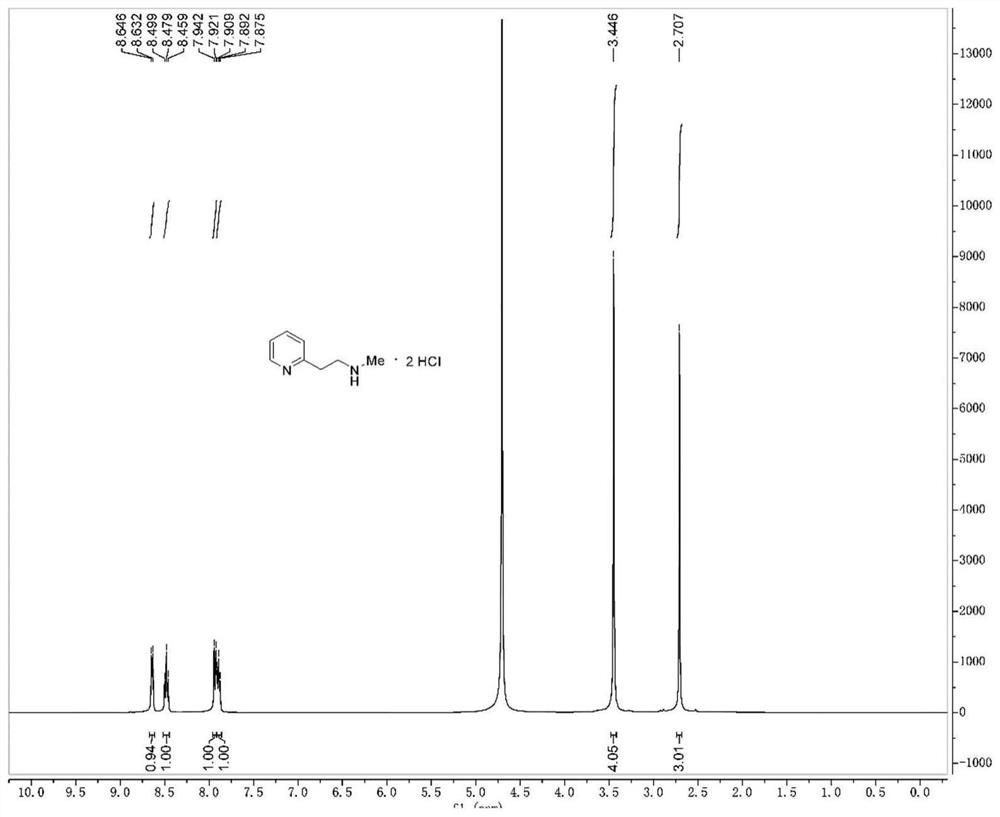
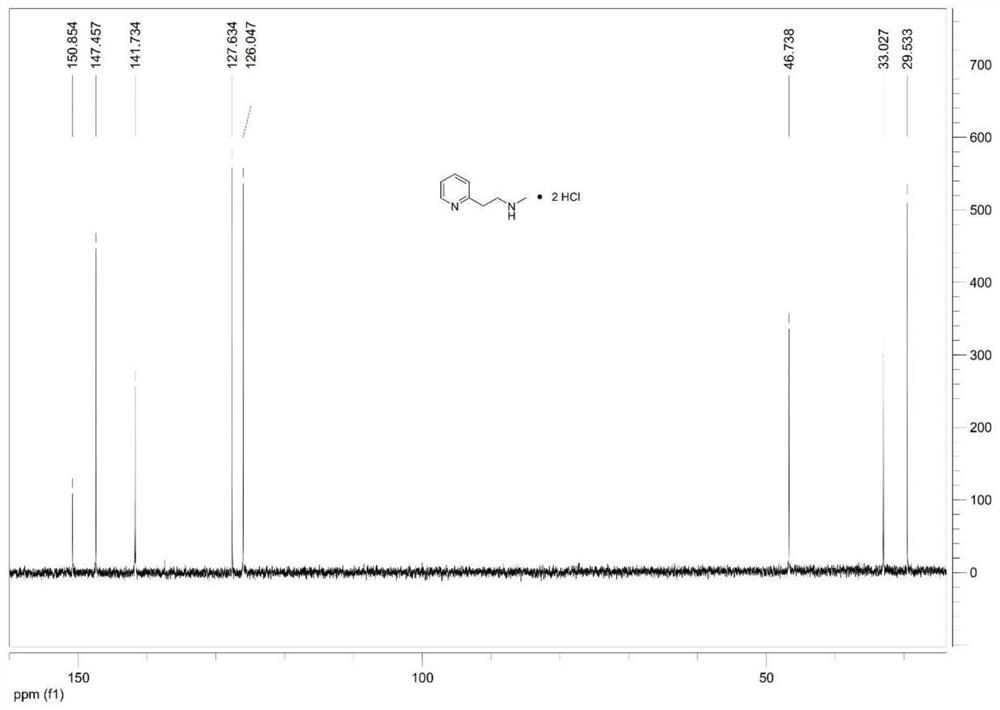
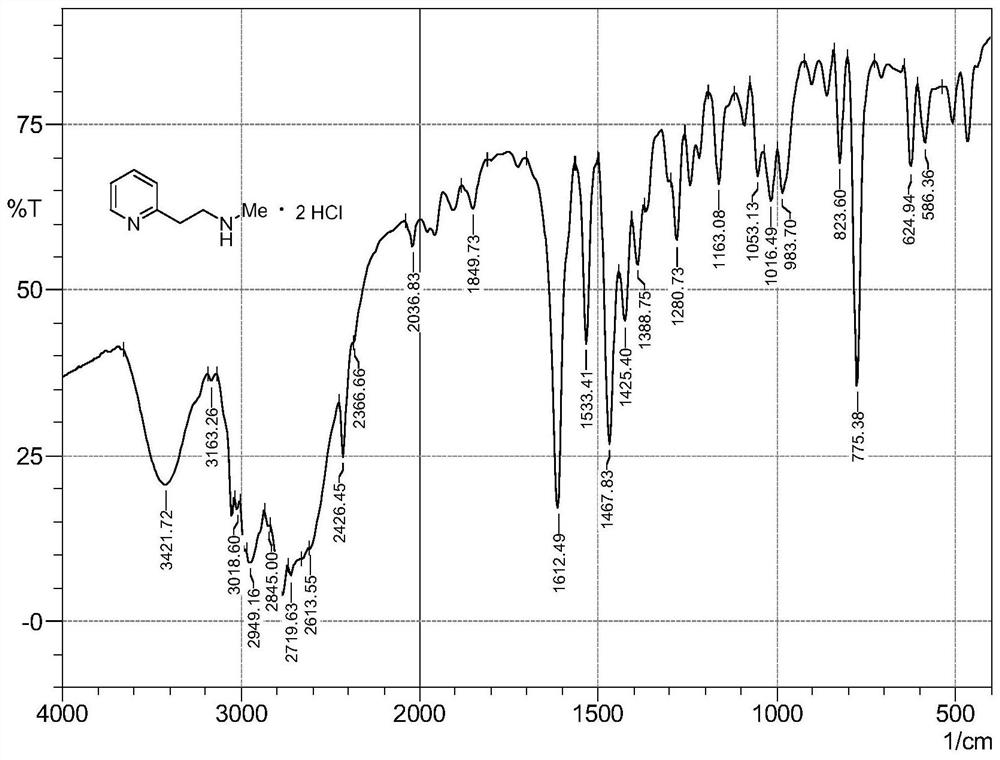
Synthesis method of betahistine hydrochloride
A technology of betahistine and a synthesis method, applied in the field of synthesis of betahistine hydrochloride, can solve the problems of difficult preparation, medium yield, unsuitable for industrialized production and the like, and achieves easy industrialized production, short reaction route and product yield. High efficiency and purity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030] At room temperature, sequentially add 0.06g 5% Pd / C, 15.90g (0.15mol) sodium carbonate, 8.10g (0.12mol) methylamine hydrochloride into a 500mL three-necked flask, depressurize the air and fill it with nitrogen, repeat Three times, the air was replaced with nitrogen. Under nitrogen protection, 12.32 g (0.1 mol) of 2-hydroxyethylpyridine and 280 mL of toluene were injected sequentially. After the addition is complete, heat to 110°C and react for 15h. The reaction solution was cooled to room temperature, 100 mL of water was added, the transition metal catalyst was recovered by suction filtration, and the catalyst was washed with a small amount of ethyl acetate. The filtrate was separated into the organic phase, and the aqueous phase was extracted with ethyl acetate (3 x 30 mL). The organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, and the organic solvent was recovered by distillation under reduced pressure. The residue was d...
Embodiment 2
[0032] At room temperature, sequentially add 0.12g of 5% Pd / C, 15.90g (0.15mol) of sodium carbonate into a 500mL three-neck flask, depressurize the air and fill it with nitrogen, repeat three times, and replace the air with nitrogen. Under nitrogen protection, 13.7mL (0.16mol) of 40% methylamine aqueous solution, 12.32g (0.1mol) of 2-hydroxyethylpyridine and 210mL of toluene were injected sequentially. After the addition is complete, heat to 90°C and react for 18h. The reaction solution was cooled to room temperature, 100 mL of water was added, the transition metal catalyst was recovered by suction filtration, and the catalyst was washed with a small amount of ethyl acetate. The filtrate was separated into the organic phase, and the aqueous phase was extracted with ethyl acetate (3 x 30 mL). The organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, and the organic solvent was recovered by distillation under reduced pressure. The resi...
Embodiment 3
[0034] At room temperature, successively add 0.25g nano-nickel powder, 13.60g (0.20mol) sodium formate, 8.10g (0.12mol) methylamine hydrochloride into a 500mL three-necked flask, decompress and extract the air and fill it with nitrogen, repeat three times, use Nitrogen replaced the air. Under nitrogen protection, 12.32 g (0.1 mol) of 2-hydroxyethylpyridine and 280 mL of benzene were injected sequentially. Addition is complete, heated to 80 ° C, the reaction 16h. The reaction solution was cooled to room temperature, 100 mL of water was added, the transition metal catalyst was recovered by suction filtration, and the catalyst was washed with a small amount of ethyl acetate. The filtrate was separated into the organic phase, and the aqueous phase was extracted with ethyl acetate (3 x 30 mL). The organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, and the organic solvent was recovered by distillation under reduced pressure. The residu...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More