Pharmaceutical composition containing oseltamivir phosphate and preparation method thereof
A technology of oseltamivir phosphate and composition, which is applied in the field of pharmaceutical composition containing oseltamivir phosphate and its preparation, can solve the problems of complex preparation process, poor patient compliance, poor product stability, etc., and achieve simple preparation process , High patient compliance and good stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0022] The first aspect of the embodiment of the present application provides a preparation method of a pharmaceutical composition containing oseltamivir phosphate, comprising the following steps:
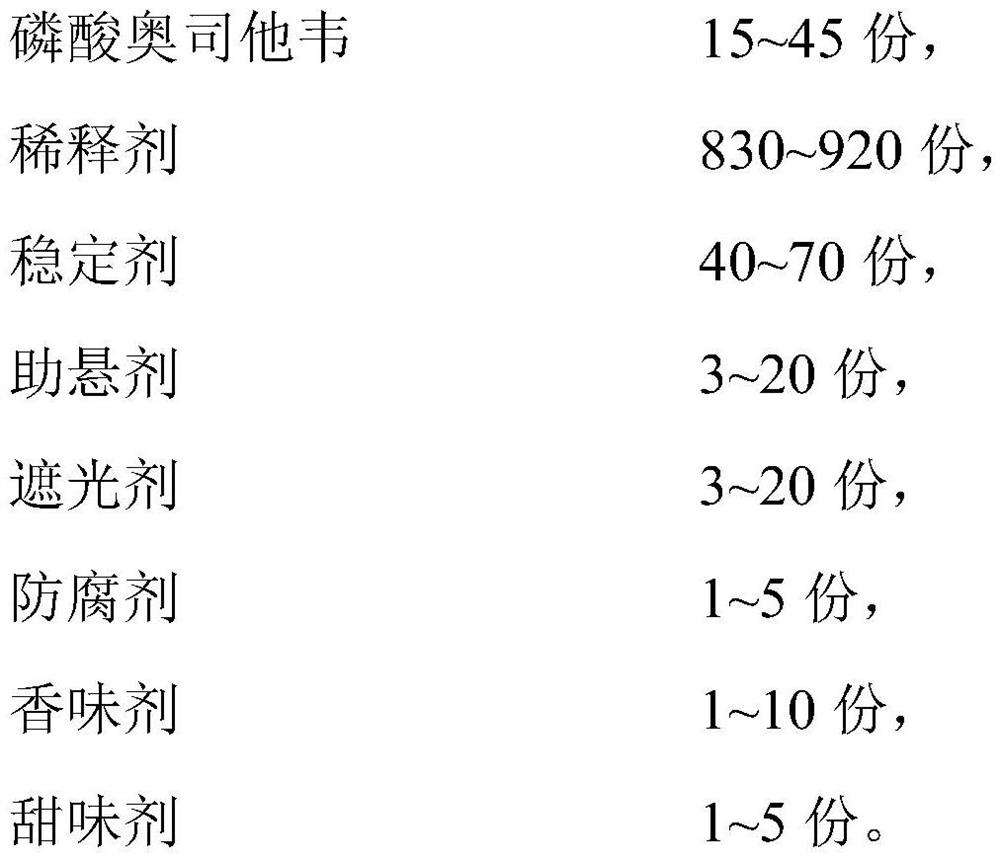
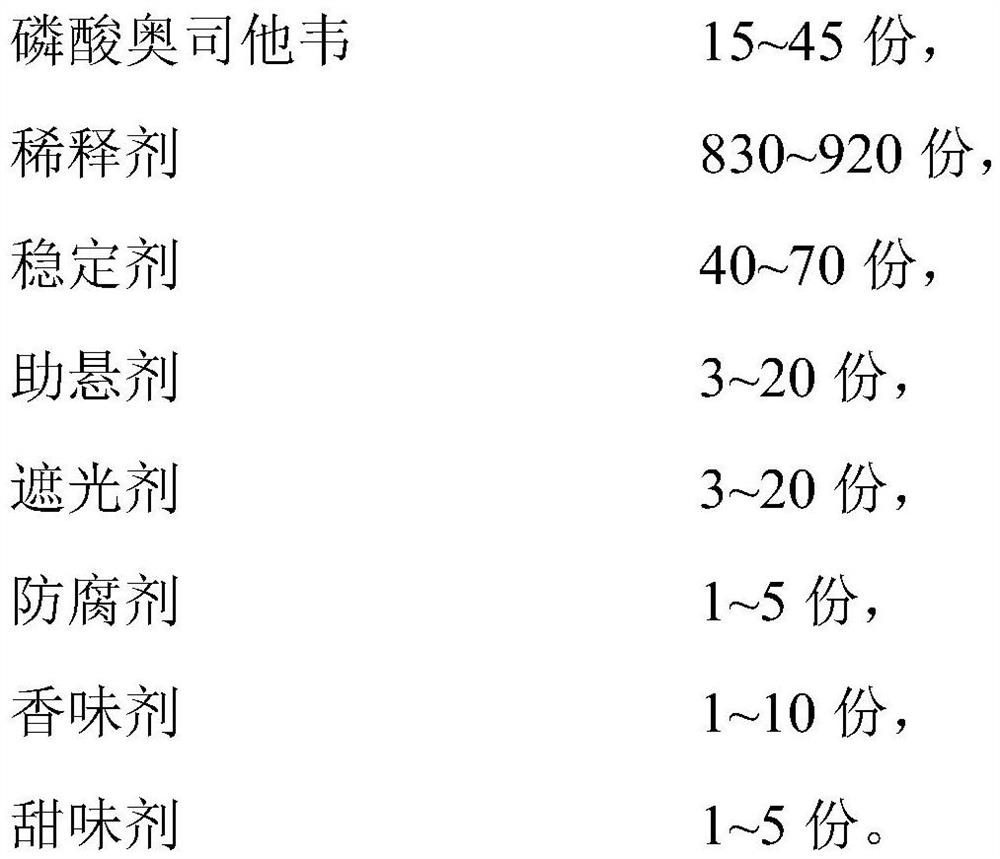
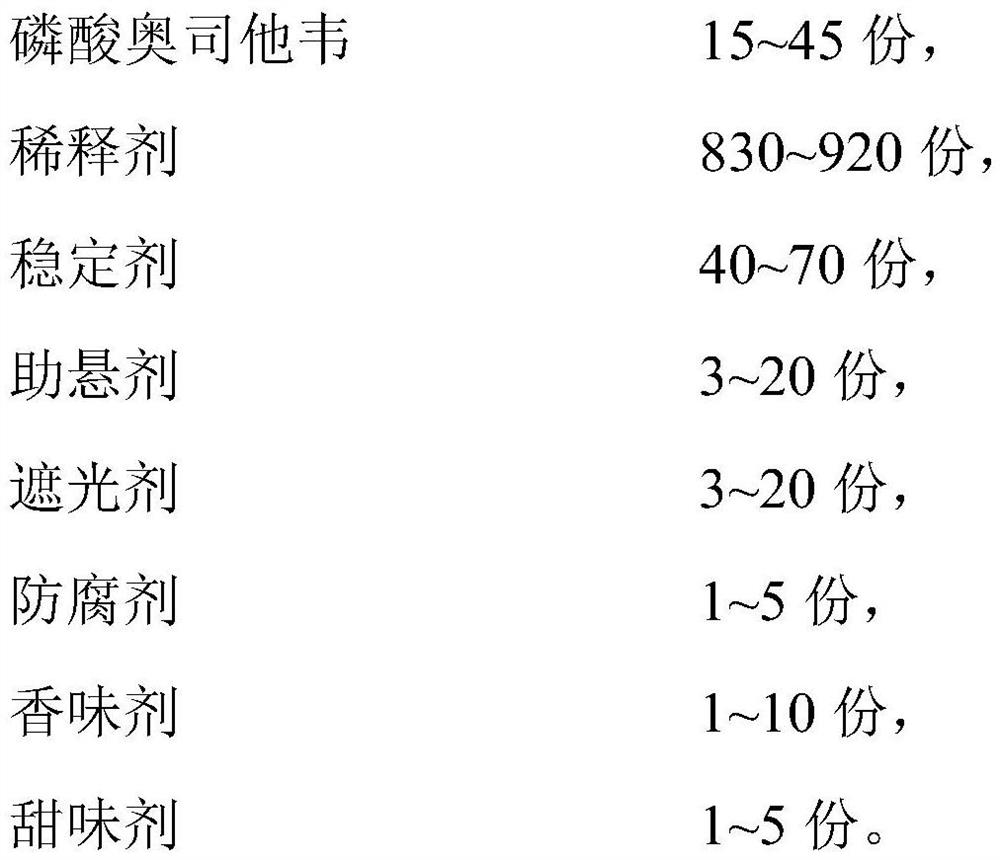
[0023] S10. Perform a first mixing treatment on 15-45 parts of oseltamivir phosphate, 40-70 parts of a stabilizer and a first diluent to obtain a first mixture;
[0024] S20. Mix 3-20 parts of suspending agent, 3-20 parts of opacifying agent, 1-5 parts of preservative, 1-10 parts of flavoring agent and 1-5 parts of sweetener with the second diluent second mixing treatment to obtain a second mixture;
[0025] S30. Perform a third mixing process on the first mixture, the second mixture and the third diluent to obtain a pharmaceutical composition containing oseltamivir phosphate;
[0026] Wherein, the total mass parts of the first diluent, the second diluent and the third diluent are 830-920 parts.
[0027] In the preparation method of the pharmaceutical composition containing oselt...
Embodiment 1
[0057] A pharmaceutical composition (in bags) containing oseltamivir phosphate, comprising the following table 1 raw material components:
[0058] Table 1
[0059]
[0060] Its preparation includes steps:
[0061] 1. Dilute the prescribed amount of oseltamivir phosphate (D 90 380 μm), monosodium citrate and 3000g sorbitol (about 1 / 3 prescription quantity) pass through a 60-mesh sieve and mix together to obtain mixture 1;
[0062] 2. Put the prescription amount of xanthan gum, titanium dioxide, sodium saccharin, sodium benzoate, orange flavor and 1000 g of sorbitol (about 12% of the prescription amount) through a 60-mesh sieve and mix to obtain mixture 2;
[0063] 3. Finally, add mixture 1, mixture 2 and 4706g sorbitol into a three-dimensional mixer, and mix at a speed of 15rpm for 20min to obtain a pharmaceutical composition containing oseltamivir phosphate.
[0064] 4. Use a granule packaging machine to pack the mixed powder, use aluminum foil bags, each bag contains 1....
Embodiment 2
[0068] A pharmaceutical composition (bottled) containing oseltamivir phosphate, comprising the following table 2 raw material components:
[0069] Table 2
[0070]
[0071] Its preparation includes steps:
[0072] 1. Dilute the prescribed amount of oseltamivir phosphate (D 90 150 μm), monosodium citrate and 3000g sorbitol (about 1 / 3 prescription quantity) pass through a 60-mesh sieve and mix together to obtain mixture 1;
[0073] 2. Put the prescription amount of xanthan gum, titanium dioxide, sodium saccharin, sodium benzoate, orange flavor and 1000 g of sorbitol (about 12% of the prescription amount) through a 60-mesh sieve and mix to obtain mixture 2;
[0074] 3. Finally, add mixture 1, mixture 2 and 4706g sorbitol into a three-dimensional mixer, and mix at a speed of 15rpm for 20min to obtain a pharmaceutical composition containing oseltamivir phosphate.
[0075] 4. Use a granule packaging machine to sub-package the mixed powder, use glass bottles, each bottle has a ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More