Fluorescent proteins from aquatic species
a technology of fluorescence proteins and aquatic species, which is applied in the field of fluorescence proteins, can solve the problems of affecting the utility of a protein in research, accumulating protein can be toxic to some mammalian cells, and high levels of gfp expression are particularly toxic, so as to improve fluorescence, enhance the effect of fluorescence and reduce toxicity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Species Collection and Animal Husbandry
[0069] Coral colonies were obtained from two sources. First, a solid, brick-red colony consisting of several Actinodiscus / Discosoma sp. polyps was obtained from a local aquarium store. Second, Montastraea cavernosa colonies were collected from reefs in the South Florida area. Both species were maintained in small aquariums with flow-through, filtered sea water.
example 2
Isolation of Total RNA, Reverse Transcription, Amplification and Recovery of cDNAs
[0070] RNA was isolated using the Totally RNA kit (Catalog #1902, Ambion, Inc., Austin, Tex.) from a single polyp (Actinodiscus / Discosoma sp.) or from a cellular mass lightly airbrushed from the underlying skeleton (M. cavernosa). Briefly, the tissue was homogenized and mixed by tube inversion with approximately 10 volumes of denaturation solution and extracted with 1 volume of phenol / chloroform. The aqueous supernatant was transferred to a clean vessel. One-tenth volume of 3M sodium acetate was added to the supernatant, which was then extracted an additional time with 1 volume of acid-phenol / chloroform. Again, the aqueous supernatant was transferred to a clean vessel. One volume of isopropanol was added to the supernatant, and total RNA was precipitated by centrifugation. The pellet was washed with 75% ethanol and resuspended in 50 uL RNAse-free water. Yield was measured with a spectrophotometer, and...
example 3
Transformation and Selection of Bacterial clones, Plasmid Preparation, and DNA Sequencing of Wild-Type Fluorescent Proteins
[0074] The gel purified cDNA fraction was ligated into the pCR II cloning vector (Invitrogen, Carlsbad, Calif.), with resultant plasmids electrotransformed into Top 10 E. coli (Invitrogen). Transformed bacteria were grown on LB-ampicillin (100 μg / ml) plates and colonies were screened for fluorescence using a Leica MZFLIII fluorescence stereo dissection microscope. Single fluorescent colonies were picked, restreaked for several rounds to resolve mosaicism, and grown in liquid culture. Plasmid DNA was prepared using the Qiagen Midi kit (Qiagen, Valencia, Calif.).
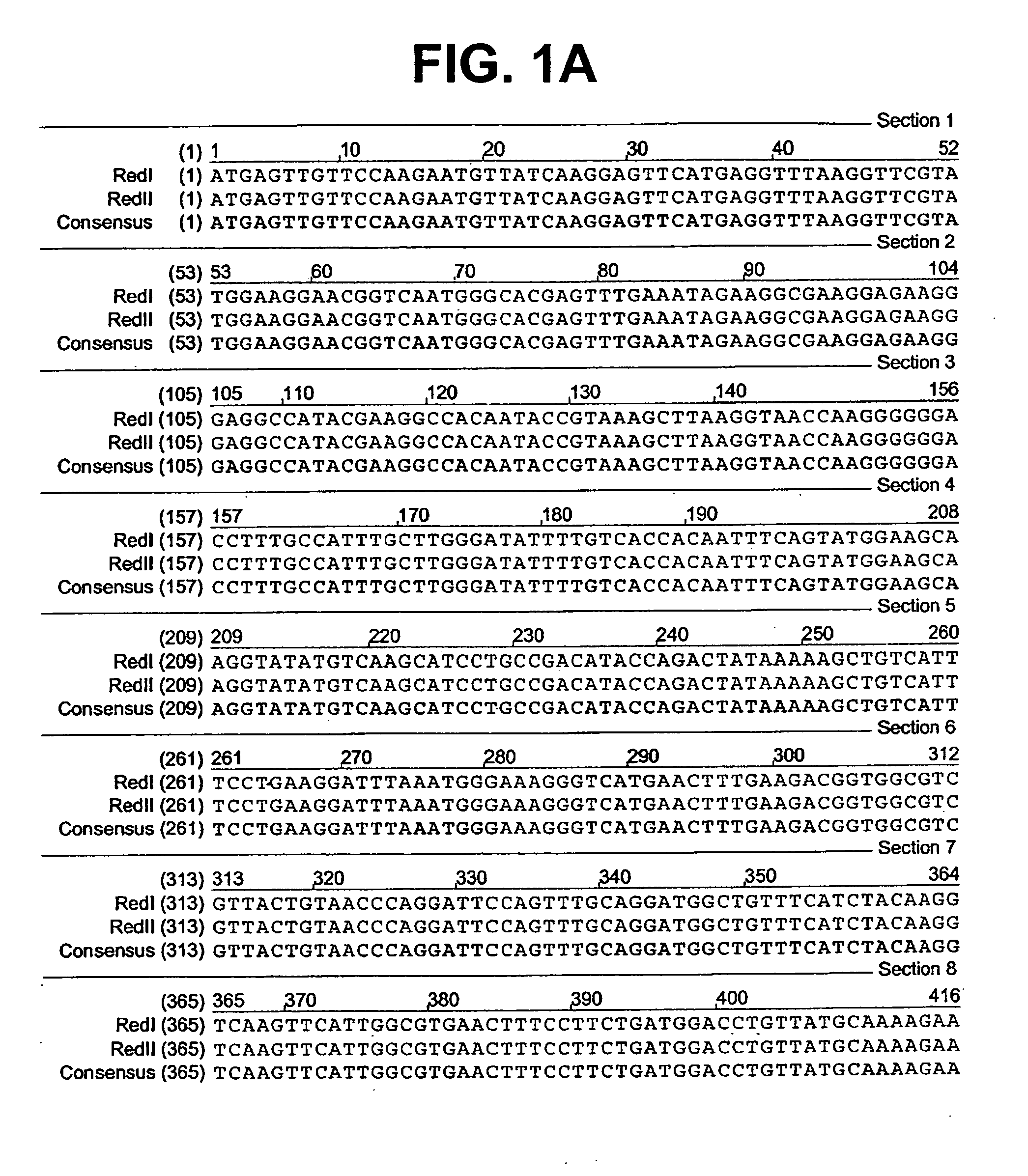
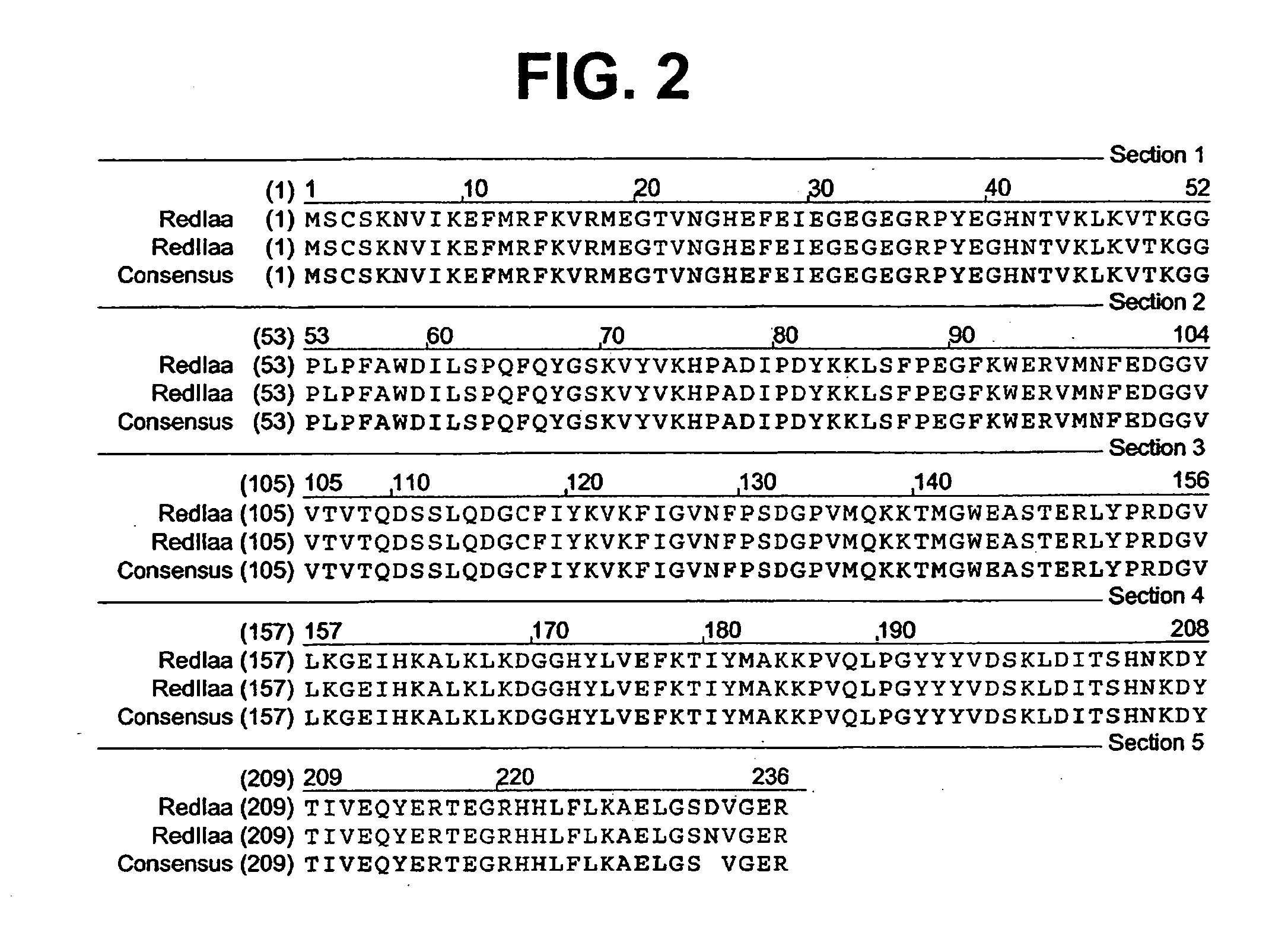
[0075] A colony expressing a wild-type red fluorescent protein (RFP) was isolated from the bacterial colonies generated from the Actinodiscus / Discosoma sp. 1 RNA. This is herein referred to as Ac / DsRFP.
[0076] In addition, a colony expressing a wild-type green fluorescent protein (GFP) was isolated from ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| optical property | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More