Broad-spectrum safe anti influenza A virus vaccine for animals
A type of influenza A virus and vaccine technology, applied in the field of genetic engineering, can solve the problems of long production cycle, leakage and spread of live virus, and high production conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 2
[0128] Example 2: Construction of insect baculovirus expression plasmids expressing M1, HA, NA and 3xM2eNP genes and synthesis of recombinant insect baculoviruses in insect cells.
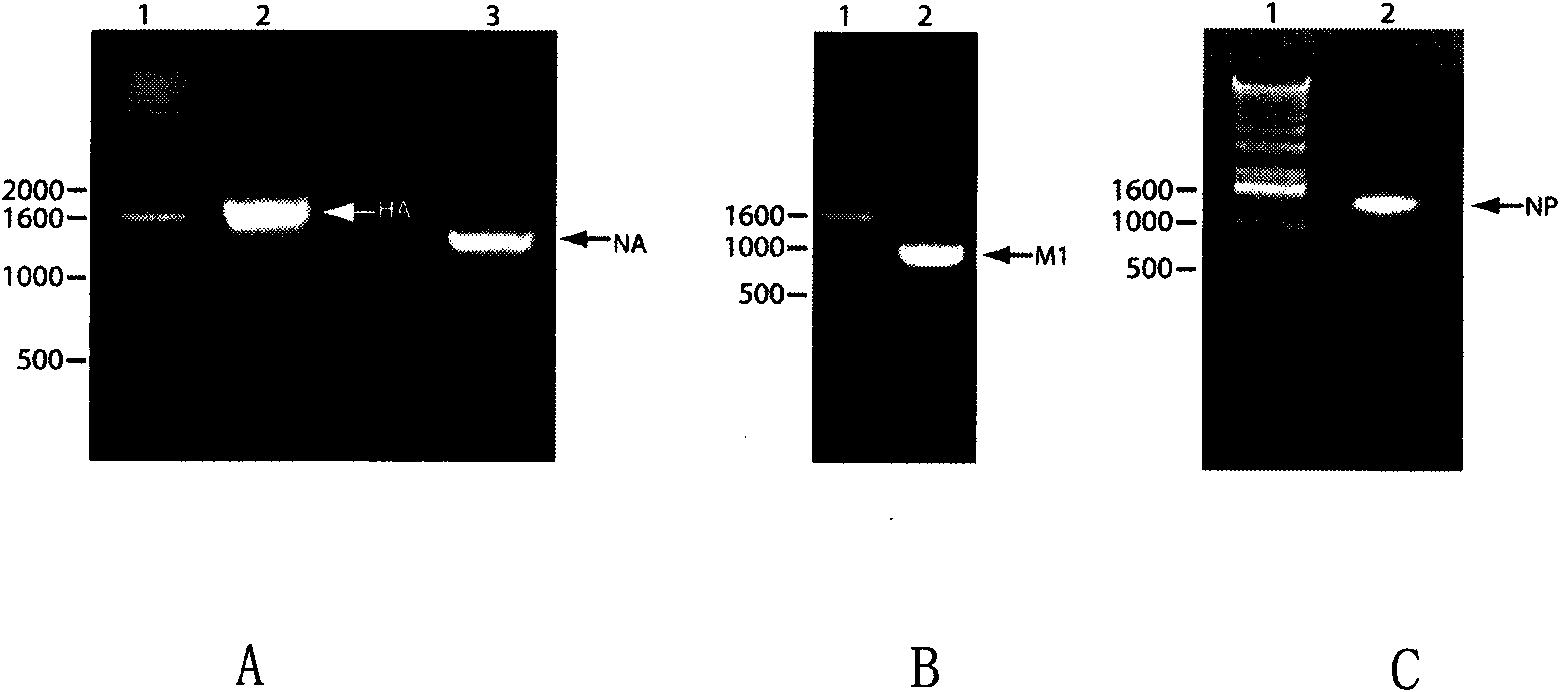
[0129] (1) Construction of a recombinant plasmid expressing the M1 gene: Insect baculovirus expression plasmid pFastBac-Dual (product of Invitrogen Company) was digested and hydrolyzed by restriction endonuclease BamHI / NotI overnight at 37°C, then gel electrophoresis and column extraction purification. Under the action of T4 DNA ligase, the digested plasmid and the digested M1 gene DNA fragment were ligated overnight at 16°C. The reaction system is as follows: 1ul of 10×T4 ligation buffer, 3ul of the DNA fragment recovered by M1 digestion, 1ul of the recovered product of pFastBac-Dual plasmid, 1ul of T4 DNA ligase, and 10ul of ddH2O. The ligation product was transformed into E.coli DH5a competent bacteria by heat shock method. The specific operation is as follows: transfer 50ul DH 5a competent ce...
Embodiment 3
[0138] Example 3: Expression and analysis of M1, HA, NA and fusion protein 3xM2eNP genes in co-transfected suspension cultured insect cells Sf-21.
[0139] (1) Gel protein electrophoresis to detect the expression of the M1 gene: the cell centrifugation precipitate used to amplify the culture of the recombinant baculovirus Bac-M1 was treated with the cell lysis buffer, centrifuged at 4°C (13,000rpm) for 10 minutes, and collected The supernatant was subjected to gel electrophoresis to analyze whether the matrix protein M1 was expressed in the insect cell Sf-21. Briefly, 10ul of 2XSDS loading buffer was added to 10ul of cell lysate supernatant, treated at 100°C for 5min, and then centrifuged rapidly for 5 seconds. All 20ul of mixed samples were added to 4%-12% SDS-polyacrylamide gel (Invitrogen company product) spotting wells. Set the electrophoresis conditions as a constant voltage of 100V, a temperature of 4°C, and a time of 2 hours. The electrophoresis buffer was Tris-glycin...
Embodiment 4
[0142] Example 4: The intervention of nucleoprotein NP can increase the yield of virus-like particles.
[0143] As mentioned above, nucleoprotein NP can couple with matrix protein M1, and can bring the M1 protein that re-enters the host cell nucleus back to the cytoplasm [Document 14]. The synthesis amount of M1 protein directly determines the formation amount of influenza A virus granules [References 5, 6]. Recombinant insect baculovirus Bac-M1 and Bac-HANA with known virus titers were co-transfected into insect cell Sf-21. At the same time, the recombinant insect baculoviruses Bac-M1, Bac-HANA and Bac-3xM2eNP with known virus titers were co-transfected into insect cells Sf-21 in another shake flask. After culturing at 27°C for three days, the cell supernatants in the two shake flasks were collected, and the HA titer of the virus-like particles was detected by microplate counting method. The specific operation is as follows: PBS is added to each well of a 96-well cell cultu...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More