in a medication infusion pump is provided. The electrical current to an infusion pump is measured. Based on the current measurements, the infusion pump detects when the plunger slide is seated in the reservoir, and detects a problem with the force sensor when the force sensor independently fails to register a value indicating that the plunger slide is seated in the reservoir.
Owner:MEDTRONIC MIMIMED INC
Method and apparatus for detecting errors, fluid pressure, and occlusions in an ambulatory infusion pump
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. A method, system, and an article of manufacture for automatically detecting a force sensor failure in a medication infusion pump is provided. The electrical current to an infusion pump is measured. Based on the current measurements, the infusion pump detects when the plunger slide is seated in the reservoir, and detects a problem with the force sensor when the force sensor independently fails to register a value indicating that the plunger slide is seated in the reservoir.
Owner:MEDTRONIC MIMIMED INC
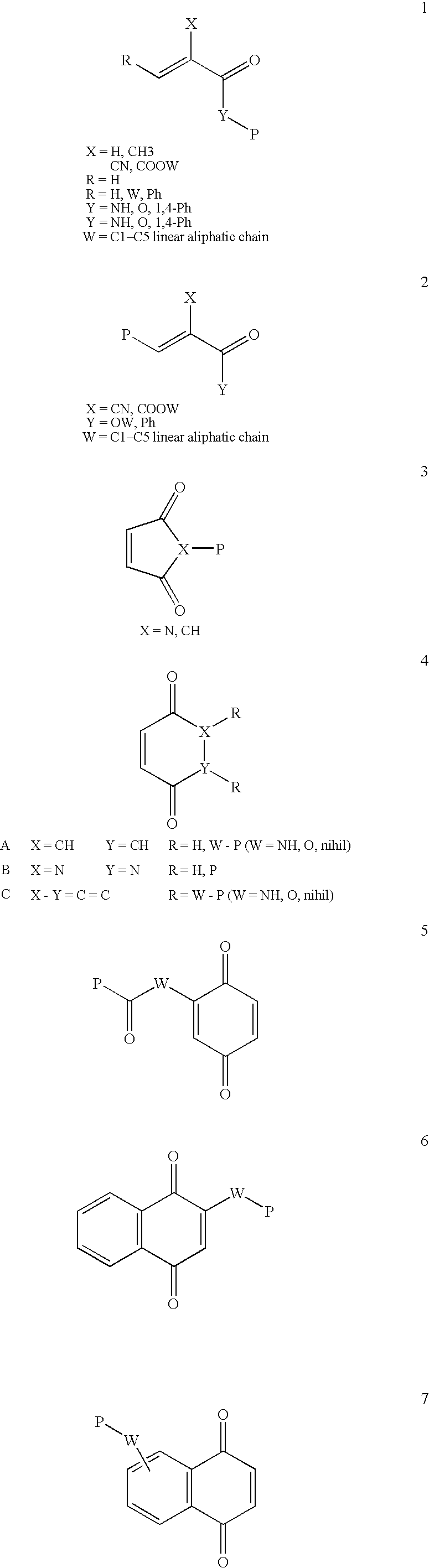
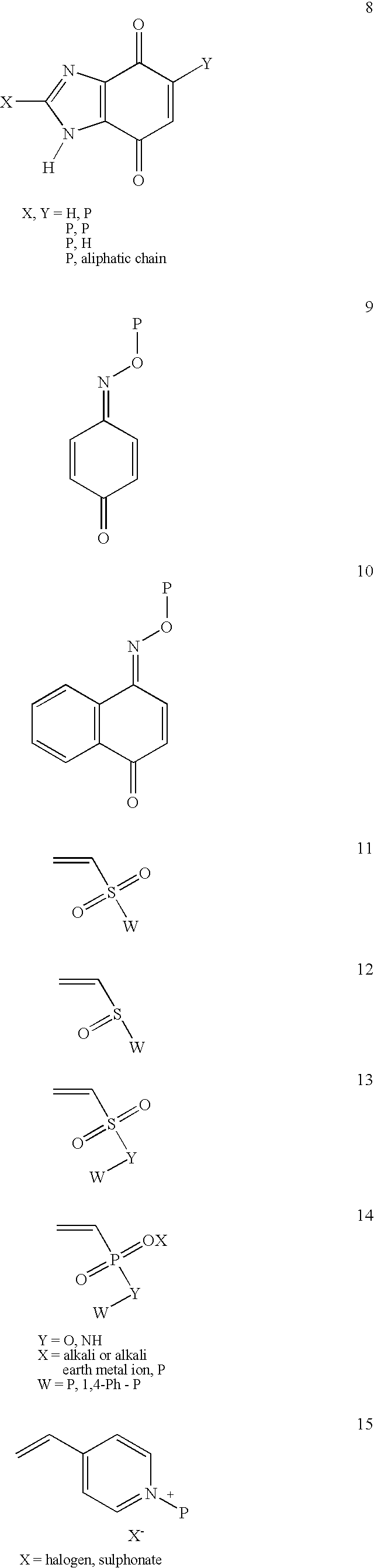
Conjugate addition reactions for the controlled delivery of pharmaceutically active compounds
InactiveUS6958212B1Reducing and delaying onsetGood water solubilitySugar derivativesPeptide/protein ingredientsBiological materialsPolymer
The invention features polymeric biomaterials formed by nucleophilic addition reactions to conjugated unsaturated groups. These biomaterials may be used for medical treatments.
Owner:ETH ZZURICH +1
Method and apparatus for detecting occlusions in an ambulatory infusion pump
An improved pump, reservoir and reservoir piston are provided for controlled delivery of fluids. A motor is operably coupled to a drive member, such as a drive screw, which is adapted to advance a plunger slide in response to operation of the motor. The plunger slide is removably coupled to the piston. A method, system, and an article of manufacture for automatically detecting an occlusion in a medication infusion pump is provided. The electrical current to an infusion pump is measured. Based on a series of measurements of one or more variables, the infusion pump detects whether there is an occlusion in the system.
Owner:MEDTRONIC MIMIMED INC
Method and apparatus for sorting items
A method and apparatus are provided for sorting items to a plurality of sort destinations. The items are loaded onto one of a plurality of independently controlled delivery vehicles. The delivery vehicles follow a track that guides the delivery vehicles to the sort destinations, which are positioned along the track. Once at the appropriate sort destination, the delivery vehicle ejects the item to the sort destination and returns to receive another item to be delivered.
Owner:OPEX