Substituted phenethylamines with serotoninergic and/or norepinephrinergic activity
a technology phenethylamine, which is applied in the direction of amine active ingredients, drug compositions, peptides, etc., can solve the problems of venlafaxine discontinuation symptoms, short half-life of venlafaxine relative to the majority of serotonin reuptake inhibitors, and high risk of sri discontinuation symptoms, etc., to achieve a more reproducible dosing effect, high crystal
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
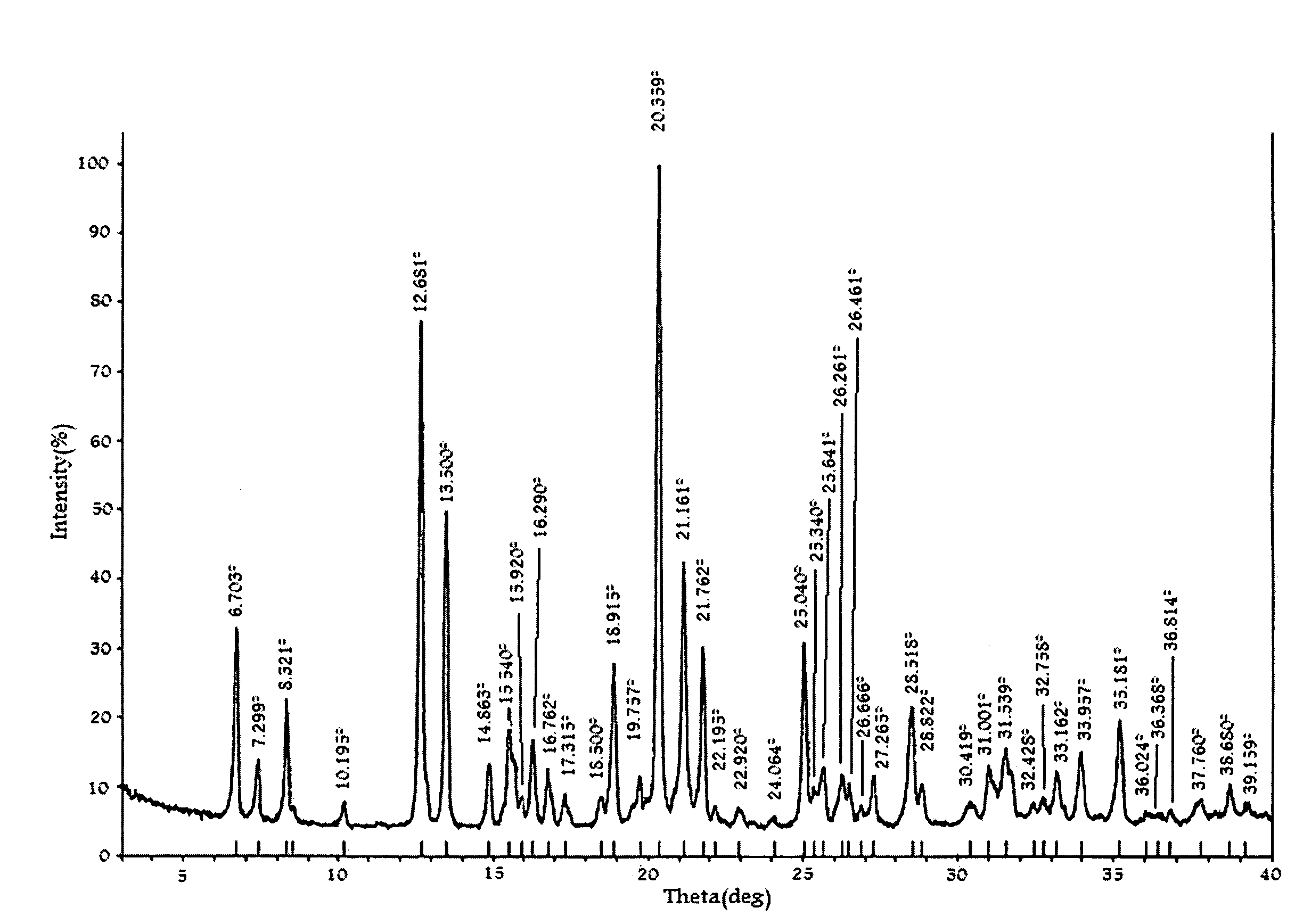
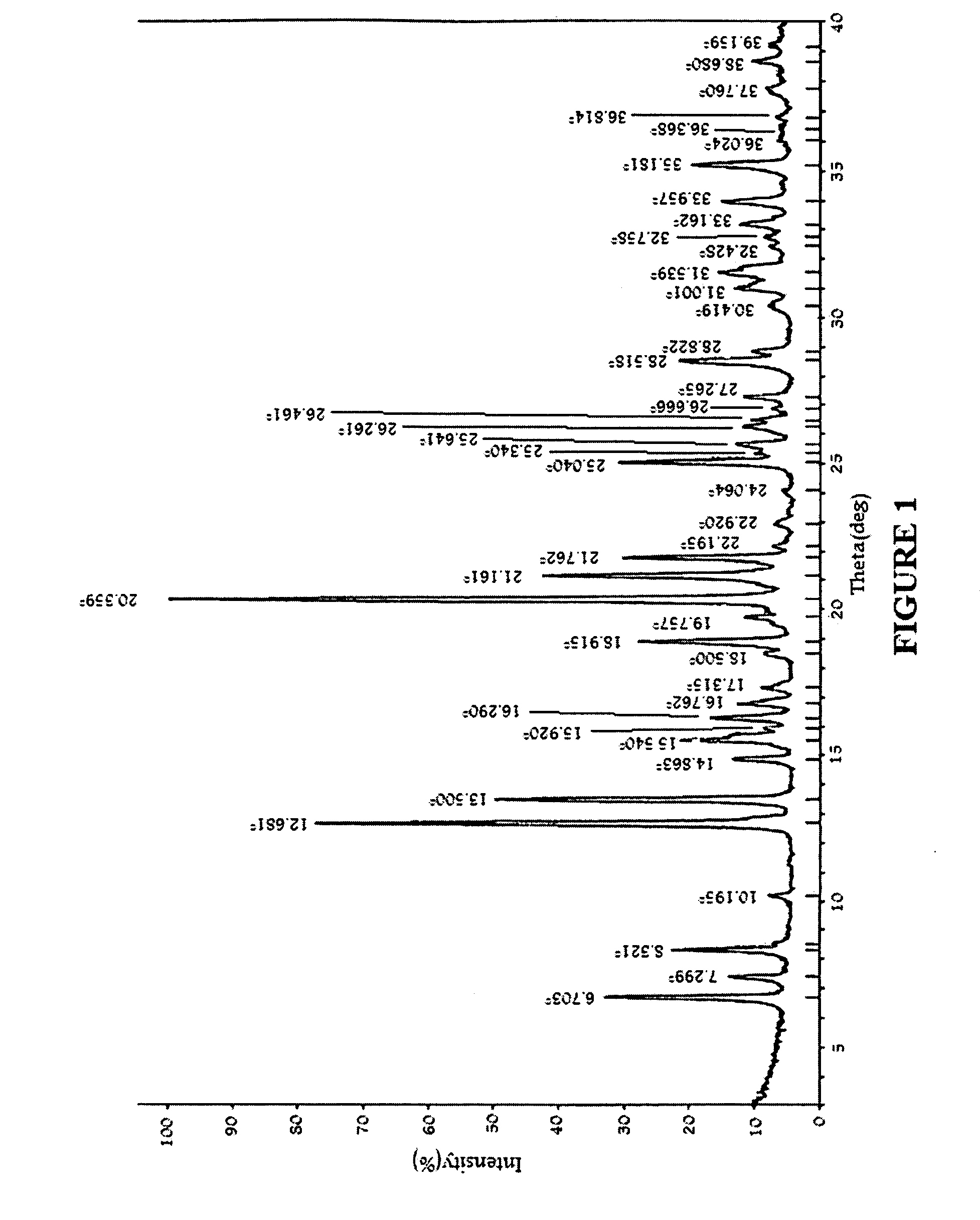
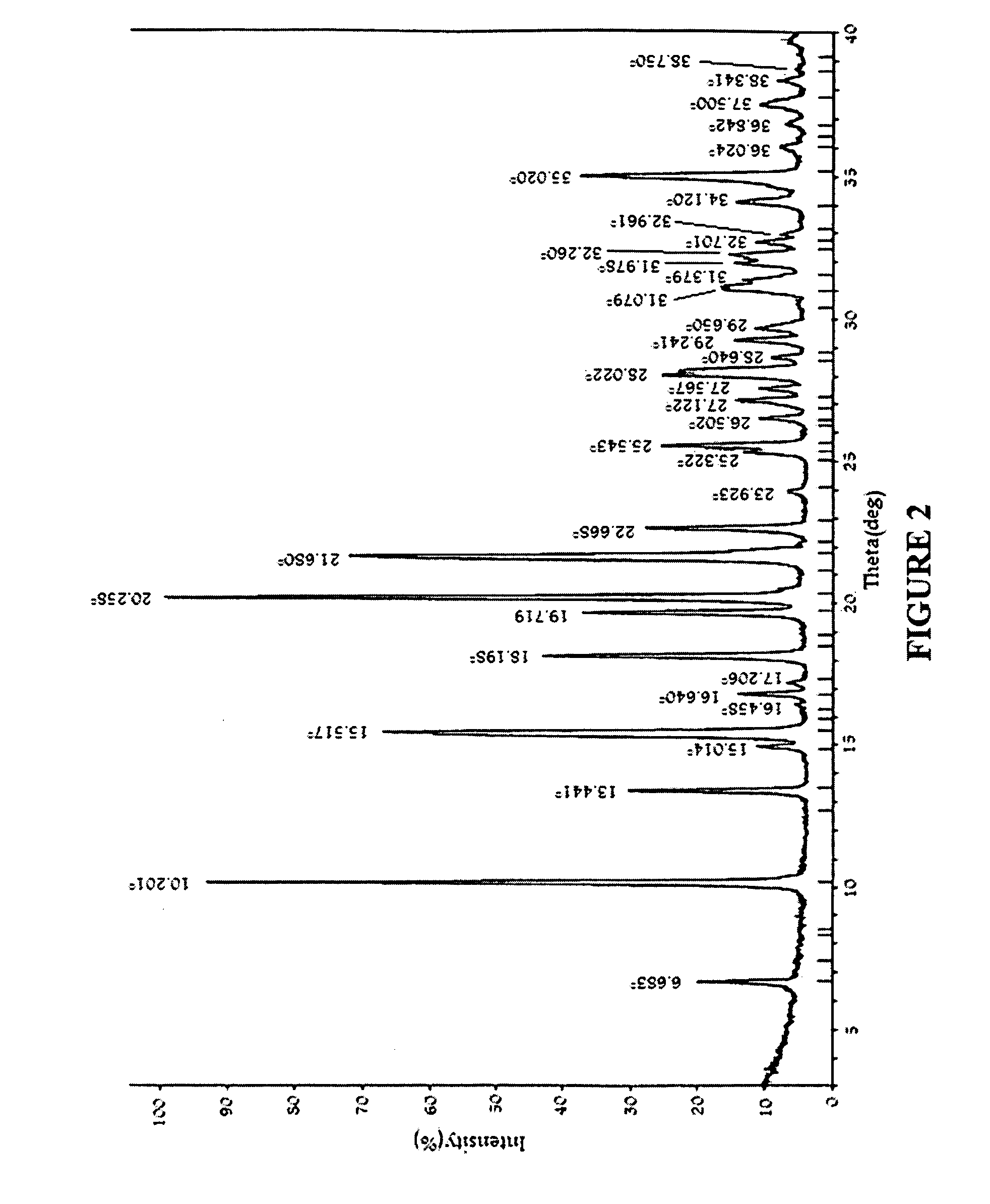
Image
Examples
example 1
d9-2-(4-Methoxyphenyl)-acetic Acid
[0351]
[0352]d9-(4-Methoxyphenyl)-acetic acid can be prepared according to known literature procedures Ouk et al., Green Chemistry, 2002, 4(5), 431-435, which is hereby incorporated by reference in its entirety, by reacting d6-(4-hydroxyphenyl)-acetic acid (1 equiv, Cambridge Isotopes Laboratories), K2CO3 (0.04 equiv) and d6-carbonic acid dimethyl ester (1.25 equiv, Cambridge Isotopes Laboratories) at 160° C. until completion.
Example 2
d15-2-(4-Methoxyphenyl)-N,N-dimethyl-acetamide
[0353]
[0354]The title compound is prepared according to the procedure described in Yardley et al, Journal of Medicinal Chemistry 1990, 33(10), 2899-2905, which is hereby incorporated by reference in its entirety. A solution of d9-(4-methoxyphenyl)-acetic acid (1 equiv) in methylene chloride is treated with oxalyl chloride (1.22 equiv) and DMF (catalytic amount) and then stirred at room temperature until all acid is converted to the acid chloride. The solvent is removed under...
example 3
d24-2-(1-Hydroxycyclohexyl)-2-(4-methoxyphenyl)-N,N-dimethylacetamide
[0355]
[0356]The title compound is prepared according to the procedure described in Yardley et al., Journal of Medicinal Chemistry 1990, 33(10), 2899-2905, which is hereby incorporated by reference in its entirety. A solution of d15-2-(4-methoxyphenyl)-N,N-dimethyl-acetamide (1 equiv) in THF is treated with n-butyllithium (1 equiv) at −78° C. The mixture is stirred for 90 minutes at −78° C.; a THF solution of d10-cyclohexanone (1.2 equiv, Sigma-Aldrich) is added, and stirring is maintained until completion. The reaction is quenched by addition of D2O (2 equiv), the mixture is warmed to room temperature and the solvent is removed under reduced pressure and the crude residue is purified by silica gel column chromatography.
example 4
d26-1-[2-Dimethylamino-1-(4-methoxyphenyl)-ethyl]-cyclohexanol
[0357]
[0358]The title compound is prepared according to the procedure described in Yardley et al., Journal of Medicinal Chemistry 1990, 33(10), 2899-2905, which is hereby incorporated by reference in its entirety. d24-2-(1-Hydroxycyclohexyl)-2-(4-methoxyphenyl)-N,N-dimethyl-acetamide (1 equiv) in THF is added dropwise to a mixture of lithium aluminum deuteride (1.6 equiv) at 0° C. and stirred until completion. The reaction is quenched with D2O, and worked up under standard conditions known to one skilled in the art. The mixture is then filtered and the precipitate is washed several times with THF. The combined filtrates are evaporated, and the residue is recrystallized from a suitable solvent.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More