Acinetobacter baumannii 1a1s_1969 recombinant protein and its preparation method and application
An Acinetobacter baumannii, recombinant protein technology, applied in the biological field, can solve the problems of complex composition of outer membrane complex, complex vaccine composition, large toxic and side effects, etc., to maintain spatial conformation and immunogenicity, and to achieve controllable quality and safety , the effect of simple steps
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0057] Embodiment 1: Cloning of Acinetobacter baumannii A1S_1969 protein
[0058] 1. First, according to the gene sequence of Acinetobacter baumannii 17978 standard strain A1S_1969, its amino acid sequence is shown in SEQ ID NO.3. Bioinformatics software was used for structural analysis, and the signal peptide sequence was as follows: Figure 5 As shown, its spatial structure analysis is as follows Figure 6 shown.
[0059] 2. According to the analysis results, the PCR method is used to amplify the gene fragments of the two fragments of the A1S_1969 protein respectively with the whole genome of Acinetobacter baumannii 17978 as a template (SEQ ID NO.4), and the amplification steps are as follows:
[0060] 1) Design PCR primers as follows, use forward primer P1A1S1969B1 (SEQ ID NO: 9), reverse primer P1A1S1969N2 (SEQ ID NO: 10) to amplify the first fragment, forward primer P2A1S1969B1 (SEQ ID NO: 11), The reverse primer P2A1S1969N2 (SEQ ID NO: 12) amplifies the second fragment...
Embodiment 2
[0103] Example 2: Acinetobacter baumannii-17978 1A1S_1969 protein induced expression, purification and expression form identification in prokaryotic expression system-Escherichia coli
[0104] 1. Induced expression of target protein
[0105] 1) Take 100 μL of the pGEX-6P-2-1A1S_1969 / XL-1blue bacterial solution that was correctly identified by double enzyme digestion and add it to 10 mL of Amp-resistant TB medium, cultivate overnight at 100 rpm at 37°C, take 2 ml of the overnight cultured bacterial solution and add it to 18 mL In the Amp-resistant TB medium (the rest of the bacterial solution is stored in a refrigerator at 4°C for later use), culture at 37°C for 2-3 hours at a rotation speed of 250rpm, and when the secondary activation reaches OD600 of 0.8-1.2, add 4.4 μL of IPTG to make it The final concentration was 200 μM, and then placed on a shaker to induce expression at 30°C for 3 hours, and then induced expression at 16°C overnight.
[0106] 2) Take out the bacterial s...
Embodiment 3
[0112] Embodiment 3: Preparation of 1A1S_1969 protein antigen
[0113] 1. Amplify culture to obtain protein
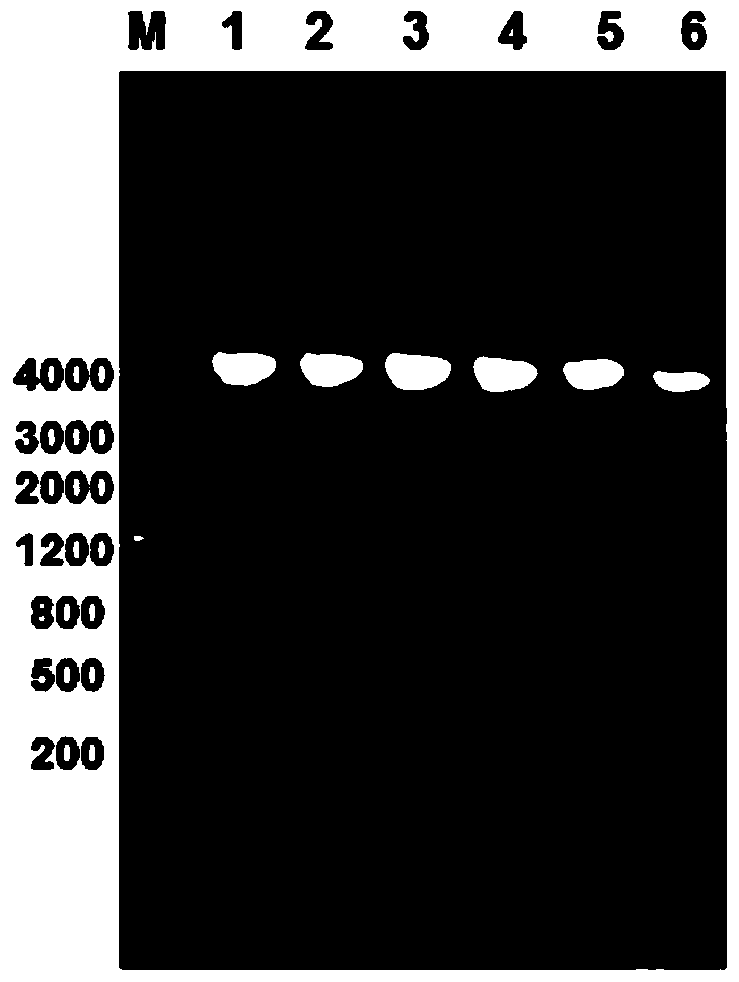
[0114] Inoculate the pGEX-6P-2-1A1S_1969 / XL-1blue strains stored in the -80°C refrigerator on LB ampicillin-resistant plates, and cultivate overnight at 37°C; pick a single colony and inoculate in 100ml LB ampicillin-resistant medium, 37 Cultivate overnight at 200rpm; add 100ml of activated bacterial liquid to 2L LB medium containing Amp resistance for secondary activation, culture at 37℃ for 3-4h until OD600 is 1.2, add 420ml of IPTG (final concentration is 200uM) After induction in a shaker at 30°C for 3 hours, centrifuge at 6000rpm for 5 minutes to collect the bacteria, add 80ml of PBS to resuspend the bacteria, then ultrasonically lyse the bacteria for 30 minutes, collect the supernatant and 4ml of glutathione-agarose by centrifugation as above Gel 4B binding; a large amount of 1A1S_1969 fusion protein containing GST tag was obtained.
[0115] 2. Use the enzyme d...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More