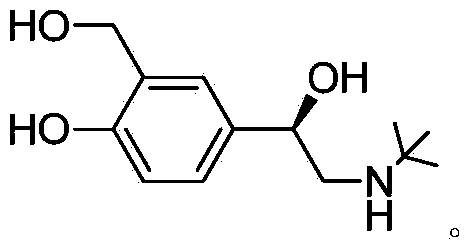
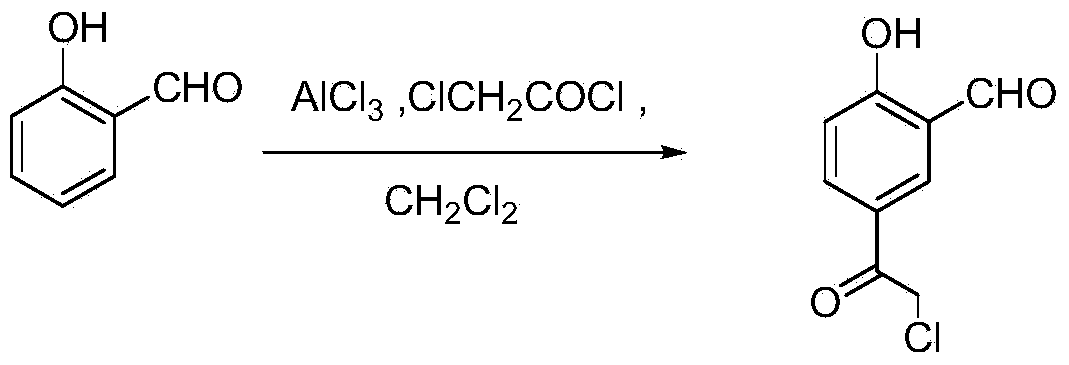Method for preparing levalbuterol
A technology of levosalbuterol and isopropanol, which is applied in the field of preparation of compound levosalbutamol, can solve the problems of low synthesis efficiency, high price, inconvenient large-scale production, etc., and achieve the effect of simple preparation process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
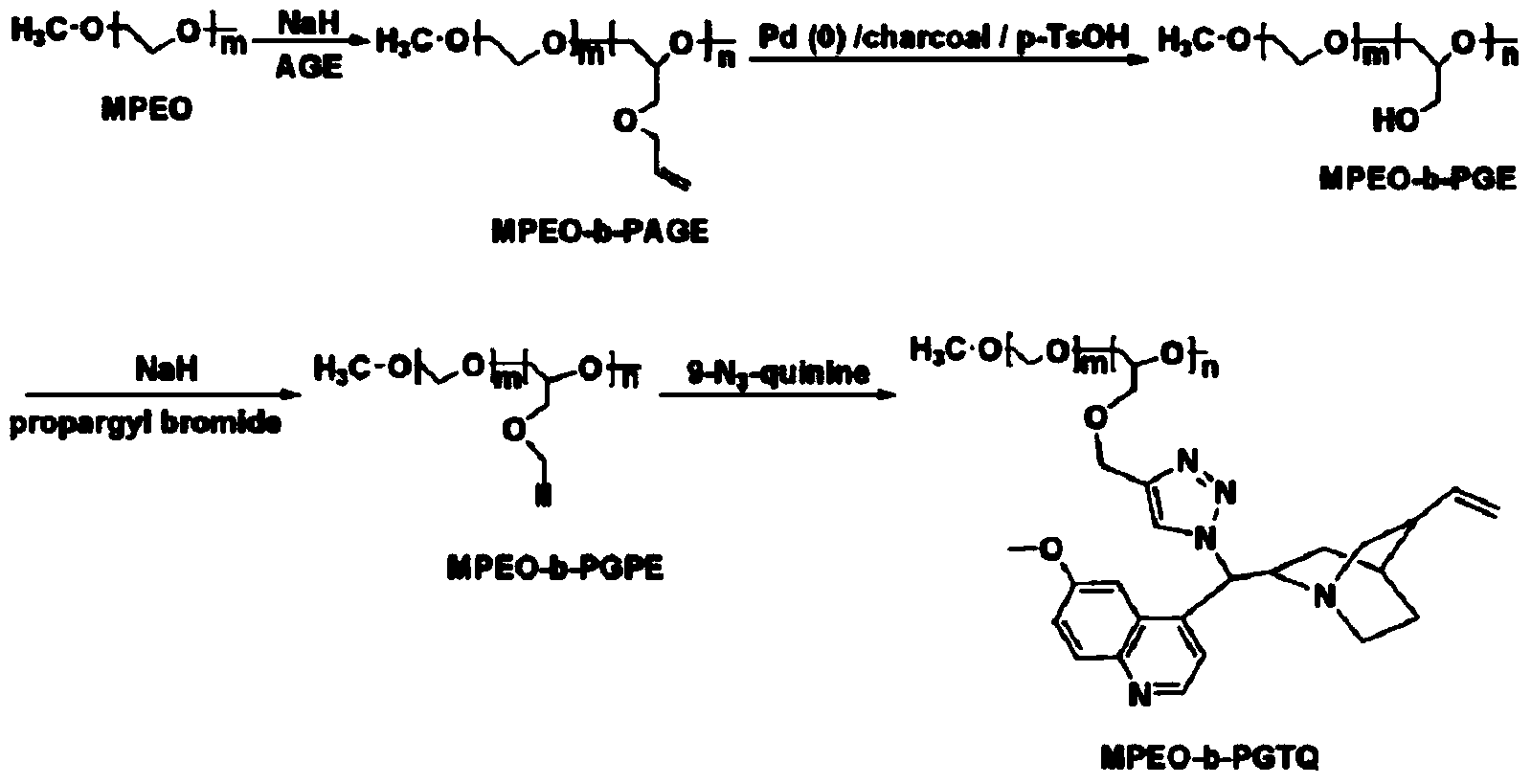
[0035] Example 1: Synthesis of chiral amphiphilic block copolymer MPEO-b-PGTQ with quinine in the side chain
[0036] (1) Vacuum degas the reaction flask three times, add 0.36g (0.015mol) of sodium hydride after elution of mineral oil and 20g (0.01mol) of dry methoxy-terminated polyethylene glycol (MPEO) under nitrogen protection , heated in an oil bath to 100°C, stirred until sodium hydride was dissolved in molten MPEO, and reacted for 1 hour ([NaH] / [OH]=1.5:1), the syringe was purged with nitrogen three times, and allyl glycidyl ether (AGE ) into the reaction bottle, and reacted for 24 hours under the protection of nitrogen at 100°C. After the reaction, the reaction mixture was cooled to room temperature. Dissolve the crude product in a certain amount of dichloromethane, add silica gel, and remove the dichloromethane by rotary evaporation to obtain silica gel powder with polymer adsorbed, which is transferred to a silica gel column, and the PAGE homopolymer is first eluted w...
Embodiment 2
[0040] Embodiment 2: the synthesis of (chloroacetyl) salicylaldehyde
[0041]
[0042] Add 20g (0.15mol) of anhydrous aluminum trichloride to a 250mL three-necked flask, add 15mL of dichloromethane dropwise while stirring, raise the temperature to 50°C, and add dropwise 10mL of a dichloromethane solution of 6.61g (0.042mol) of chloroacetyl chloride , stirred for 30min. 3.66g (0.03mol) of salicylaldehyde was dissolved in 10mL of dichloromethane, dropped into the reactant at 40°C, and refluxed for 12h. The reaction solution was slowly poured into 120 g of crushed ice, 15 mL of water and 30 mL of dichloromethane under stirring. Adjust the pH to 2, stir for 20 min, separate the organic layer, wash the aqueous layer with dichloromethane (30mI×3), combine the dichloromethane layers, wash with water (20mL×2), wash with saturated brine (50mL), anhydrous sulfuric acid Sodium dry. Concentrate to obtain a purple-black oil, which is washed with 40 mL of dichloromethane to obtain a d...
Embodiment 3
[0044] Embodiment 3: [[(1,1-dimethylethyl) amino] acetyl] the synthesis of salicylaldehyde hydrochloride
[0045]
[0046] Suspend 2.43g (0.01mol) of the compound (chloroacetyl) salicylaldehyde in 10mL of isopropanol, add 3.15mL (0.03mol) of tert-butylamine dropwise in an ice-water bath, and stir the reaction at 40°C for 2h. A mixture of 2.55 mL of concentrated hydrochloric acid and 2.55 mL of isopropanol was slowly added dropwise, and the reaction was stirred at room temperature for 10 h. Filter, dry the filter cake to obtain the crude product, wash with a little isopropanol, and dry to obtain the light yellow solid compound [[(1,1-dimethylethyl) amino] acetyl] salicylaldehyde hydrochloride, 2.3g, Yield 85%, mp 232--234°C.
[0047] 1 H NMR (DMSO-d 6 ): 1.15-1.36 (S, 9H), 4.70 (S, 1H), 7.10 (d, 1H), 8.11-8.31 (t, 2H), 10.30 (s, 1H).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More