Isoindigo derivative polymer with butadiene as space isolation group, preparation and use thereof
A space isolation, butadiene technology, applied in semiconductor/solid-state device manufacturing, photovoltaic power generation, electrical components and other directions, can solve the problems of IID distortion, high TIIHOMO energy level, reduce dihedral angle, etc., to achieve low synthesis cost, Simple and effective synthesis method
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
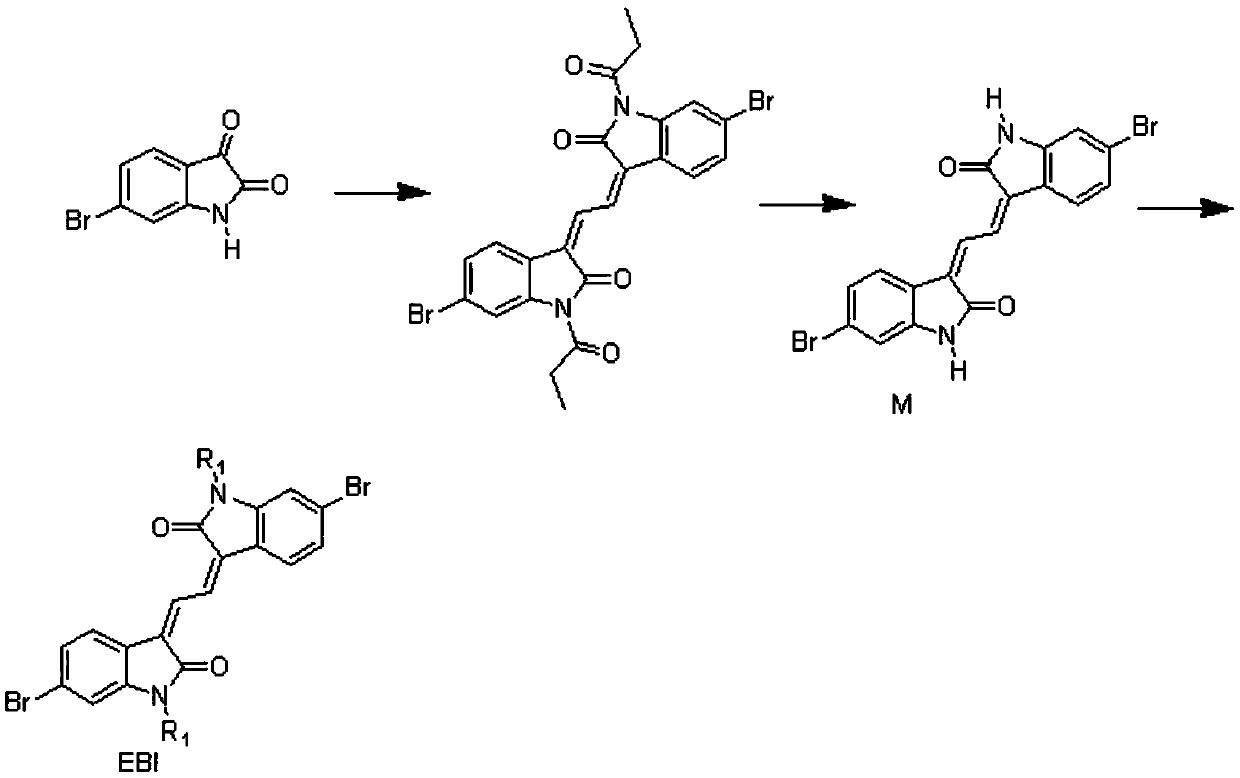
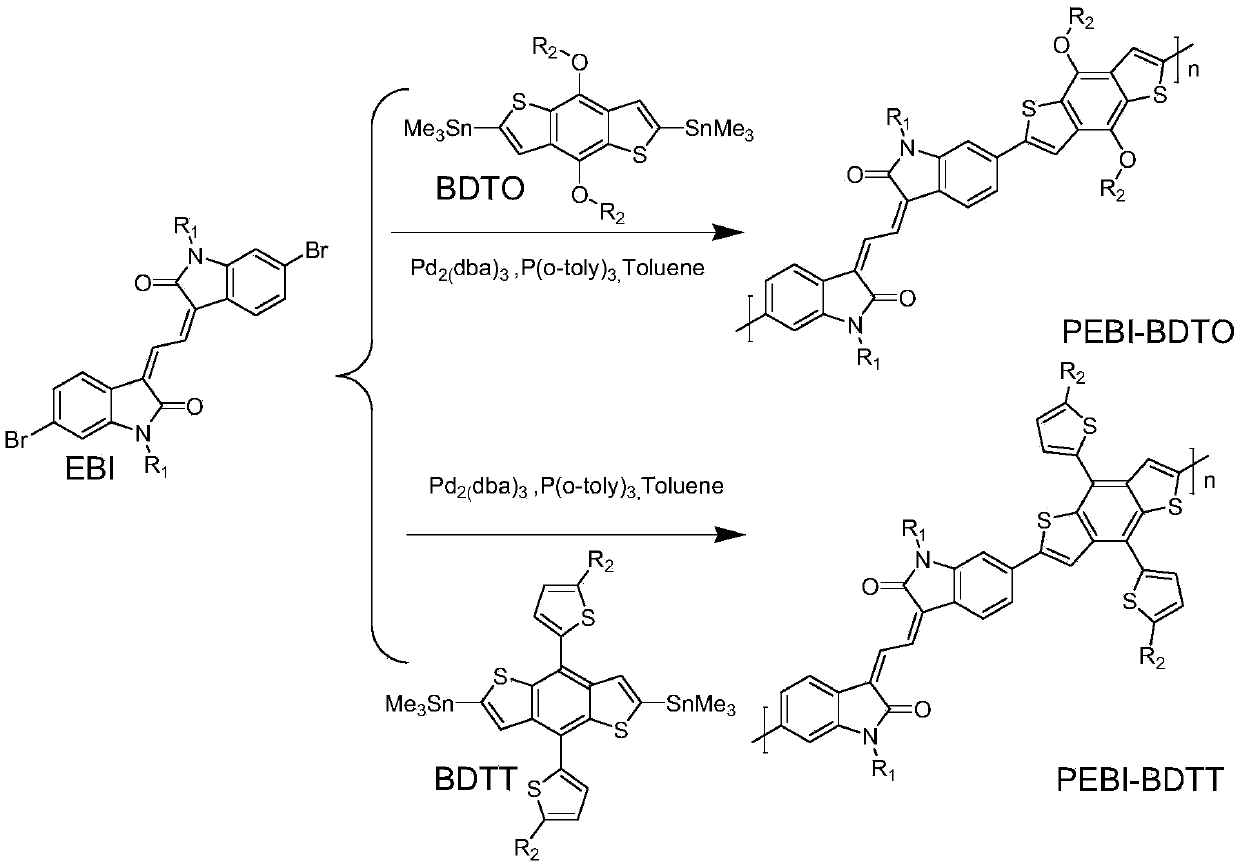
[0031] This embodiment provides soluble extended isoindigo EBI-based conjugated polymers PEBI-BDTO and PEBI-BDTT, the structural formula of which is shown in Table 1 (wherein, 100>n>1), and the synthetic route of its monomer EBI see figure 1 , the synthesis of polymers PEBI-BDTO and PEBI-BDTT see figure 2 shown.
[0032]
[0033]
[0034] This embodiment also relates to the preparation method of polymer PEBI-BDTO and PEBI-BDTT, comprising the following steps:
[0035] (a) Synthesis of intermediate compound benzodithiophene bistin monomer
[0036] The structural formula of benzodithiophene ditin monomer is
[0037]
[0038] For its detailed preparation method, see the literature "Hwang, Y.J; Kim, F.S; Xin, H; Jenekhe, S.A; New Thienothiadiazole-Based Conjugated Copolymers for Electronics and Optoelectronics. ;FullereneDerivative-DopedZincOxideNanofilmastheCathodeofInvertedPolymerSolarCellswithLow-BandgapPolymer(PTB7-Th)forHighPerformance.Adv.Mater.2013,25,4766–477...
Embodiment 2
[0053] This example relates to the ultraviolet absorption spectrum and electrochemical properties of the polymers PEBI-BDTO and PEBI-BDTT of Example 1. Figure 5 The UV spectral properties of polymers in chloroform solution are given, and the λ of polymers PEBI-BDTO and PEBI-BDTT in chloroform solution UV-vis max abs Very close, 663 and 668nm respectively. Figure 6 The ultraviolet spectral properties of the polymer film state are given. Compared with the solution, the UV-vis of the film is red-shifted, λ max abs are 667nm and 676nm, red-shifted by 4nm and 8nm, respectively. Figure 7 The cyclic voltammetry curves of the compound polymers are given. The cyclic voltammetry test is carried out on the computer-controlled CHI610D telephone line analyzer, using the traditional three-electrode test system, the platinum electrode is the working electrode, the silver / silver ion electrode is the reference electrode, and the electrolyte is tetra-n-butyl hexafluorophosphoric acid Am...
Embodiment 3
[0055] This example relates to the use of the polymers PEBI-BDTO and PEBI-BDTT of Example 1 as active layer materials in thin-film solar cells. Using polymers PEBI-BDTO and PEBI-BDTT as donor materials (D), fullerene (PC 71 BM) As the acceptor material (A), the photovoltaic performance of the material was tested using the bulk heterojunction structure. The test conversion efficiency data of polymer solar cell devices is shown in Table 2, and the I-V curve is shown in Figure 8 . The highest conversion efficiency of PNDFT-DTBT in the preliminary test is 4.59%, and the polymer has high open circuit voltage and fill factor.
[0056] Table 2
[0057]
[0058] In summary, the conjugated polymer of isoindigo derivatives in which butadiene is a spacer group involved in the present invention has good planar conjugation and good ultraviolet spectrum absorption. The material can be solution-processed and has a narrow energy band gap, good UV absorption and good thermal stability....
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com