Detection of mycobacteria
a technology of mycobacteria and detection method, which is applied in the field of detection of mycobacteria, can solve the problems of limited antigen detection assays for direct diagnosis of tuberculosis, low detection thresholds, and inability to identify subjects with latent infection, and achieve the effect of low detection thresholds
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 2
Production of Murine Monoclonal Antibody
[0154] The monoclonal antibody designated as TBC27 was produced for use in the ELISA method of the present invention. This monoclonal was developed by immunization with M. tuberculosis alpha antigen obtained using the methods of Example 1. Six week-old female Balb / c mice (Charles River, Mass.) were immunized with 50 .mu.g alpha antigen in incomplete Freund's adjuvant (DIFCO, Detroit, Mich.). Twice, animals were boosted with additional 50 .mu.g of antigen at two week intervals, and sacrificed 2 weeks after the last boost. Spleen cells were fused to SP2 / 0 Ag 14 myeloma cells (ATCC) using polyethylene glycol (Sigma), and were propagated in Dulbecco's modified Eagle' medium (Sigma) with added hypoxanthine, aminopterin, and thymidine. Supernatants of wells with visible growth were tested for antibody by ELISA. The wells of Immulon 2 plates (PGC) were sensitized overnight (i.e., 12-18 hours), with 50 .mu.l M. tuberculosis filtrate (10 .mu.g / ml), and...
example 3
Detection of Alpha Antigen in M. tuberculosis Culture Filtrates
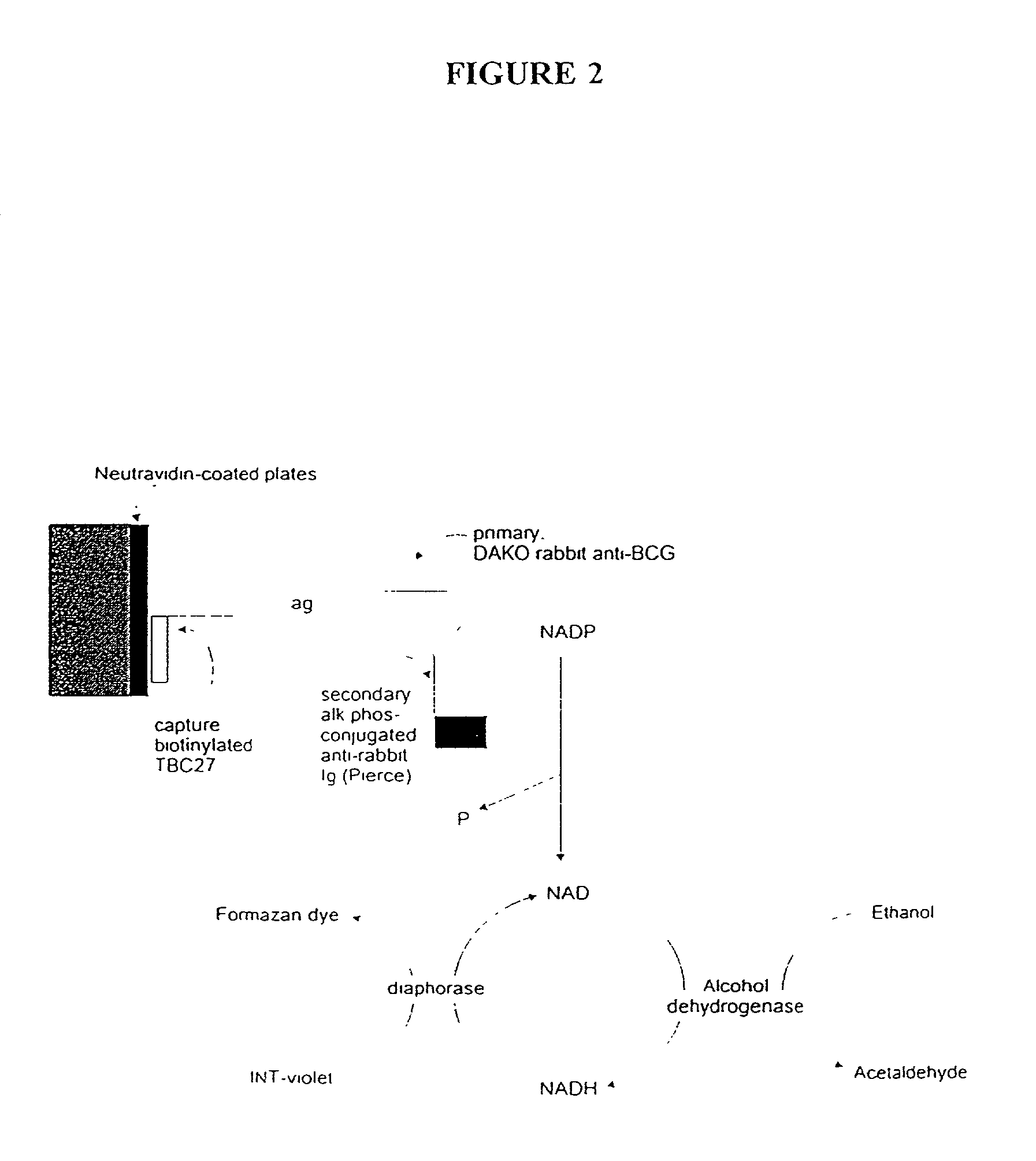
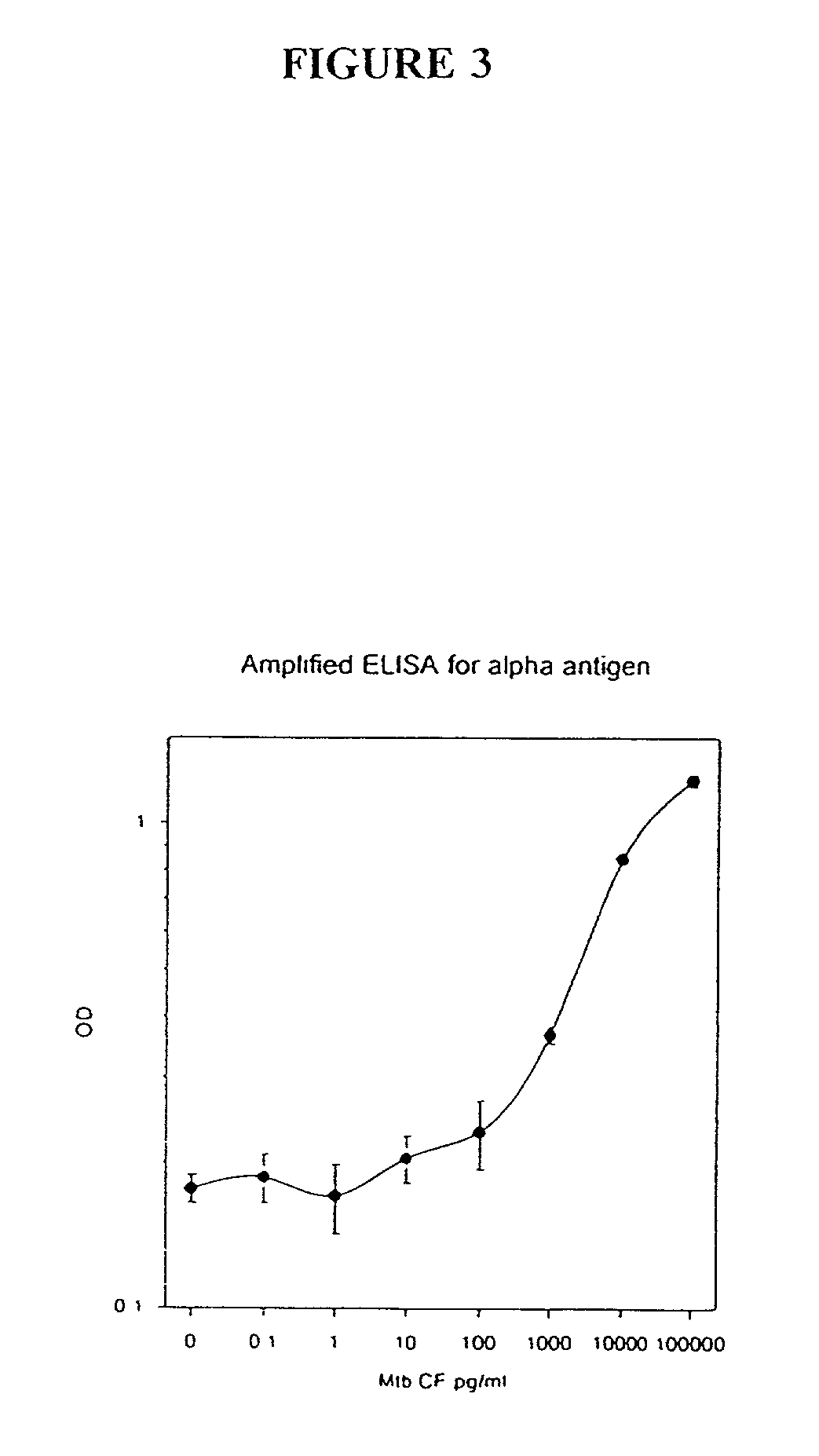
[0156] In this Example, an ELISA method (C. J. Stanley et al., "Enzyme amplification can enhance both the speed and the sensitivity of immunoassays," J. Immunol. Meth., 83:89-95 [1985]; and C. H. Self, "Enzyme amplification--a general method applied to provide an immunoassisted assay for placental alkaline phosphatase," J. Immunol. Meth., 76:389-93 [1985]) that substantially increases the speed and sensitivity of ELISA, was extensively modified for use for the detection of alpha antigen, including the addition of another step. In this method, an alkaline phosphatase conjugate was used to convert NADP to NAD, which then entered a regenerating cycle leading to production of a colored formazan dye. A schematic of the reaction is shown in FIG. 2. Its use in the alpha antigen assay has resulted in over a 1000 fold reduction in the lower limit of detection of the assay (i.e., from 20 ng / ml, to from 1 to 10 pg / ml). The samples ...
example 4
Recovery of M. tuberculosis Alpha Antigen in Spiked Samples
[0162] In this Example, the same methods as described in Example 3 were used for testing of serum or urine, with the exception that M. tuberculosis culture filtrates were added to normal pooled normal human serum or urine, as described above. This experiment was conducted in order to determine whether alpha antigen from M. tuberculosis filtrate could be detected in such samples. The results of this experiment are shown in FIG. 4. As can be seen from FIG. 4, the sensitivity of the assay was not substantially affected by either diluent (i.e., normal serum or urine). However, the study suggests that separate diluents for standards be used, depending upon the type of clinical sample.
PUM
| Property | Measurement | Unit |
|---|---|---|
| volume | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| power | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More