Synthesis method of fluxapyroxad based on Suzuki reaction
A technology of fluconazole and a synthesis method, which is applied in the field of organic chemical synthesis of pesticide molecules, can solve the problems of high safety risk of fluconazole and the like, and achieves the effects of great commercial value and potential, low preparation cost and mild reaction conditions.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment approach
[0017] According to one embodiment of the present invention, the synthesis method comprises the following steps:
[0018] S1. Condensation of 3-difluoromethyl-1-methyl-1-piperazole-4-carboxylic acid with 2-iodoaniline in organic solvent under the action of coupling reagent and 4-dimethylaminopyridine Reaction to generate 3-difluoromethyl-N-(2-iodophenyl)-1-methyl-1-piperazole-4-amide;
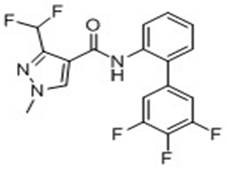
[0019] S2.3-Difluoromethyl-N-(2-iodophenyl)-1-methyl-1-piperazole-4-amide occurs in organic solvents in the presence of palladium metal catalysts and alkaline reagents Suzuki reaction to 3-difluoromethyl-1-methyl-N-[3'-4'-5'-trifluoro-(1,1'-biphenyl)-2-yl]-1-piperene Azol-4-amide, the product of flufenapyramide.
[0020] Preferably, in step S1, the coupling reagent is 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride, and the organic solvent is dichloromethane; in step S2 Among them, the palladium metal catalyst is tetrakis(triphenylphosphine) palladium, the basic reagent is potass...
Embodiment 1
[0030]
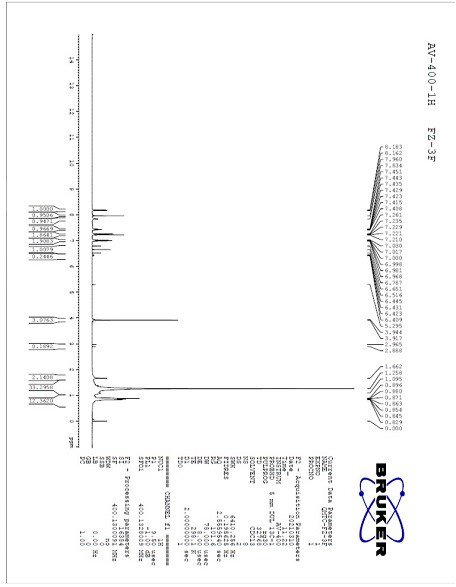
[0031] Take a round bottom flask with a built-in stirring bar. To this was added 3-difluoromethyl-1-methyl-1-pyrazole-4-carboxylic acid (1.00 g, 5.70 mmol), 2-iodoaniline (1.50 g, 6.84 mmol), 1-(3-di Methylaminopropyl)-3-ethylcarbodiimide hydrochloride (1.20 g, 6.27 mmol), 4-dimethylaminopyridine (0.84 g, 6.84 mmol) and dichloromethane (20 ml). The resulting mixture was stirred overnight at room temperature. After the completion of the reaction was monitored by TLC, distilled water (20 ml) was added to the reaction system. The organic phase was separated and the aqueous phase was washed 3 times with dichloromethane (20 ml). The organic phases were combined and evaporated to dryness, the resulting crude product was purified by column chromatography to give 3-difluoromethyl-N-(2-iodophenyl)-1-methyl-1-pyrazole- The 4-amide is pure, with a yield of 81%, and a purity calculated by H NMR spectrum of 91%.
Embodiment 2
[0033]
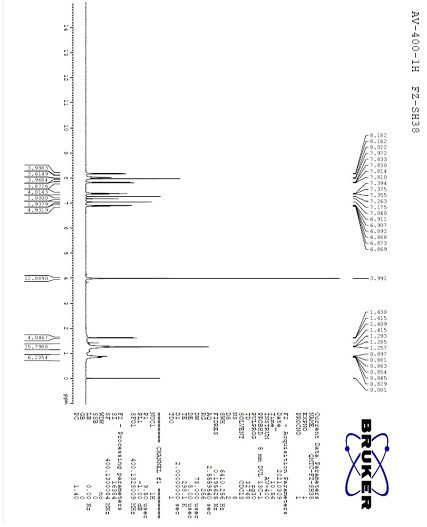
[0034] Take a round bottom flask with a built-in stirring bar. To this was added 3-difluoromethyl-N-(2-iodophenyl)-1-methyl-1-piperazole-4-amide pure (0.31 g, 0.82 mmol), 3,4,5-tris Fluorophenylboronic acid (0.17 g, 0.98 mmol), tetrakis(triphenylphosphine)palladium (300 mg), potassium carbonate (0.57 g, 4.10 mmol) and tetrahydrofuran (9 ml). The resulting mixture was heated to reflux under nitrogen atmosphere and reacted overnight. After the reaction was detected by thin layer chromatography, water (20 ml) and dichloromethane (50 ml) were added thereto, and stirred for 30 minutes. The organic phase was separated, washed with saturated ammonium chloride solution (10 ml), dried and concentrated to dryness. The obtained crude product was purified by column chromatography to obtain pure flufenapyroxad as a light yellow solid with a yield of 92% and a purity of 95% calculated by H NMR spectroscopy.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More