[0005] ①This
emulsion is a heterogeneous liquid preparation, which is a thermodynamically and kinetically unstable
system. Therefore, when the
emulsion is placed for a long time, the
emulsion droplet particles are easy to merge to increase the particle size, or cause
instability such as oil floating and layering of the emulsion;
[0007] ③ During the high
temperature treatment in the preparation process and the long-term storage process of this emulsion, the drugs,
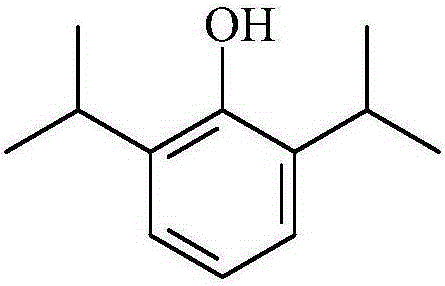
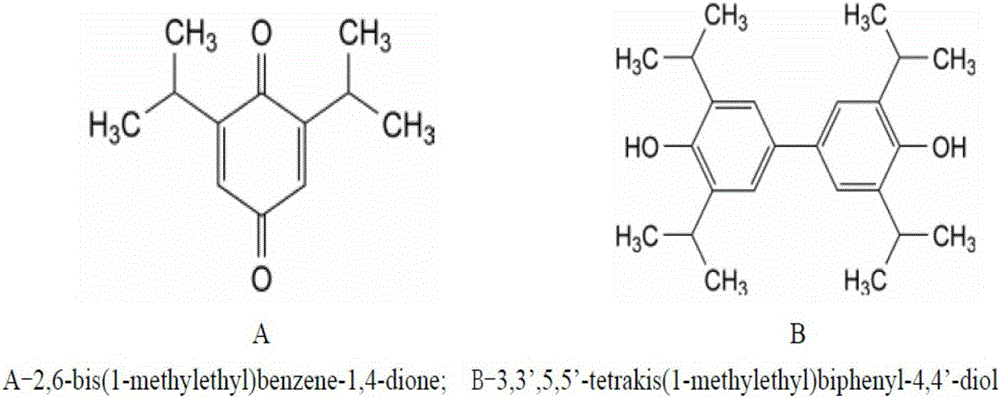
soybean oil and lecithin in the emulsion are easily oxidized and degraded, thus causing allergies and other side effects: there are phenolic hydroxyl groups in the structure of propofol, which are easily destroyed Oxidation to generate 2,6-diisopropyl-1,4-
benzoquinone or
polymerization to generate 3,3,5,5'-tetraisopropyl biphenol (see
chemical structure figure 2 ), these two impurities have greater
toxicity;
[0008] ④ In order to prevent propofol from being oxidized, auxiliary materials such as EDTA-2Na,
sulfite, and
thiosulfate are often added to the preparation, but these auxiliary materials are also likely to cause allergic reactions, especially for patients with idiopathic
asthma;
[0009] ⑤ The fat, phospholipids and other substances in this preparation are good substrates for microbial production in a liquid environment, so there is a risk of bacterial and fungal infection during storage and use, and if used improperly, it is easy to cause consequences such as infection;
[0010] ⑥ The incidence of pain at the
injection site of this preparation is as high as 28% to 90%, which has seriously affected the patients;
[0011] ⑦ This emulsion is likely to cause
hypertriglyceridemia after intravenous administration, especially for patients who receive long-term infusion;
[0012] ⑧ This preparation should avoid high and low temperature storage, and at the same time, it should not be shaken violently, so this product is not convenient for transportation and storage
[0014] The
patent application No. 201510021939.0 prepared propofol
fat emulsion injection, which overcomes the defects of excessive particle size deviation of
colostrum, easy emulsion breaking, and excessive particle size deviation of emulsion after high-pressure homogenization, but it uses natural Egg
yolk lecithin and
soybean oil are used as excipients, which can easily cause allergic reactions
[0015] The
patent application number is 201410819727.2 prepared propofol fat emulsion injection, which does not contain
preservative disodium edetate, which reduces the toxic and side effects of the preparation, but the natural egg
yolk lecithin and soybean oil it uses still cause allergies Phenomenon
[0016] The patent with authorized notification number CN101006992B prepares propofol freeze-dried milk, which increases the stability of the emulsion, but it uses natural egg
yolk lecithin and soybean oil as auxiliary materials, which can easily cause allergic reactions
[0017] The
patent application No. 201410130319.6 prepared propofol liposomes, which have the advantages of high encapsulation efficiency, high
drug loading, and
small particle size, but it uses emulsifier Tween-80 and natural egg yolk lecithin, which is easy to
allergic reaction[0018] The patent with authorized announcement number CN102805728B prepared a kind of propofol fat emulsion injection, which solved the problem of
large particle size and easy demulsification of propofol fat emulsion, and at the same time alleviated the pain of injection, but it uses Tween-80 and Natural egg yolk lecithin and soybean oil, prone to allergic reactions
[0019] The patent application No. 201210419120.6 has prepared a stable and efficient propofol medium / long-chain fat emulsion injection, which is more stable than ordinary propofol fat emulsion, and at the same time, can significantly reduce the incidence of injection pain and improve Improve
patient compliance, but the natural egg yolk lecithin it contains is prone to allergic reactions
[0020] The patent with authorized notification number CN102552136B has prepared a medium /
long chain injection of propofol, which does not contain
preservative disodium edetate, which reduces the
irritation of the preparation, but it uses natural egg yolk lecithin and large
Soybean oil can still cause allergies
[0021] The patent application number is CN104288130A has prepared a kind of propofol composition for injection, and it increases propofol in preparation by adding cosolvent
propylene glycol,
nonionic surfactant Tween 20, Tween 80,
polyoxyethylene castor oil etc. The encapsulation rate in the medium reduces the injection pain caused by free propofol, but the excipients it adds will produce allergic
irritation Login to View More
Login to View More  Login to View More
Login to View More