ELISPOT detection kit for detecting bovine brucellosis
A detection kit, the technology of Brucella bovis, applied in the field of diagnosis of bovine brucellosis, the field of ELISPOT detection kits for detecting bovine brucellosis, can solve the problem of unsatisfactory specificity and the inability to meet the requirements of bovine Brucella Specific detection of bacterial infection, can not meet the early detection of Brucella bovine infection and other problems, to eliminate the interference of false positives, good specificity, strong affinity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment approach
[0019] As an embodiment of the present invention, the first bovine interferon-gamma monoclonal antibody is bovine interferon-gamma monoclonal antibody 2G5, and the bovine interferon-gamma monoclonal antibody 2G5 is obtained from a hybridoma with a deposit number of CCTCCNO: C2012107 The cell line 2G5 or its passage cell line is secreted; and the second bovine interferon-gamma monoclonal antibody is bovine interferon-gamma monoclonal antibody 5E11, and the bovine interferon-gamma monoclonal antibody 5E11 has a preservation number of CCTCCNO: C2012108 The hybridoma cell line 5E11 or its subcultured cell line is secreted and produced.
[0020] Hybridoma cell line 2G5 and hybridoma cell line 5E11 are disclosed in patent CN102965343B.
[0021] As an embodiment of the present invention, the supporting medium is a microporous membrane filter plate. More preferably, the supporting medium is a PVDF film-coated microwell culture plate. Wherein, preferably, there is a PVDF membrane on ...
Embodiment 1
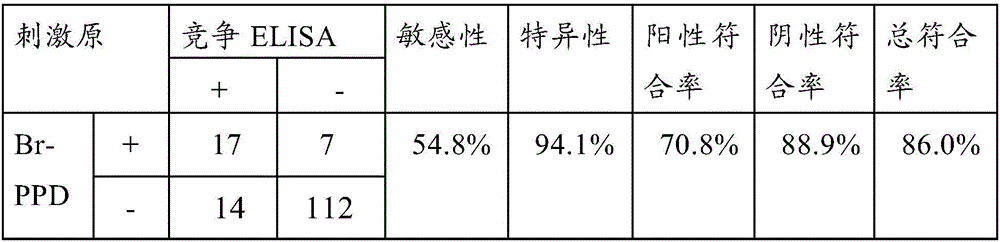
[0047] Embodiment 1 The preparation of the bovine brucellosis ELISPOT detection kit based on Br-PPD of the present invention
[0048] The ELISPOT kit assembly steps are as follows:
[0049] 1) Purification of bovine gamma interferon monoclonal antibody 2G5
[0050] The hybridoma cell line secreting bovine gamma interferon monoclonal antibody 2G5 was injected into the peritoneal cavity of mice. After ascites was produced, the ascites was collected, and the collected 2G5 ascites was purified by ProteinG affinity chromatography.
[0051] 2) Preparation of biotin-labeled bovine interferon-γ monoclonal antibody (referred to as Bio-5E11)
[0052] The hybridoma cell line secreting bovine gamma interferon monoclonal antibody 5E11 was injected into the peritoneal cavity of mice. After ascites was produced, the ascites was collected, and the collected 5E11 ascites was purified by ProteinG affinity chromatography.
[0053] Label the purified 5E11 monoclonal antibody with standard bioti...
Embodiment 2
[0064] Embodiment 2 detects bovine peripheral blood sample based on the ELISPOT detection kit of non-specific stimulant PWM
[0065] 1. Experimental materials
[0066] The bovine brucellosis ELISPOT detection kit prepared in Example 1 was used.
[0067] 2. Anticoagulation incubation and detection
[0068] ① Sterilely take 1mL of bovine blood and add it into blood collection tubes containing sodium heparin, and mix it upside down after blood collection to obtain anticoagulant blood;
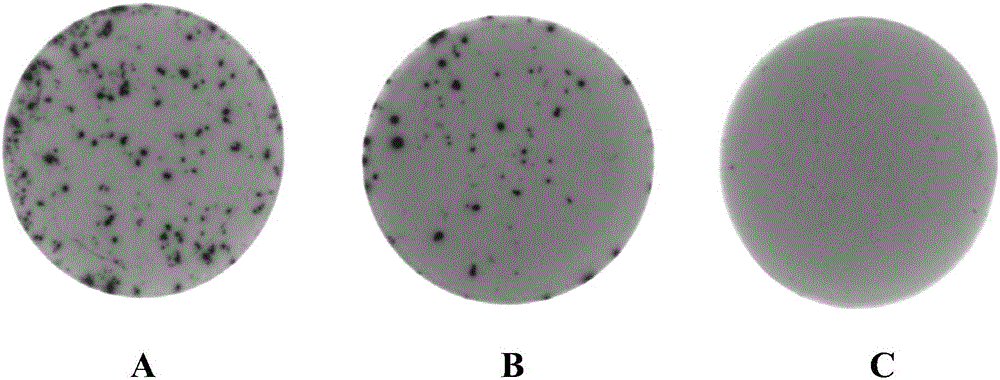
[0069] ②Add the following reagents to the above-mentioned coated 96-well filter plate: each anticoagulant sample includes 2 test wells, 50 μL of cell culture solution to the negative control well, 50 μL of PWM diluted in the cell culture solution to the positive control well (final Concentration is 5 μg / mL), 50 μL bovine anticoagulant blood was added to each well, and the 96-well filter plate was placed at 37 ° C, 5% CO 2 Cultivate in the incubator for 24-48 hours;
[0070] ③Take out the 96-we...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More