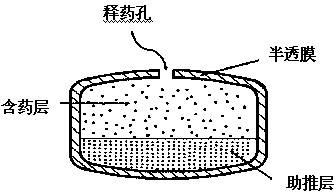
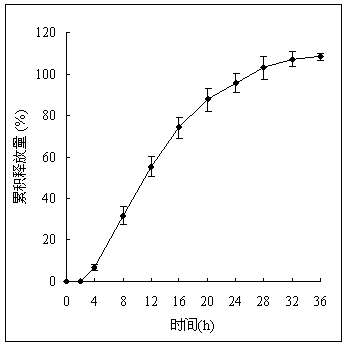
Nifedipine controlled release composition and preparation method thereof
A composition and nitrobenzene technology, applied in the field of medicine, can solve the problems of affecting product quality, organic solvent residues, not being well solved, etc., and achieve the effects of avoiding raw material aggregation, removing acetone residues, and improving production efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0071] The preparation of the drug-containing layer, the specific prescription components are as follows:
[0072] .
[0073] i) Preparation of suspension for granulation: Weigh purified water, add the prescribed amount of binder while stirring, stir until the binder is completely swollen, add nifedipine while stirring to form a suspension, pass through 200 mesh Sieve, rinse the screen with purified water, merge into the suspension, keep stirring, and set aside;
[0074] ii) Take polyoxyethylene by weighing and add in the fluidized bed granulator, preheat;
[0075] iii) top spray granulation;
[0076] iv) After stopping the liquid spraying, dry, and the particles pass through a 24-mesh sieve;
[0077] v) Add the prescribed amount of magnesium stearate and mix well.
[0078] The particle size distribution is shown in the table below:
[0079] .
Embodiment 2
[0081] The preparation of the booster layer, the specific prescription components are as follows:
[0082] .
[0083] i) Preparation of solutions and suspensions for granulation
[0084] ① Preparation of sodium chloride solution: Dissolve the prescribed amount of sodium chloride in purified water and set aside;
[0085] ② Adhesive solution: Weigh purified water, add the prescribed amount of adhesive while stirring, stir until completely dissolved, and set aside;
[0086] ii) Take polyoxyethylene by weighing and add in the fluidized bed granulator, preheat;
[0087] iii) top spray granulation, successively spray sodium chloride solution and binder solution into polyoxyethylene;
[0088] iv) After stopping the liquid spraying, dry, and the particles pass through a 24-mesh sieve;
[0089] v) Add the prescribed amount of magnesium stearate and mix well.
[0090] The particle size distribution is shown in the table below:
[0091] .
Embodiment 3
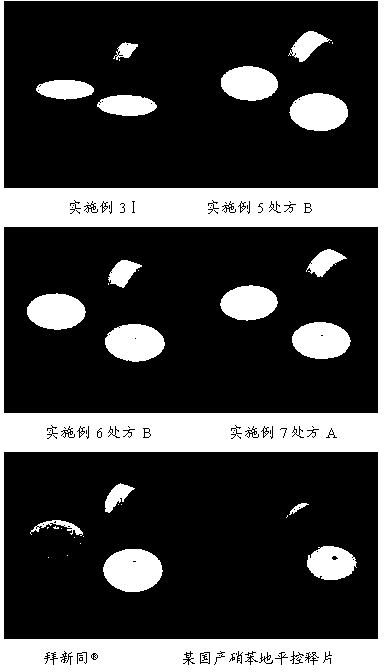
[0106] Embodiment 3 Tablet comparative investigation
[0107] The granules in the prescription B of Example 1 and the prescription B of Example 2 and the mixed powders in Comparative Example 1 and Comparative Example 2 were selected respectively, and the same double-layer tablet press was used to compress the double-layer tablet with the same parameters, and the results are shown in the following table Shown:
[0108] serial number Drug-containing layer booster layer result Ⅰ Embodiment 1 prescription B Embodiment 2 prescription B There is no mixture of two layers of color, and almost no flower flakes are found. Ⅱ Comparative example 1 Comparative example 2 The colors are mixed, and the flower flakes are obvious. Ⅲ Embodiment 1 prescription B Comparative example 2 There are white spots and plaques on the surface of the yellow layer. Ⅳ Comparative example 1 Embodiment 2 prescription B There are yellow spots on the surface o...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com