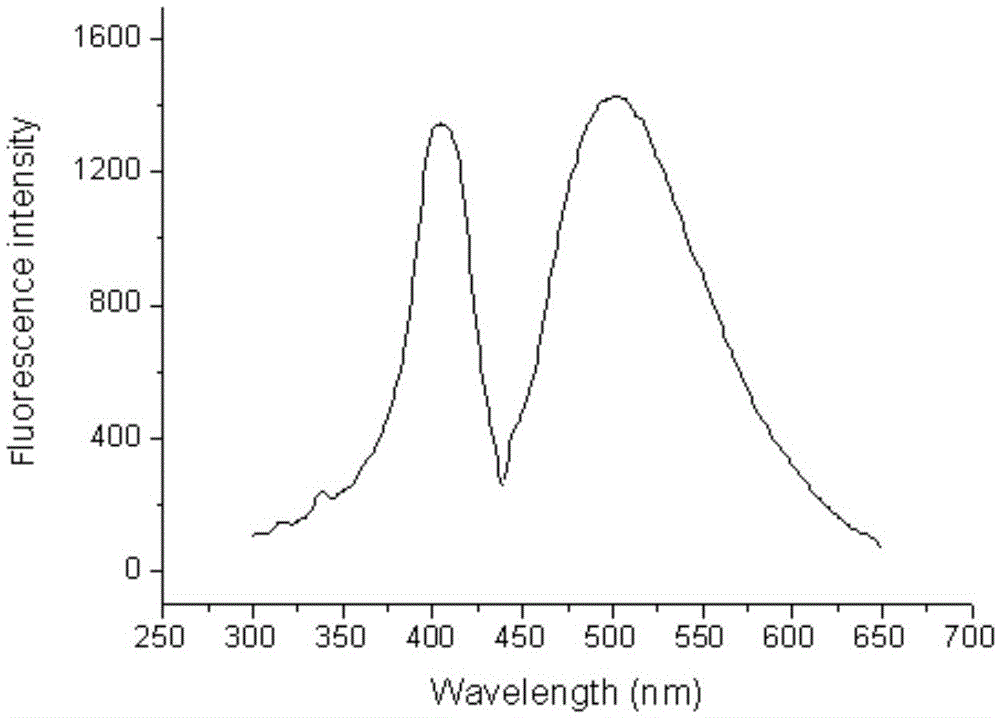
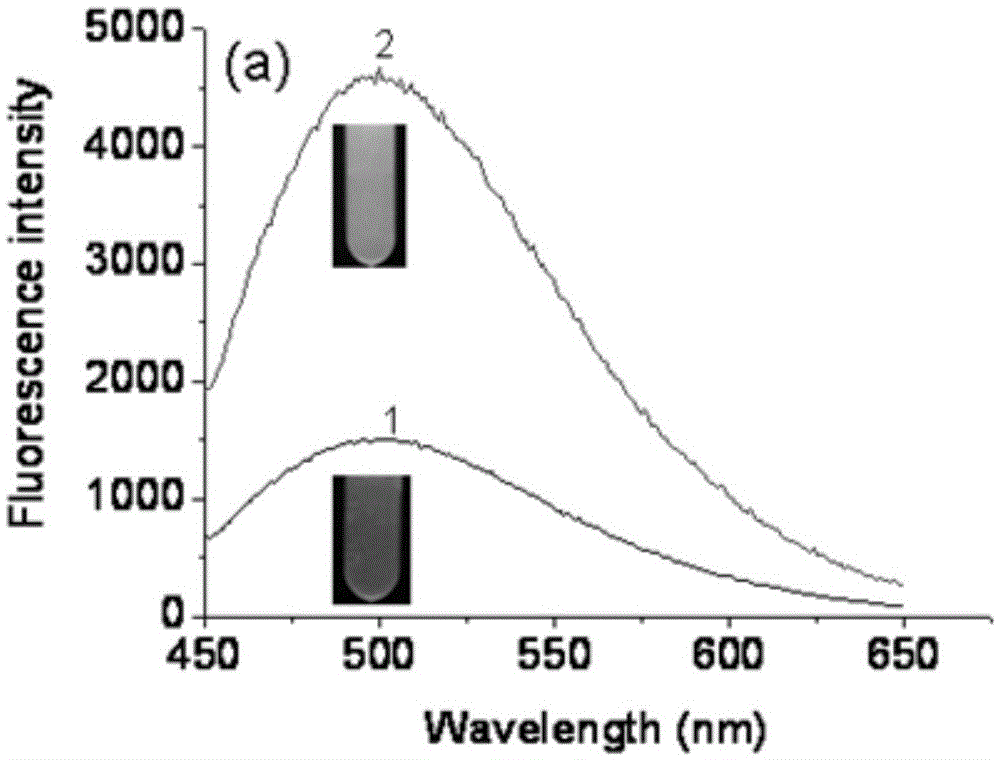
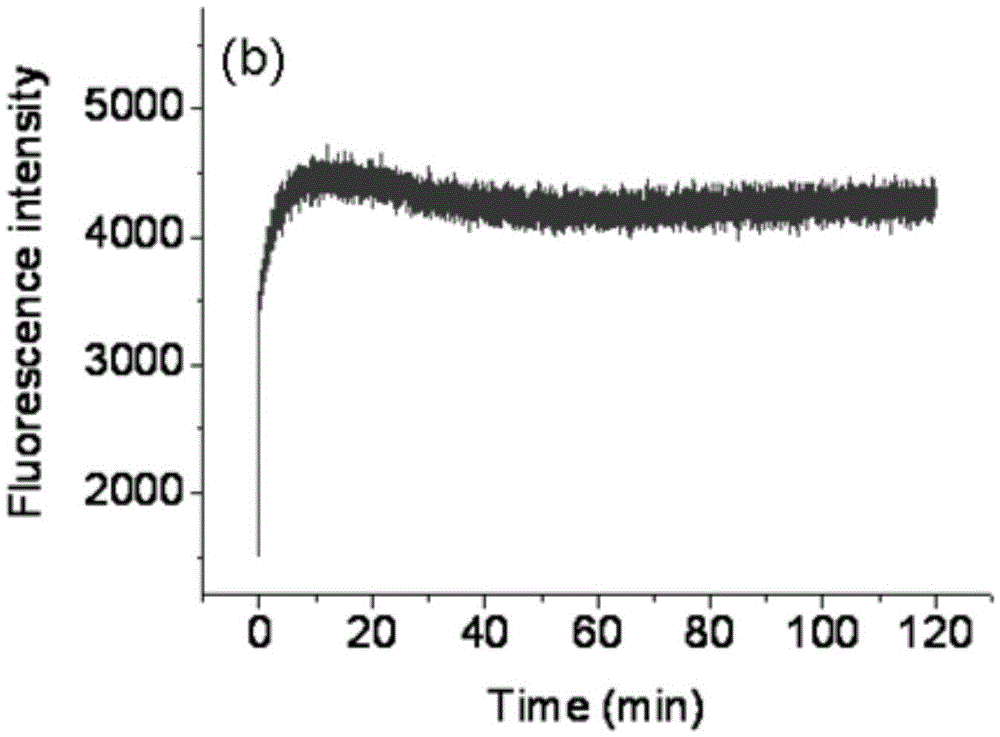
Fluorescent probe for rapidly responding to hydrogen peroxide as well as preparation method and application thereof
A fluorescent probe and fast-response technology, applied in chemical instruments and methods, fluorescence/phosphorescence, and material analysis by optical means, can solve the problem of long response time, high reactivity, and steady-state concentration of probes and hydrogen peroxide Low-level problems, to achieve the effect of good dyeing effect, simple synthesis steps, and high dyeing efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0038] Under the protection of argon, the compound TPE-Br (0.982g, 2mmol) and diboronic acid pinacol ester (1.27g, 5mmol) were dissolved in anhydrous DMF20mL, and anhydrous potassium acetate (1.374g, 14mmol) and pd( dppf)Cl 2 (0.08g, 0.11mmol, react at 85°C for 24h. After the reaction, cool to room temperature, extract the mixture with dichloromethane and water, dry the organic phase with anhydrous magnesium sulfate overnight. Redissolve with dichloromethane, column Chromatographic separation (eluent: petroleum ether: ethyl acetate 15:1, V / V) gave 0.67 g (57%) of a milky white solid.
Embodiment 2
[0040] Under the protection of argon, the compound TPE-Br (1mmol) and diboronic acid pinacol ester (5mmol) were dissolved in anhydrous DMF 20mL, anhydrous sodium carbonate (8mmol) and tetrakistriphenylphosphopalladium (0.11mmol) were added, Reaction at 100°C for 20h. After the reaction was completed, it was cooled to room temperature, the mixture was extracted with dichloromethane and water, and the organic phase was dried overnight with anhydrous magnesium sulfate. It was redissolved with dichloromethane and separated by column chromatography (petroleum ether: ethyl acetate 15:1, V / V) to obtain 0.67 g (57%) of a milky white solid.
Embodiment 3
[0042] Under the protection of argon, the compound TPE-Br (2mmol) and diboronic acid pinacol ester (6mmol) were dissolved in anhydrous DMF 20mL, and anhydrous cesium carbonate (15mmol) and ferrocenetriphenylphosphorous palladium chloride ( 0.11 mmol), reacted at 75°C for 35h. After the reaction was completed, it was cooled to room temperature, the mixture was extracted with dichloromethane and water, and the organic phase was dried overnight with anhydrous magnesium sulfate. It was redissolved with dichloromethane and separated by column chromatography (petroleum ether: ethyl acetate 15:1, V / V) to obtain 0.67 g (57%) of a milky white solid.
[0043] NMR and mass spectrometry characterization:
[0044] 1 HNMR (300MHz, CDCl 3 ,δ):7.54(d,J=1.5Hz,2H),7.51(d,J=1.5Hz,2H),7.06-7.10(m,6H),7.05-6.99(m,8H),1.32(s, 24H)
[0045] 13 CNMR (100MHz, CDCl 3 ,δ):146.80,146.62,143.61,143.45,141.34,134.23,134.14,131.41,130.76,127.79,127.66,126.62,126.55,83.78,24.99
[0046] 11 BNMR (400...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More